A diagnostic test, first offered in the United States at University Hospitals Harrington Heart & Vascular Institute, has now shown through a clinical study to significantly decrease cardiovascular mortality, reduce additional non-invasive heart testing, and increase cath lab efficiency.
Tag: Diagnostic Testing
‘Just breathe’: Missouri S&T researchers ready for clinical trials for diagnostic instrument
When an ill person schedules a doctor visit, blood work or other diagnostic testing is often required before a diagnosis is determined. Missouri University of Science and Technology electrical engineering researcher Dr. Jie Huang is developing technology that would allow patients to “just breathe” and avoid that testing.
AACC Releases Guidance on Procalcitonin Testing to Improve Care for Patients with Bacterial Infections
AACC has issued expert guidance on clinical testing for procalcitonin, a blood marker that helps detect serious bacterial infections and sepsis. By giving clinicians and laboratory professionals much-needed clarity on how to best use procalcitonin tests, the new guidelines could improve the treatment of critically ill patients and those with certain lower respiratory infections.
Breaking Research That Could Improve Cardiac Care for Children Published in AACC’s The Journal of Applied Laboratory Medicine
A first-of-its-kind study has established pediatric reference intervals for two common tests for cardiovascular disease. Published in AACC’s The Journal of Applied Laboratory Medicine, these findings are crucial to advancing diagnosis and treatment of heart conditions in children.
A Novel Test Could Make Sure Newborns With a Serious Genetic Disease Get Essential Treatment
Scientists have developed a test that could greatly improve quality of life for infants with homocystinuria (HCU)—a congenital disease that, if not treated early, causes serious complications. Research demonstrating the efficacy of this test was published today in AACC’s Clinical Chemistry journal.
ORTHO RANKS NO.1 IN CUSTOMER SATISFACTION FOR 5 YEARS IN A ROW
QuidelOrtho Corporation (NASDAQ: QDEL) (“QuidelOrtho”) announced today that its subsidiary, Ortho Clinical Diagnostics, a leading provider of in vitro diagnostics solutions and services to hospitals, clinical laboratories and blood banks around the world, has earned the No. 1 ranking in the diagnostics industry for Overall Customer Satisfaction in the 2022 IMV ServiceTrak™ Awards for the fifth consecutive year. The ranking is based on interviews conducted with laboratory professionals, including Ortho’s customers, from more than 1,200 clinical testing locations operating over 1,900 collective instruments. The prestigious annual IMV ServiceTrak™ Awards rankings affirm the continued power and impact of Ortho’s innovative Ortho Care® Services and Informatics platform.
Rapid, reliable test for COVID and other infections, created by McMaster researchers, moves toward marketplace
McMaster University researchers behind a new form of rapid, accurate and portable diagnostic test are moving their work toward the marketplace.
Expanding diagnostics business: SCHOTT signs agreement to acquire Applied Microarrays Inc.
SCHOTT MINIFAB, a subsidiary of SCHOTT that develops and manufactures microfluidic devices for point-of-care and life sciences consumables, has had a long-standing close customer relationship with Applied Microarrays Inc. (AMI). Together, they develop biotech substrates for diagnostics applications.
No lab required: New technology can diagnose infections in minutes
Engineering, biochemistry and medical researchers from McMaster University have created a hand-held rapid test for bacterial infections that can produce accurate, reliable results in less than an hour, eliminating the need to send samples to a lab.
New wiki on salivary proteins may transform diagnostic testing and personalized medicine
To improve the development of new saliva-based diagnostic tests and personalized medicine, the National Institute of Dental and Craniofacial Research (NIDCR) has supported the development of the Human Salivary Proteome Wiki, the first public platform that catalogs and curates data on each of the thousands of proteins within our saliva.
Grant accelerates McMaster University, SQI Diagnostics effort to move infection testing innovation from lab to market
A new grant is helping McMaster University engineers and a Toronto precision-medicine diagnostics company to get infection-testing technology to market while generating opportunities for students.
Experience “Innovation Uninterrupted” with Sysmex America at AACC 2020
Sysmex America, Inc., a leading diagnostic solutions company offering hematology, urinalysis, information systems and flow cytometry testing technology for optimal clinical laboratory performance, will be exhibiting virtually at the 72nd AACC Annual Scientific Meeting & Clinical Lab Expo, December 14 – 17.
Advances in COVID-19 Testing, Artificial Intelligence, Cancer Therapies, and The Future of Precision Medicine to Be Explored at The All-Virtual 2020 AACC Annual Scientific Meeting
In the wake of the novel coronavirus pandemic, the need for reliable, accurate, and accessible laboratory testing is more evident than ever before. At the 2020 AACC Annual Scientific Meeting & Clinical Lab Expo, laboratory medicine experts will present the cutting-edge research and technology that is revolutionizing clinical testing and patient care for COVID-19 and across the spectrum of healthcare.
Baebies Selected as Finalist for the 2020 AACC Disruptive Technology Award
Baebies has been named a finalist for the 2020 American Association for Clinical Chemistry’s (AACC) Disruptive Technology Award for FINDER®, a near-patient testing platform. As a growth-stage company developing diagnostic products to provide a healthy start for children everywhere, Baebies FINDER tests for diseases from low blood volume (50 µL) with a turn-around time of approximately 15 minutes after sample introduction.
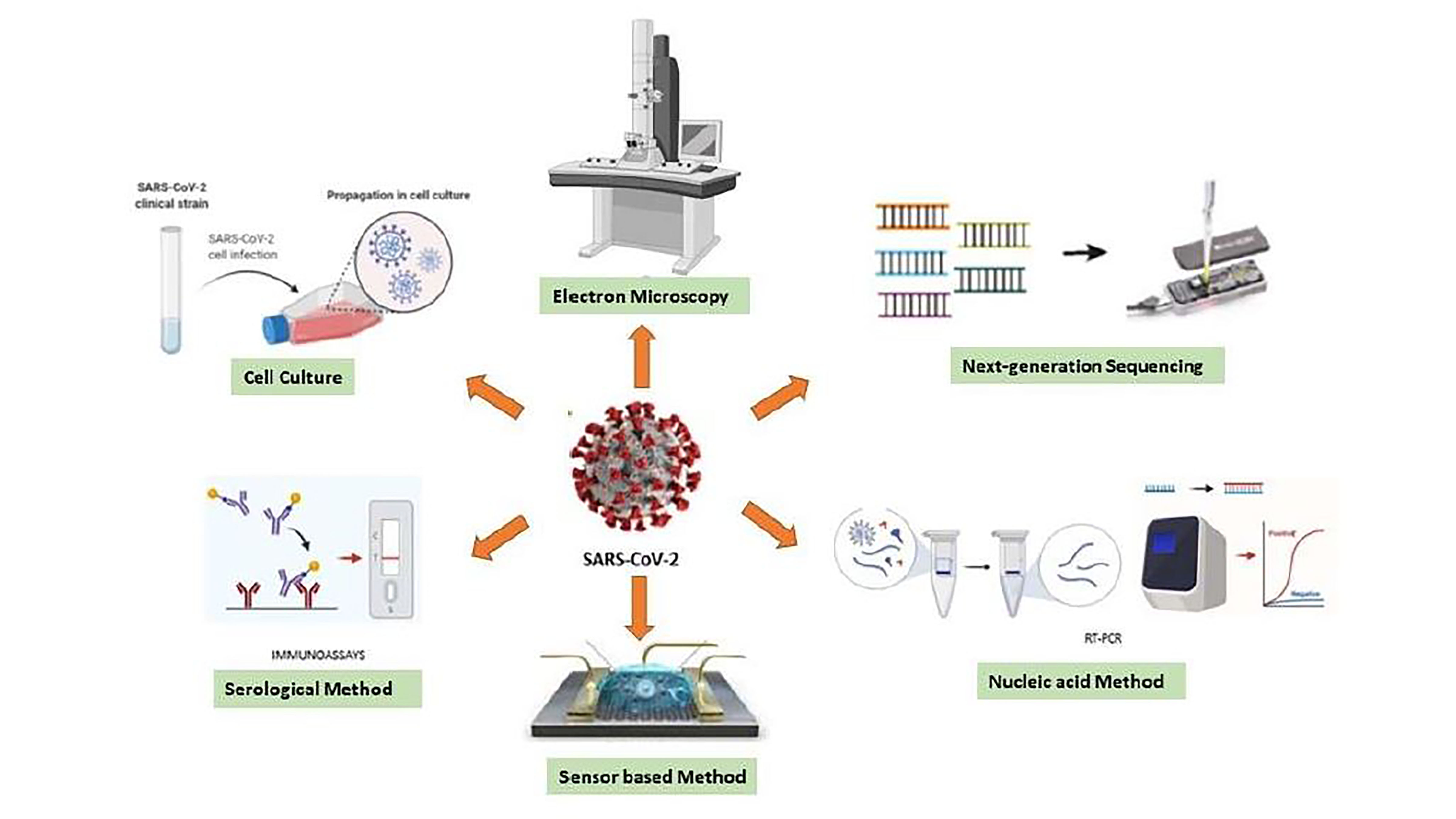
Out of Many COVID-19 Tests, Which One to Choose?
Curbing the coronavirus pandemic relies heavily on how quickly a potentially exposed individual can be tested and quarantined. However, the current diagnostic techniques vary in reliability and relevance, so an understanding of which test is most appropriate for a given circumstance is necessary to avoid false reports. Researchers evaluated the available diagnostic techniques and determined key steps required for better testing moving forward. They present their findings in the journal APL Bioengineering.

NIH RADx initiative advances six new COVID-19 testing technologies
NIH, with BARDA, today announced a third round of contract awards for scale-up and manufacturing of new COVID-19 testing technologies.
AACC Launches a New Competition to Support Innovative Research in Diagnostic Testing
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce a new competition that will support cutting-edge research that could significantly improve diagnostic testing and patient care. Winners of the competition will each receive a sample set from AACC’s Universal Sample Bank, which includes blood samples from hundreds of healthy individuals that were collected to aid medical studies.
AACC Releases Guidance Document on Using Point-of-Care Tests to Improve Patient Care
AACC has issued a new guidance document detailing best practices that hospitals and other healthcare institutions should follow when running a point-of-care testing program. As point-of-care tests emerge for more and more conditions—including COVID-19—the guidance emphasizes that it is essential for laboratory professionals and clinicians to collaborate on point-of-care testing programs to ensure this testing benefits patients.
10 thousand Utahns will be tested for COVID-19 under Utah HERO
The David Eccles School of Business at the University of Utah and University of Utah Health announce the start of Utah HERO (Health & Economic Recovery Outreach), a massive undertaking that will begin with the testing of 10,000 Utahns across four counties. The data gathered will inform decision-makers in the state as they work to help keep residents safe and get people back to work.
NIH mobilizes national innovation initiative for COVID-19 diagnostics
NIH today announced a new initiative aimed at speeding innovation, development and commercialization of COVID-19 testing technologies, a pivotal component needed to return to normal during this unprecedented global pandemic.
AACC Announces 2020 Award Winners; Celebrates Achievements in Laboratory Medicine
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce the recipients of the 2020 AACC and AACC Academy Awards. Through this annual awards program, AACC and its academy recognize individuals worldwide for exceptional research and service in the field of laboratory medicine, and strive to raise awareness that clinical laboratory testing is vital to quality patient care.
AACC Reschedules 2020 AACC Annual Scientific Meeting & Clinical Lab Expo to December to Ensure a Healthy and Safe Meeting Experience for Attendees
AACC is pleased to announce that the organization will be able to preserve the complete 2020 AACC Annual Scientific Meeting & Clinical Lab Expo experience for its attendees by moving the meeting to December 13-17, 2020. After carefully monitoring the ongoing global crisis caused by the COVID-19 outbreak, the association’s leadership determined that rescheduling is the best way to safeguard the health of meeting attendees, partners, and staff, which is AACC’s number one priority.
New Legislation Would Jeopardize Patient Access to Medical Tests Across the Board by Restricting Policy that Removed Barriers to Coronavirus Testing
On March 5, U.S. House and Senate lawmakers introduced the VALID Act, which would give the Food and Drug Administration (FDA) new, expansive powers to regulate laboratory developed tests—tests that are already regulated by the Centers for Medicare and Medicaid Services (CMS) and are subject to stringent personnel, quality control, and proficiency testing requirements. This bill promotes duplicative, costly federal regulations for clinical laboratories that will result in decreased patient access to essential medical tests. AACC urges Congress not to act on this bill until its impact on healthcare can be thoroughly evaluated.
AACC Statement on New FDA Guidance That Allows Certified Labs to Perform Coronavirus Testing
AACC thanks the FDA for being responsive to the concerns of the clinical laboratory community and amending the coronavirus guidance to allow CMS-certified labs to develop and implement new tests for coronavirus prior to FDA approval.
As U.S. Struggles to Get Coronavirus Testing Up and Running, AACC Calls on FDA to Allow Clinical Labs to Develop Their Own Tests for the Virus
In a letter to the Food and Drug Administration (FDA), AACC is urging the agency to allow clinical laboratories to develop coronavirus tests without going through FDA review. Lifting this regulatory requirement is key to ensuring that all patients have access to high-quality coronavirus testing and that healthcare workers have the tools they need to control the spread of this disease in the U.S.
Two New Rapid Coronavirus Tests Could Play Key Role in Efforts to Contain Growing Epidemic
Breaking research in AACC’s Clinical Chemistry journal shows that two new tests accurately diagnose coronavirus infection in about 1 hour. These tests could play a critical role in halting this deadly outbreak by enabling healthcare workers to isolate and treat patients much faster than is currently possible.
Top Doctors Limit Number of Tests They Order to Signal Diagnostic Prowess to Peers
A new study by Carey Business School researchers notes that some expert medical diagnosticians may order fewer patient tests as a way to indicate a high level of competence to their peers. They do so despite an increase in diagnostic techniques that can assess patient condition more accurately than former methods.
New Method Detects Toxin Exposure from Harmful Algal Blooms in Human Urine
A newly developed method can detect even low-dose human exposure to microcystins and nodularin in human urine. During harmful algal blooms (HABs), species of cyanobacteria release toxic peptides, including microcystins and nodularin into waterways, impacting wildlife and humans living in these marine environments. These findings are the first to report microcystin concentrations directly from exposed residents impacted by cyanobacteria in Florida, and is a critical step in developing and interpreting clinical diagnostic tests for HABs exposure worldwide.
New AACC Guidance Document Outlines How Healthcare Professionals Can Prevent the Widespread Supplement Biotin From Interfering With Medical Tests
Biotin’s upsurge in popularity has led to a parallel rise in incidents of this health supplement interfering with critical medical tests. A new guidance document from AACC urges clinicians and laboratory experts to collaborate to prevent this potentially harmful test interference, and to ensure that patients taking biotin receive high quality care.
Paper-based test could diagnose Lyme disease at early stages
Researchers reporting in ACS Nano have devised a blood test that quickly and sensitively diagnoses the disease at early stages.
The Journal of Applied Laboratory Medicine Is Now Indexed in PubMed
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce that its peer-reviewed publication The Journal of Applied Laboratory Medicine has been accepted for indexing in PubMed, one of the largest and most respected databases of medical research in the world.
AACC’s Journals, Clinical Chemistry and The Journal of Applied Laboratory Medicine, to Be Published through Oxford University Press
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce a publishing partnership with Oxford University Press for its peer-reviewed journals, Clinical Chemistry and The Journal of Applied Laboratory Medicine. Both journals will join Oxford University Press’s world-class science portfolio starting in January 2020. The partnership will increase researchers’ and healthcare practitioners’ access worldwide to the important science published in the journals.