The Association for Molecular Pathology (AMP), the premier global, molecular diagnostic professional society, will host a free webinar on the US Food and Drug Administration (FDA) final rule that seeks to regulate laboratory developed tests (LDTs) as medical devices.
Tag: Testing
This Alloy is Kinky
A team led by Berkeley Lab has revealed a new metal alloy that resists damage at both hot and cold temperature extremes due to an atomic-level effect called kink bands, making it potentially suitable for demanding applications like more powerful aerospace engines.
MSU expert: What to know about flu, RSV, new COVID-19 variant and new vaccine
The new COVID-19 EG.5 variant is responsible for around 22% of current cases. The World Health Organization has classified it as a “variant of interest,” meaning countries should monitor it more closely than other strains — and cases have only increased in the past few weeks. Peter Gulick is an expert on infectious diseases, and he provides insight on what this new variant could mean and what you should know.
‘Placenta-on-a-chip’ Mimics Malaria-infected Nutrient Exchange between Mother-Fetus
Combining microbiology with engineering technologies, this novel 3D model uses a single microfluidic chip to study the complicated processes that take place in malaria-infected placenta as well as other placenta-related diseases and pathologies. The technology supports formation of microengineered placental barriers and mimics blood circulations, which provides alternative approaches for testing and screening.
New Low-cost Device Rapidly, Accurately Detects Hepatitis C Infection
The entire virus detection process is executed inside a uniquely designed, portable, inexpensive, disposable, and self-driven microfluidic chip. The fully automated sample-in–answer-out molecular diagnostic set-up rapidly detects Hepatitis C virus in about 45 minutes and uses relatively inexpensive and reusable equipment costing about $50 for sample processing and disease detection. The disposable microfluidic chip also offers shorter times for a reliable diagnosis and costs about $2.
UCLA launches effort to expand its COVID test to detect other emerging viruses
UCLA Health is joining a new effort to increase the ability to quickly respond to emerging pathogens by creating the capability to detect all existing and new respiratory RNA viruses in a single test.
Study Examines Data Transparency, Health Equity in U.S. COVID-19 Response
State governments varied widely in COVID-19 pandemic mitigation measures and how they addressed immediate and long-standing health disparities and associated inequities.
New Tool Assesses How Well People Read Kids’ Emotions
Researchers have developed and validated a tool for assessing how accurate people are at recognizing emotion in elementary school-aged children. The technique facilitates research on understanding emotions of children – and shows that adults are often wrong when assessing the emotions of children.
Important signs of blood clots in the lungs missed in patients with dementia
Patients with dementia who had signs and risk factors of a pulmonary embolism, or a blood clot in the lungs, were much less likely to be tested for pulmonary embolism than patients without dementia who had the same signs and risk factors.
Inflammatory Proteins Help Better Diagnose Progression of Alzheimer’s Disease
Testing for some inflammatory proteins associated with the nervous and immune systems will help diagnose the earlier onset and progression of Alzheimer’s disease, according to a Rutgers study.
Connect Chicago Initiative Expands Community COVID-19 Testing
As COVID-19 cases rise in the U.S., Connect Chicago — new initiative between the Chicago Department of Public Health, Rush University Medical Center, and Esperanza Health Centers — is aiming to redouble testing efforts in Chicago communities that need it most.
Early CHIS 2021 Data Estimates Show how Californians Dealt With COVID-19
With California state vaccination rates slowing, and guidelines on mask wearing and social gatherings changing, the UCLA Center for Health Policy Research (CHPR) has released new data from the 2021 California Health Interview Survey (CHIS) that sheds light on Californians’ views on getting the vaccine and following suggested safety protocols.
New partnership to develop a new ultra-fast test for COVID-19 antibodies
The NanoSpot.AI test is estimated to be significantly less expensive to manufacture than other SARS-CoV-2 antibody tests and has the potential to be more affordable than currently available tests, making it possible to extend the test to every corner of the world. Clinical studies validating NanoSpot.AI are currently underway.
Tennessee-Based Veteran-Owned Start-Up to Develop New COVID-19 Screening Method
DHS S&T awarded $199,653 in Phase 1 funding to Farmspace Systems, LLC, a veteran-owned start-up based in Alamo, Tenn., to enhance its COVID Finder technology, a non-thermal detection COVID-19 screening method.
Atlantic Health System introduces rapid test for COVID-19 variants
Atlantic Health System is among the first hospitals in the region to be able to rapidly screen for key viral variants, a key advance for public health amid the pandemic.
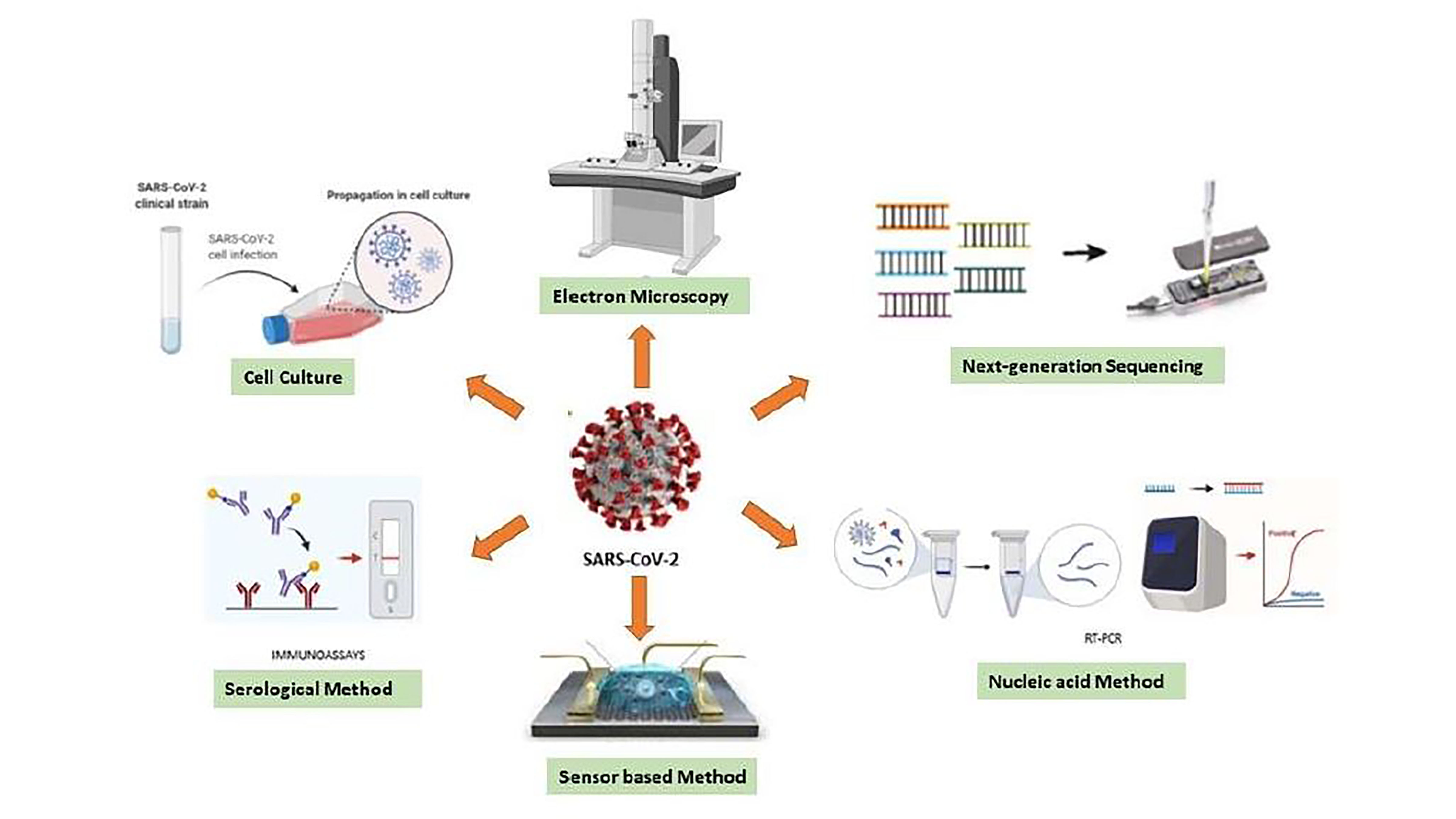
Out of Many COVID-19 Tests, Which One to Choose?
Curbing the coronavirus pandemic relies heavily on how quickly a potentially exposed individual can be tested and quarantined. However, the current diagnostic techniques vary in reliability and relevance, so an understanding of which test is most appropriate for a given circumstance is necessary to avoid false reports. Researchers evaluated the available diagnostic techniques and determined key steps required for better testing moving forward. They present their findings in the journal APL Bioengineering.
Arizona State University releases first comprehensive survey on how companies are protecting their employees from COVID-19
A new global business survey conducted by the College of Health Solutions at Arizona State University (ASU) and the World Economic Forum (WEF), with support from The Rockefeller Foundation, finds that less than 20% of employers report testing their workers for COVID-19, and 35% have permanently reduced their workforce. The survey, which was completed by 1,125 employers from 29 countries with the majority over a period of six weeks, September to October, found that for companies with employees onsite at the workplace, many are taking some steps to reduce the risk of spreading the virus. Nearly three-fourths (74%) of these companies report they require masks for their employees, and nearly 80% make masks and hand sanitizer available.

Rush University Medical Center Surpasses 100,000 COVID-19 Tests
Throughout the COVID-19 pandemic, Rush University Medical Center has been a leader in the Chicago area in developing, expanding and evolving testing efforts. On Friday, Nov. 13, the Medical Center performed its 100,000th test for COVID-19.
Where COVID-19 tests fail: Researcher ready to comment.
MOSCOW (MIPT) — As countries like Great Britain consider introducing testing regimes for international arrivals to ease travel restrictions, even as the media continue to cite coronavirus infection statistics by the hour, more people start wondering about the limitations of COVID-19 test…
UC San Diego Partners with San Ysidro Health to Expand COVID-19 Testing
Researchers at University of California San Diego School of Medicine, in collaboration with local partners, have been awarded a $5 million grant from the National Institutes of Health (NIH) to implement a program of widespread testing for COVID-19 in San Ysidro, focused on pregnant women and children.
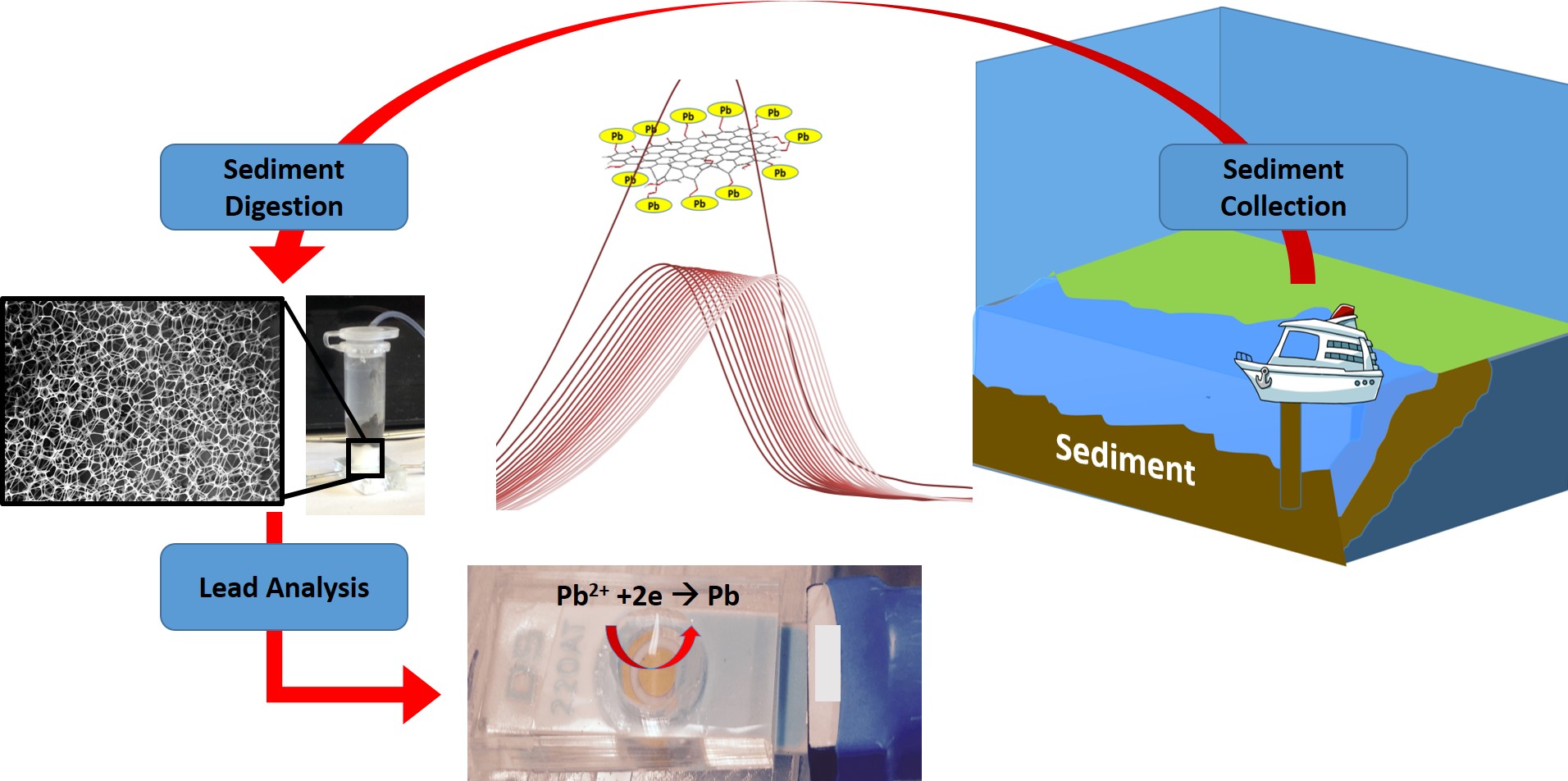
New Device Can Measure Toxic Lead Within Minutes
Rutgers researchers have created a miniature device for measuring trace levels of toxic lead in sediments at the bottom of harbors, rivers and other waterways within minutes – far faster than currently available laboratory-based tests, which take days. The affordable lab-on-a-chip device could also allow municipalities, water companies, universities, K-12 schools, daycares and homeowners to easily and swiftly test their water supplies. The research is published in the IEEE Sensors Journal.
Gaps in early surveillance of coronavirus led to record-breaking US trajectory
Research from the University of Notre Dame provides insight into how limited testing and gaps in surveillance during the initial phase of the epidemic resulted in so many cases going undetected.
FDA approval of rapid saliva test could be a game changer for COVID-19 testing, says expert
Emergency authorization by the Food and Drug Administration to use a new and inexpensive rapid saliva test for COVID-19 could be a game changer for tracking and slowing the spread of the virus, says Virginia Tech epidemiologist Charlotte Baker. Quoting…
U.S. COVID-19 Testing Too Slow, Survey Says
Rutgers scholar Katherine Ognyanova is available to comment on the latest Rutgers-Harvard-Northeastern-Northwestern survey data on waiting times for diagnostic tests across the United States from The COVID-19 Consortium for Understanding the Public’s Policy Preferences Across States. The researchers surveyed 19,058 people…

UC Davis Health tests monoclonal antibodies as potential COVID-19 treatment
Timothy Albertson has received a BARDA grant to lead a new clinical trial at UC Davis Health. The trial evaluates the efficacy, safety and tolerability of an antibody cocktail in hospitalized adult patients with COVID-19.
Removing CDC from tracking COVID-19 outbreaks poses risk to public health, says expert
With cases of the coronavirus spiking in many areas nationwide, public health officials caution that funneling COVID-19 hospitalization data to the HHS rather than the Centers for Disease Control and Prevention (CDC) could slow efforts to end the coronavirus pandemic.…

Asymptomatic Testing Central to UC San Diego’s Return to Learn for Fall Quarter
The University of California San Diego today announced the next step in its Return to Learn program, which will guide an incremental repopulation of the campus while offering broad, asymptomatic testing for faculty, staff and students on a recurring basis to detect the presence of SARS-CoV-2, the novel coronavirus that causes COVID-19.

FAU Now Offers COVID-19 Contact Tracing and Risk-Reduction Public Health Certificate
In response to the high demand for one of the fastest-growing jobs in the U.S., FAU has launched a new, online public health certificate course on COVID-19, contact tracing and risk-reduction. The five-week, 15-hour course does not require a college degree and is scheduled from June 29 to Aug. 7. The program is open to the general public for adults age 18 and older with a high school diploma or equivalent and a variety of work experiences and educational backgrounds.

COVID-19 Test That Relies on Viral Genetic Material Gives False Negative Results if Used Too Early in Those Infected
In a new study, Johns Hopkins researchers found that testing people for SARS-CoV-2 — the virus that causes COVID-19 — too early in the course of infection is likely to result in a false negative test, even though they may eventually test positive for the virus.

LLNL providing critical assistance in addressing national swab shortage for COVID-19 testing
To address the nationwide shortage of specialized nasal swabs used for COVID-19 diagnoses, Lawrence Livermore National Laboratory engineers formed an ad hoc, rapid response team that tested more than a dozen novel, 3D-printed swab designs from a grassroots coalition of commercial and academic partners. The mechanical tests performed at the Lab provided valuable feedback that improved the designs, enabling them to meet requirements for COVID-19 testing. The Lab’s work on swabs is continuing with funding from the U.S. Department of Energy (DOE) and expanding into other 3D-printed components for COVID-19 test kits.
Mount Sinai Health System and Renalytix Form Joint Venture, Kantaro Biosciences, To Develop and Scale Production of COVID Antibody Test Kits
– Kantaro Biosciences partners with Bio-Techne for manufacturing and global kit distribution
– Scaled kit production to enable clinical laboratories to conduct 10M tests per month is planned to begin in July

McMaster chemists develop foolproof new test to track the fats we eat
A team of researchers at McMaster University has developed a reliable and accurate blood test to track individual fat intake, a tool that could guide public health policy on healthy eating.

FDA Approves First At-Home Saliva Collection Test for Coronavirus
Rutgers’ RUCDR Infinite Biologics received an amended emergency use authorization from the FDA late Thursday for the first SARS-CoV-2 coronavirus test that will allow people to collect their own saliva at home and send to a lab for results. The decision follows the FDA’s recent emergency approval to RUCDR Infinite Biologics for the first saliva-based test, which involves health care workers collecting saliva from individuals at testing sites.

A New Way to Accurately Estimate COVID-19 Death Toll
A Rutgers engineer has created a mathematical model that accurately estimates the death toll linked to the COVID-19 pandemic in the United States and could be used around the world. The model, detailed in a study published in the journal Mathematics, predicted the death toll would eventually reach about 68,120 in the United States as a result of the SARS-CoV-2 coronavirus that causes COVID-19. That’s based on data available on April 28, and there was high confidence (99 percent) the expected death toll would be between 66,055 and 70,304.
Occupational Medicine Physicians Help Businesses Safely Return Employees to Work
The American College of Occupational and Environmental Medicine has launched a campaign to inform businesses and the public about the importance of returning to work safely and the specific role that occupational medicine physicians can and should play in this process.
UCLA Biodesign Fellow Focuses Surgical, Medical and Biomedical Engineering Insights on a Weak Link in COVID-19 Testing: Swabs Shortage
When the COVID-19 outbreak became a pandemic, and the scarcity of testing supplies became a crisis, the leaders of UCLA Biodesign saw the experience and background of Gabriel Oland, MD, as the ideal combination to help reinforce one link in the strained supply chain: the nasopharyngeal swabs used to collect patient specimens for testing.

Working behind the scenes: UAB pathologists play key role in fighting coronavirus pandemic
Testing has been a major cause for concern worldwide ever since the pandemic began, but clinicians and researchers with UAB’s Department of Pathology have been working around the clock to make testing available for as many people as possible, making sure accurate results are available in a timely manner.
BIDMC-led clinical trial identifies four novel 3D-printed swabs for use in COVID-19 testing
A clinical trial conducted by a multi-disciplinary team from Beth Israel Deaconess Medical Center has identified four novel prototypes of 3D-printed swabs that can be used for COVID-19 testing.
Coriell Institute for Medical Research Fills Gaps in COVID-19 Testing Materials Supply
Answering the growing need for additional testing, and in direct response to shortages encountered in testing kit supply pipelines, the Coriell Institute for Medical Research is now offering services to create viral testing collection tubes containing different media and/or buffer formulations, for use in COVID-19 testing kits. Coriell is also exploring the feasibility of saliva as a means for testing for the new coronavirus.

New Rutgers Saliva Test for Coronavirus Gets FDA Approval
The FDA has granted emergency use authorization (EUA) to Rutgers’ RUCDR Infinite Biologics and its collaborators for a new collection approach that utilizes saliva as the primary test biomaterial for the SARS-CoV-2 coronavirus, the first such approval granted by the federal agency. The new saliva collection method, which RUCDR developed in partnership with Spectrum Solutions and Accurate Diagnostic Labs (ADL), will allow for broader population screening than the current method of nose and throat swabs.

Data Visualization Tool Examines Community Factors Underlying COVID-19 Outcomes
A new data visualization tool examines how and why COVID-19 impacts regions differently. Using daily updated data, COVIDMinder compares community risks, mediation tools, and outcomes related to COVID-19 by state across the United States, and by county within New York state.

How Old are Whale Sharks? Nuclear Bomb Legacy Reveals Their Age
Nuclear bomb tests during the Cold War in the 1950s and 1960s have helped scientists accurately estimate the age of whale sharks, the biggest fish in the seas, according to a Rutgers-led study. It’s the first time the age of this majestic species has been verified. One whale shark was an estimated 50 years old when it died, making it the oldest known of its kind. Another shark was an estimated 35 years old.

Tulane School of Medicine launches COVID-19 testing lab to improve capacity
The test is another way that Tulane is working to ease the testing crisis and stop the spread of COVID-19, which has hit the New Orleans area especially hard.
Rutgers Launches Genetic Testing Service for New Coronavirus
Rutgers’ RUCDR Infinite Biologics has launched a test for the SARS-CoV-2 coronavirus and is using its automation experience and infrastructure to test as many as tens of thousands of samples daily. RUCDR has also submitted an emergency use authorization request for a saliva collection method that will allow for broader population screening.

New COVID-19 test gives results in four hours
A laboratory at Tulane Medical Center is conducting a new test for COVID-19 that can yield results within four hours.
Mount Sinai Developing an “End-to-End” Diagnostics Solution for COVID-19 That Incorporates Diagnosis, Treatment Selection, and Monitoring of Disease Course
An expert team of researchers and clinicians have been working together to design, validate, and implement an “end-to-end” clinical pathology laboratory solution that will allow for the testing of approximately several hundred people per day in order to rapidly diagnose and help guide the selection of treatment and monitor disease course.
Rutgers Expert Available to Discuss Importance of Accuracy of Coronavirus Testing
A Rutgers University population health epidemiologist is available to comment on the importance of understanding the specificity, sensitivity, and the positive and negative predictive values of the accuracy and precision of Coronavirus testing, and how that knowledge may impact our…
New Software Tests Asphalt Performance More Efficiently
New Brunswick, N.J. (Feb. 26, 2020) – Rutgers University–New Brunswick researchers have created a software tool that more efficiently analyzes how asphalt performs, saving transportation agencies time and money. As performance testing for asphalt pavement has evolved, the focus has shifted…

Validating NIH Toolbox to help evaluate cognitive processing in people with intellectual disability
Cognitive processing in people with intellectual disability can now be accurately assessed thanks to UC Davis Health researchers who updated and validated series of tests, part of the NIH Toolbox Cognitive Battery.
LLNL develops 3D ‘brain-on-a-chip’ device capable of long-term recording of neural activity
Lawrence Livermore National Laboratory engineers and biologists have developed a “brain-on-a-chip” device capable of recording the neural activity of living brain cell cultures in three-dimensions, a significant advancement in the realistic modeling of the human brain outside of the body.