A new study, led by professors at the University of Chicago and Duke University, found that COVID-19 cases in the southern state of Karnataka, India, are nearly 95 times greater than reported.
Tag: coronavirus testing
Media Advisory: AnteoTech enters high sensitivity Assay Development market built on proprietary AnteoBind™ technology.
AnteoTech marks its entry into the Assay Development market through the design of a high sensitivity COVID-19 Antigen Rapid Test. The test uses an AnteoBind™ activated Europium particle to enable the detection of low viral loads. AnteoTech is pleased to announce the expansion of its business to include Assay Development in addition to its existing Raw Material Supply business.
New Saliva-Based Antibody Test for SARS-CoV-2 Highly Accurate in Initial Study
A new saliva-based test developed by a team at Johns Hopkins Bloomberg School of Public Health has been found to accurately detect the presence of antibodies to SARS-CoV-2, the virus that causes COVID-19.

The Medical Minute: The three types of COVID-19 tests
Eight months into the COVID-19 pandemic, questions about testing abound. With COVID-19 cases trending upward, it’s important to understand when individuals should seek getting tested and what type of test they should have.
Scientists pioneer faster, cheaper COVID-19 testing technology
The U.S. Food and Drug Administration has granted emergency use authorization for UCLA Health to launch a new method of COVID-19 detection using sequencing technology called SwabSeq. Capable of testing thousands of samples at once, the method returns accurate, individual results in 12 to 24 hours.
Trump’s positive test ‘demolishes’ his COVID-19 narrative
On Friday, President Trump announced that he and First Lady Melania Trump tested positive for COVID-19. Doug Kriner, Cornell University professor of government and an expert on presidential power, says that the president’s test undermines his narrative about the virus.…
AACC Urges Congress to Fund Laboratory Training Programs to Better Prepare the U.S. for Future Pandemics
In the face of a chronic shortage of professionals who are qualified to perform clinical laboratory tests—including those for COVID-19—AACC released a position statement today calling on Congress to provide federal funding to expand clinical laboratory training programs. This will help to ensure that labs have the staffing they need to deliver timely, accurate test results, particularly during public health emergencies such as the current coronavirus pandemic.
A New Test Could Identify COVID-19 Patients Who Are at Risk for Severe Illness
A novel study in AACC’s Clinical Chemistry journal shows that a new test for SARS-CoV-2 viral proteins known as antigens could determine which COVID-19 patients are most likely to become critically ill. This test could help improve management of COVID-19 by enabling hospitals to ensure that these patients have access to intensive care.
U.S. COVID-19 Testing Too Slow, Survey Says
Rutgers scholar Katherine Ognyanova is available to comment on the latest Rutgers-Harvard-Northeastern-Northwestern survey data on waiting times for diagnostic tests across the United States from The COVID-19 Consortium for Understanding the Public’s Policy Preferences Across States. The researchers surveyed 19,058 people…
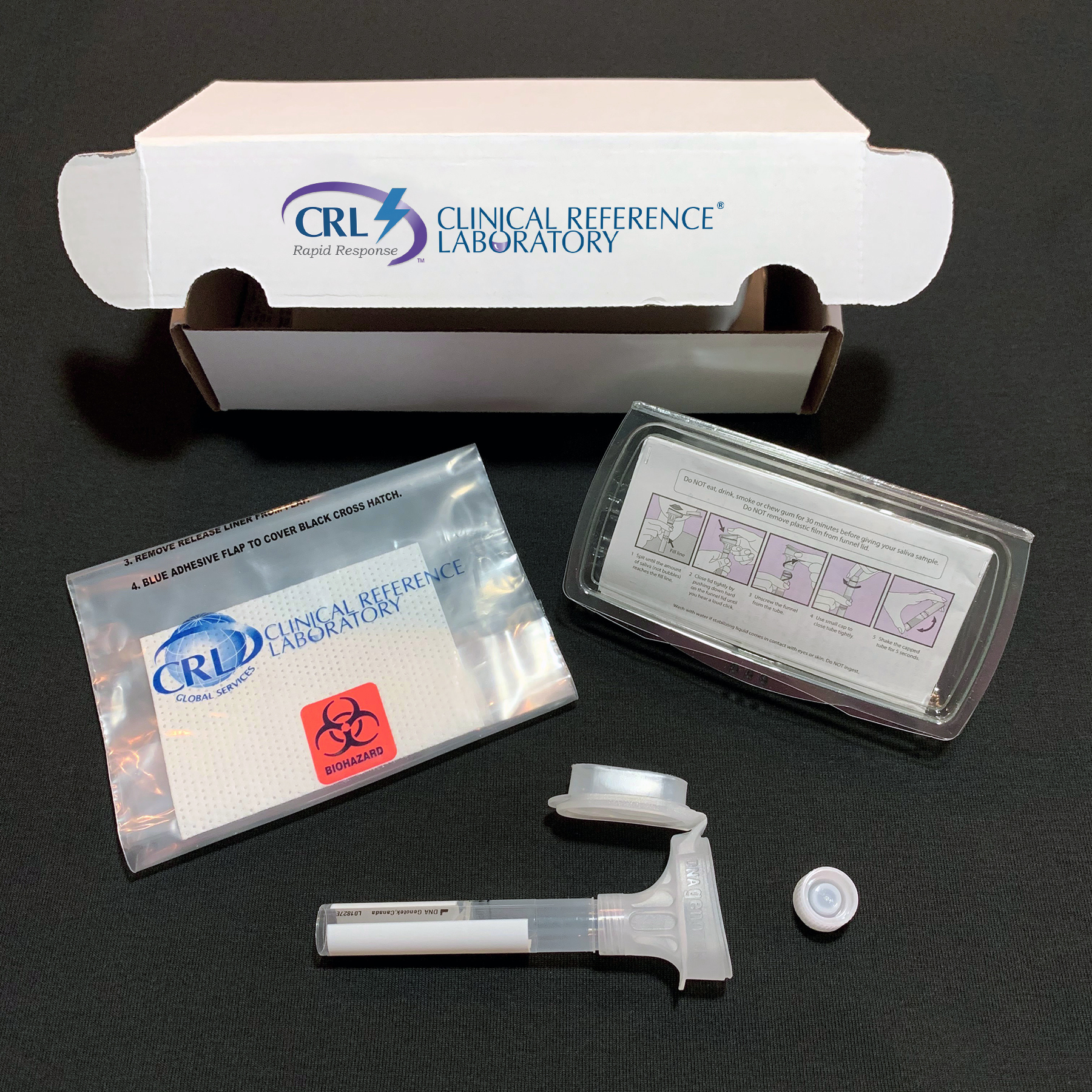
Clinical Reference Laboratory Receives FDA Emergency Use Authorization for Best-in-Class Self-Collected COVID-19 Saliva Test
Today, Clinical Reference Laboratory (CRL), one of the largest privately held clinical testing laboratories in the U.S., announced that it received FDA Emergency Use Authorization (EUA) and is scaling up capacity for CRL Rapid Response™, a saliva-based COVID-19 RT-PCR test that can be self-collected at home, work or any other setting. The test, shown in CRL’s EUA studies to be more sensitive and accurate than the standard COVID-19 anterior nasal swab test, detects the presence of coronavirus in the saliva of the test taker. In addition, the test is more comfortable and easier to administer, is not “technique dependent” and virtually anyone can self-collect an adequate sample for testing, with test results available in 24-48 hours of receipt at CRL. CRL Rapid Response™ is ready for immediate commercial launch, making it the first large-scale service of its kind focused on the American workforce. Testing is critical to safely helping America get back to work, which is why CRL is offering
Coronavirus testing, immunity: What we know
Questions around coronavirus testing and immunity are top of mind as the pandemic continues to spread and potential vaccines undergo trials.
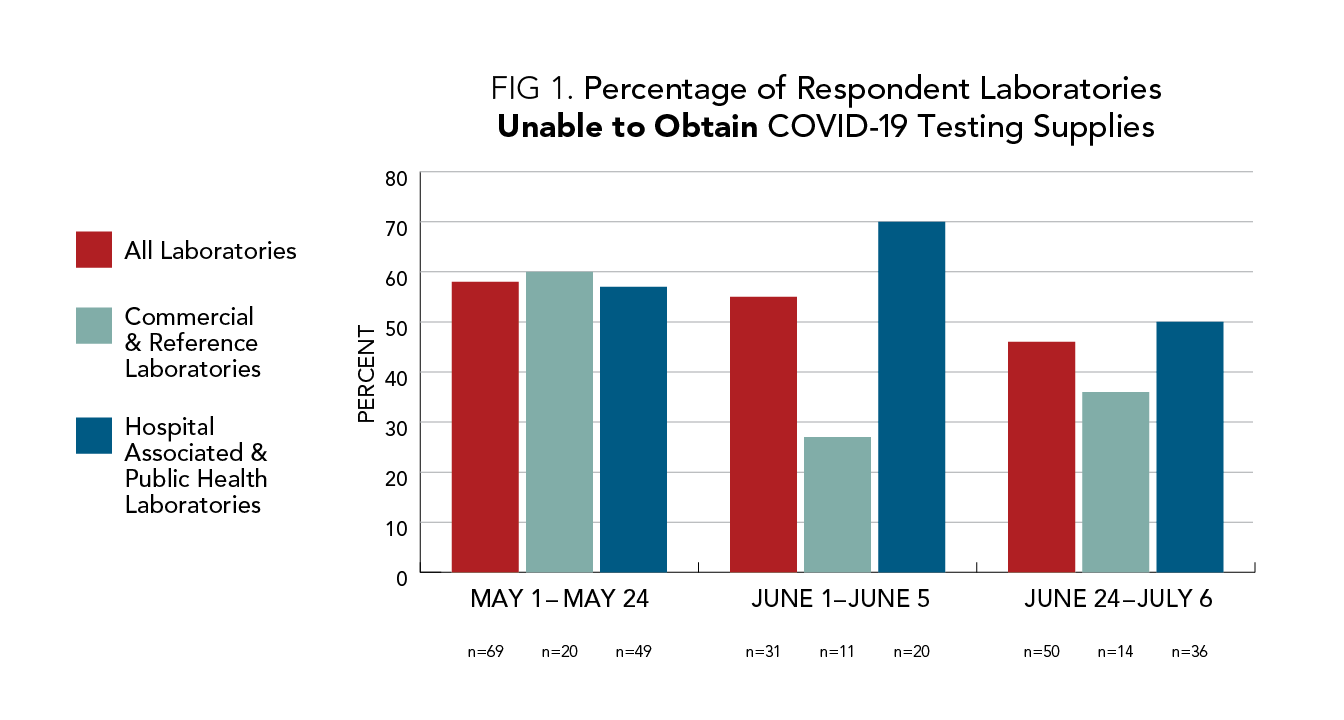
Labs Still Do Not Have Supplies for COVID-19 Testing, AACC Survey Finds
A new survey of U.S. clinical laboratories conducted by AACC has found that nearly half of all responding labs still do not have the supplies they need to run COVID-19 tests. AACC presented these findings to the White House Coronavirus Task Force today in a letter that calls on the federal government to take a more active role in alleviating this problem, so that labs can increase their testing capacity in the midst of the virus’s latest surge.
Arizona State University and World Economic Forum, with support from The Rockefeller Foundation, announce COVID-19 Diagnostics Commons to help companies get back to work
To help companies safely move their employees back to the workplace, Arizona State University’s College of Health Solutions and the World Economic Forum, with support from The Rockefeller Foundation, announced today the COVID-19 Diagnostics Commons — an interactive hub for the global community to access the very latest information about testing options and to share knowledge and practices for safely bringing back and keeping employees in the workplace during the COVID-19 era.
Why Is It So Hard to Get Tested for Covid-19?
Dr. Wiley sheds light on why testing capacity remains frustratingly limited, explaining that shortages of crucial supplies are a significant obstacle to widespread testing—and what the federal government can do to remedy this issue.
AACC Is Honored to Participate in a New Federal Initiative to Improve COVID-19 Testing Across the U.S.
We at AACC would like to thank Assistant Secretary for Health Admiral Brett P. Giroir, MD, for his leadership in establishing the National Testing Implementation Forum to address the persistent challenges that the U.S. is facing with COVID-19 testing and for inviting us to participate in this initiative.
Rapid coronavirus test speeds up access to urgent care and will free up beds ahead of winter
• The SAMBA II diagnostic device cuts COVID-19 test result times from over 24 hours to just two hours.
• First “real-world” study of SAMBA devices on hospital wards finds patient time on COVID ‘holding wards’ was almost halved.
• Researchers say faster tests helped free up beds and expedite access to life-saving treatments such as organ transplants – and might make all the difference later this year.
To Prevent a Second Wave of COVID-19, AACC Calls on Senate to Include 5 Recommendations in Upcoming Coronavirus Bill
Now that the latest coronavirus relief package, known as the Heroes Act, has moved forward to the U.S. Senate, AACC has sent a letter to Senate leadership outlining five key recommendations that will improve COVID-19 testing capacity across the U.S. AACC urges the Senate to ensure these recommendations are addressed within the Heroes Act, as they are critical to preventing a second wave of the pandemic.
AACC Issues Recommendations on the Use of COVID-19 Antibody Tests
To ensure the proper use of antibody testing for the novel coronavirus, AACC today issued a public statement detailing the role these tests should play in the management of COVID-19 patients and in the development of public health policy. In particular, the organization emphasizes that healthcare professionals and policymakers should work closely with laboratory experts on antibody testing to ensure that these tests are validated, used appropriately, and interpreted correctly.
Not All FDA-Authorized COVID-19 Antibody Tests Are Equally Reliable
A first-of-its-kind study published today in AACC’s Clinical Chemistry journal compared the performance of two COVID-19 antibody tests and found that—even though both tests are FDA authorized—one produced more incorrect results than the other. These findings could help healthcare professionals and researchers to better select antibody tests, which are essential to both treating COVID-19 patients and determining the full extent of the outbreak.
Intel from an Outpatient COVID-19 Clinic
A new report offers insights that can help clinicians distinguish between patients with COVID-19 infections and those with other conditions that may mimic COVID-19 symptoms.
The Nation Needs More Evidence Before Relying on Home Test Collection Kits for COVID-19
With the first home collection test kit for COVID-19 now authorized by the FDA, AACC is warning that more evidence is needed before the country can rely on home-based kits.
Early Predictor of Severe Respiratory Failure in Patients With COVID-19 Identified
A very high level of a protein known as suPAR in the blood of patients with COVID-19 may be a predictor of severe respiratory failure, according to new research published in the Journal of Critical Care on April 30.
AACC Launches Free Video Series to Help Explain COVID-19 Testing
To address some of the confusion surrounding COVID-19 testing, AACC today launched a video series in which leading laboratory experts answer common questions about tests for the pandemic. From persistent supply shortages to the emergence of antibody tests, these videos quickly answer the questions that patients and the general public have been asking.
Lab Experts Can Fill Gaps in FDA Regulation by Validating COVID-19 Antibody Tests
Now that diagnostic companies can sell COVID-19 antibody tests without FDA authorization, healthcare teams should work closely with clinical laboratory experts to ensure that these tests are thoroughly validated and used appropriately. A new opinion piece in AACC’s Clinical Chemistry journal emphasizes that this is critical to minimizing the risk of inaccurate results from these tests, which could have potentially life-threatening consequences.
AACC Commends Congress for Including Funding for Testing Improvements in the Latest Coronavirus Relief Package
AACC commends Congress for responding to the concerns of the clinical laboratory community and including $25 billion in the Paycheck Protection Program and Health Care Enhancement Act that is broadly marked for expanding the nation’s coronavirus testing capacity.
Georgia Tech Produces Key Components for Coronavirus Test Initiative
With not nearly enough coronavirus tests to go around, researchers usually seeking new scientific insights have reworked their labs to produce the enzymes for 3,000 new tests per day.
The Federal Government Must Act on Supply Chains to Enable COVID-19 Testing in High Volumes
With shortages of test components and swabs, labs still face huge obstacles to COVID-19 testing. With new White House guidelines reliant on more testing, AACC is urging the administration to find and coordinate resources so lab experts can do their jobs.

SLAC joins the global fight against COVID-19
The lab is responding to the coronavirus crisis by imaging disease-related biomolecules, developing standards for reliable coronavirus testing and enabling other essential research.

UChicago Medicine expands COVID-19 testing to South Side partners, community organizations
The University of Chicago Medicine health system, including Ingalls Memorial, will expand COVID-19 testing for up to 1,000 symptomatic people each day, a roughly fivefold increase over the average 200 daily tests that were performed since onsite screenings began on March 15.

Coronavirus Testing Begins at the Weizmann Institute of Science
On April 12, the Weizmann Institute began using its cutting-edge PCR facilities to perform coronavirus testing after an agreement was reached with Israel’s Ministry of Health.
The Need for FDA to Regulate Poorly Performing Commercial COVID-19 Tests Should Not Hinder Laboratory-Developed Tests
In the wake of FDA’s decision to loosen its emergency use authorization criteria for COVID-19 tests, reports have now been surfacing about unreliable commercial COVID-19 serological tests. While this is a major concern, AACC wants to emphasize that these problematic commercial tests are not the same as laboratory-developed tests–and that we still strongly support FDA’s decision to step back from regulating lab-developed tests for COVID-19.
Coriell Institute for Medical Research Fills Gaps in COVID-19 Testing Materials Supply
Answering the growing need for additional testing, and in direct response to shortages encountered in testing kit supply pipelines, the Coriell Institute for Medical Research is now offering services to create viral testing collection tubes containing different media and/or buffer formulations, for use in COVID-19 testing kits. Coriell is also exploring the feasibility of saliva as a means for testing for the new coronavirus.

Digital Tool Helps Hospital Make Important Coronavirus Retest Decisions
The dearth of coronavirus tests and the many false negatives confront doctors with a difficult decision this new tool helps them make.

New Rutgers Saliva Test for Coronavirus Gets FDA Approval
The FDA has granted emergency use authorization (EUA) to Rutgers’ RUCDR Infinite Biologics and its collaborators for a new collection approach that utilizes saliva as the primary test biomaterial for the SARS-CoV-2 coronavirus, the first such approval granted by the federal agency. The new saliva collection method, which RUCDR developed in partnership with Spectrum Solutions and Accurate Diagnostic Labs (ADL), will allow for broader population screening than the current method of nose and throat swabs.

FDA Approves COVID-19 Innovations; Ohio State Wexner Medical Center To Share Nationwide
The Food and Drug Administration has approved solutions created by scientists at The Ohio State University Wexner Medical Center that both expand and accelerate COVID-19 testing across Ohio.
College of American Pathologists launches quality control program for COVID-19
The College of American Pathologists (CAP), in response to the coronavirus disease (COVID-19) pandemic, released a new proficiency testing (PT) program for the detection of severe acute respiratory syndrome coronavirus 2 (SARS CoV-2).

Virus-Scanning Tool Could Detect Previous COVID-19 Infections and Inform Vaccine Development
Using a research assay called VirScan, scientists plan to study how antibodies from people who have had COVID-19 attack the virus that causes it.

Biostatistics expert: ‘We’re not testing as much as we should be’ for COVID-19’s spread
A Virginia Tech professor who specializes in statistical modeling and disease surveillance says the United States has to do a better job of testing for the novel coronavirus if the country is to be successful in mitigating the virus’ spread.…
AACC Applauds the Senate for Updating Its Legislation to Ensure that All Patients Have Access to COVID-19 Testing
AACC applauds the U.S. Senate for responding to the concerns of the clinical laboratory community and revising the Coronavirus Aid, Relief, and Economic Security (CARES) Act to ensure that it provides insurance coverage for all COVID-19 tests, regardless of whether or not they are performed under an FDA emergency use authorization (EUA).
AACC Launches Directory of U.S. Labs That Are Performing COVID-19 Testing
To facilitate rapid identification of COVID-19 cases, AACC has launched a directory of U.S. clinical laboratories that are, or will be, performing testing for SARS-CoV-2, the virus that causes COVID-19. This directory is designed to help healthcare providers quickly find laboratories that can diagnose patients suspected to have COVID-19.
AACC Urges Congress to Ensure That All COVID-19 Tests Are Covered by Insurance Now That FDA Has Released Guidance That Will Expand Testing Access
On March 16, FDA updated its guidance on COVID-19 testing to allow clinical labs to create and perform COVID-19 tests without pursuing FDA emergency use authorization (EUA). AACC commends this decision, which will help speed the expansion of COVID-19 testing at this crucial time. However, the association remains concerned that the Families First Coronavirus Response Act does not provide coverage for COVID-19 tests unless the tests are performed under an EUA. AACC urges Congress to rectify this problem before passing the bill so that all patients will have access to coronavirus testing.
Families First Coronavirus Response Act Would Prohibit Insurers from Paying for Some COVID-19 Tests
AACC greatly values the work that the U.S. House of Representatives has done to support American families in the midst of the coronavirus outbreak and is supportive of the goals of H.R. 6201, the Families First Coronavirus Response Act. However, we are concerned that the language as currently drafted does not provide coverage for COVID-19 tests performed prior to those tests receiving Emergency Use Authorization (EUA) from the Food & Drug Administration (FDA).
New Legislation Would Jeopardize Patient Access to Medical Tests Across the Board by Restricting Policy that Removed Barriers to Coronavirus Testing
On March 5, U.S. House and Senate lawmakers introduced the VALID Act, which would give the Food and Drug Administration (FDA) new, expansive powers to regulate laboratory developed tests—tests that are already regulated by the Centers for Medicare and Medicaid Services (CMS) and are subject to stringent personnel, quality control, and proficiency testing requirements. This bill promotes duplicative, costly federal regulations for clinical laboratories that will result in decreased patient access to essential medical tests. AACC urges Congress not to act on this bill until its impact on healthcare can be thoroughly evaluated.