AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce that the journal Clinical Chemistry received an impact factor of 9.3 —the second highest in the journal’s history—and the Journal of Applied Laboratory Medicine (JALM) received its first impact factor, of 2.0, according to the newly released 2022 Clarivate Journal Citation Reports.
Tag: american association for clinical chemistry
AACC and National Kidney Foundation Release Guidance to Combat Racial and Gender Inequalities in Chronic Kidney Disease Care
Today, AACC—in collaboration with the National Kidney Foundation (NKF)—released guidance to reduce racial and gender disparities in the care of patients with chronic kidney disease (CKD). The document gives members of the healthcare team actionable, evidence-based tools to improve equity in kidney health, including recommendations for using an updated algorithm that does not disproportionately affect any one group of individuals.
AACC Honors 2023 Award Winners for Advancing Laboratory Medicine and Patient Care
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce the recipients of the 2023 AACC and AACC Academy Awards. Through this annual awards program, AACC strives to support laboratory medicine professionals in all stages of their careers and to build awareness of the essential role that clinical laboratory testing plays in improving patient health.
AACC Releases Guidance on Procalcitonin Testing to Improve Care for Patients with Bacterial Infections
AACC has issued expert guidance on clinical testing for procalcitonin, a blood marker that helps detect serious bacterial infections and sepsis. By giving clinicians and laboratory professionals much-needed clarity on how to best use procalcitonin tests, the new guidelines could improve the treatment of critically ill patients and those with certain lower respiratory infections.
Breaking Research That Could Improve Cardiac Care for Children Published in AACC’s The Journal of Applied Laboratory Medicine
A first-of-its-kind study has established pediatric reference intervals for two common tests for cardiovascular disease. Published in AACC’s The Journal of Applied Laboratory Medicine, these findings are crucial to advancing diagnosis and treatment of heart conditions in children.
A Novel Test Could Make Sure Newborns With a Serious Genetic Disease Get Essential Treatment
Scientists have developed a test that could greatly improve quality of life for infants with homocystinuria (HCU)—a congenital disease that, if not treated early, causes serious complications. Research demonstrating the efficacy of this test was published today in AACC’s Clinical Chemistry journal.
New AACC Guidance Aims to Serve as a Roadmap for Healthcare Professionals Navigating Cervical Cancer Testing Options
With the goal of improving cervical cancer care, AACC has published a new guidance document with expert recommendations on detecting this cancer. The guidance aims to update healthcare and laboratory medicine professionals on the latest advancements in this field, and to help them select the most effective cervical cancer detection strategy for their patients.
Research That’s Using Data Science to Revolutionize Patient Care Highlighted in the January Issue of AACC’s The Journal of Applied Laboratory Medicine
In this special Data Science Issue, AACC’s The Journal of Applied Laboratory Medicine highlights the groundbreaking work that laboratory medicine experts are doing to advance patient care through data analytics and to make this game-changing technology a routine part of healthcare.
Data Analytics Could Prevent Testing Bottlenecks During Future Pandemics
Breaking research demonstrates the efficacy of two data analytics-based strategies that clinical labs employed to meet COVID-19 testing demands during the height of the pandemic. These findings, published in the Data Science Issue of AACC’s The Journal of Applied Laboratory Medicine, give labs a blueprint for using data analytics to ensure patient access to testing during future infectious disease outbreaks.
AACC Statement on Excluding VALID from End of Year Legislative Package
AACC applauds the decision of the United States Congress to exclude the Verifying Accurate Leading-edge IVCT Development (VALID) Act from its end of year omnibus bill.
A New Urine Test Could Help Curtail the Use of the Latest Synthetic Cannabinoids to Enter the Drug Scene
A first-of-its-kind study published today in AACC’s Clinical Chemistry journal shows that a novel drug test detects a new class of synthetic cannabinoids called OXIZIDs. This test could enable drug enforcement agencies to identify OXIZID users and could play a critical role in efforts to stop the spread of these drugs.
Innovative Smartphone and Artificial Intelligence-Based Tests Featured at the 2022 AACC Annual Scientific Meeting
Scientists have developed a cheap, convenient smartphone test for monitoring patients at risk for dangerous blood clots. Research demonstrating that this test works, along with a second study on using artificial intelligence (AI) to improve testing and treatment for people with kidney stones, will be presented at the 2022 AACC Annual Scientific Meeting & Clinical Lab Expo. Both studies could help resolve longstanding challenges in healthcare and laboratory testing.
New Tests That Could Solve Persistent Challenges in Children’s Healthcare Presented at the 2022 AACC Annual Scientific Meeting
A first-of-its-kind test could make it easier for newborns to get care for spinal muscular atrophy, a common genetic disease that is life-threatening but treatable if caught in time. Findings on this method and a second innovative test that could improve diagnosis of pediatric urinary tract infections (UTIs) will be discussed today at the 2022 AACC Annual Scientific Meeting & Clinical Lab Expo.
Novel Research Sheds Light on Antibody Responses to the SARS-CoV-2 Virus and Vaccines
Two studies presented today at the 2022 AACC Annual Scientific Meeting & Clinical Lab Expo reveal how SARS-CoV-2 antibody levels vary among recipients of COVID-19 vaccines and naturally infected individuals. These findings add to a growing body of knowledge that is essential for guiding public health initiatives, and that might one day enable clinicians to assess individuals’ immunity to SARS-CoV-2.
AACC Announces 2022 Corporate Supporter Award Recipients; Thanks Organizations for Their Invaluable Support
AACC is pleased to announce the winners of its 2022 Top Corporate Supporter Awards. This year, AACC recognizes 35 different companies and organizations that generously support the association through advertising, sponsorships, and exhibiting. These significant contributions make it possible for AACC to further its mission of better health through laboratory medicine.
Artificial Intelligence in Personalized Medicine, Genomic Sequencing Advances, Human Brain Organogenesis, Building Trust with Patients, Guiding Patient Decisions with Mass Spectrometry, and Much More to Be Explored at 2022 AACC
At the 2022 AACC Annual Scientific Meeting & Clinical Lab Expo, laboratory medicine experts will present the cutting-edge research and technology that is revolutionizing clinical testing and patient care.
Medical and Patient Communities Unite to Urge Congress to Stop Bill That Would Impede Patient Access to Essential Diagnostic Tests
AACC joins more than 100 other medical and patient organizations that have expressed concerns about the Senate HELP committee rushing to pass the VALID Act, a bill that would limit patient access to vital laboratory developed tests and the lifesaving diagnoses these tests enable. Specifically, the medical and patient communities urge Congress to remove the VALID Act from the FDASLA Act, a piece of must-pass legislation that the Senate is on the cusp of ratifying.
AACC Announces 2022 Award Winners; Celebrates Achievements in Laboratory Medicine
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce the recipients of the 2022 AACC and AACC Academy Awards. Through this annual awards program, AACC and its academy recognize individuals around the world for outstanding research and service in the field of laboratory medicine, and strive to raise awareness of the vital contribution made by all lab professionals to patient care.
AACC Calls on Congress to Halt Legislation That Would Hinder Patient Access to Essential Medical Tests
AACC has sent comments to the U.S. Senate Health, Education, Labor & Pensions (HELP) Committee urging the committee not to include the VALID Act in its omnibus Food and Drug Administration (FDA) user fee legislation. VALID would limit the availability of vital laboratory-developed tests, decreasing patient access to life-saving diagnostic tools.
AACC Statement on Non-Invasive Prenatal Screening Tests
We share the FDA’s goal of alerting the public to the potential misuse of non-invasive prenatal screening (NIPS) tests. This is why we’ve been advocating for the modernization of how laboratory-developed tests (LDTs) are regulated by the Centers for Medicare and Medicaid Services (CMS).
The Importance of Laboratory Developed Tests in Diagnosing COVID-19
Laboratory developed tests play a vital role in getting patients accurate diagnoses and effective care, and at no time has their importance been clearer than during the current pandemic. In this briefing, AACC’s leading experts in laboratory medicine will discuss why laboratory developed tests are crucial to fighting the coronavirus, as well as the regulatory barriers that nearly prevented labs from developing and introducing these tests for the virus.
AACC Urges Congress to Fund Laboratory Training Programs to Better Prepare the U.S. for Future Pandemics
In the face of a chronic shortage of professionals who are qualified to perform clinical laboratory tests—including those for COVID-19—AACC released a position statement today calling on Congress to provide federal funding to expand clinical laboratory training programs. This will help to ensure that labs have the staffing they need to deliver timely, accurate test results, particularly during public health emergencies such as the current coronavirus pandemic.
AACC and CDC Partner to Improve Cardiovascular Disease Testing Around the World
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce a new collaboration with the Centers for Disease Control and Prevention (CDC) and the CDC Foundation that aims to expand lipid testing in resource-limited countries. Improving access to this essential testing could help reduce the high worldwide mortality rate from cardiovascular disease by enabling patients to get treated for this condition earlier.
A New Test Could Identify COVID-19 Patients Who Are at Risk for Severe Illness
A novel study in AACC’s Clinical Chemistry journal shows that a new test for SARS-CoV-2 viral proteins known as antigens could determine which COVID-19 patients are most likely to become critically ill. This test could help improve management of COVID-19 by enabling hospitals to ensure that these patients have access to intensive care.
AACC Honors Senator Roy Blunt for His Dedication to Improving Pediatric Medical Testing
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce that Senator Roy Blunt (R-Missouri) is the recipient of AACC’s Outstanding Legislator Award for the 116th Congress. This award recognizes Sen. Blunt for his tireless efforts to improve children’s healthcare by advancing the development of pediatric reference intervals.
AACC Welcomes HHS Decision to Clarify Federal Regulation of Laboratory Developed Tests
AACC welcomes a decision from the Department of Health and Human Services that the Food and Drug Administration does not have authority to regulate laboratory developed tests (LDTs) without formal notice-and-comment rulemaking.
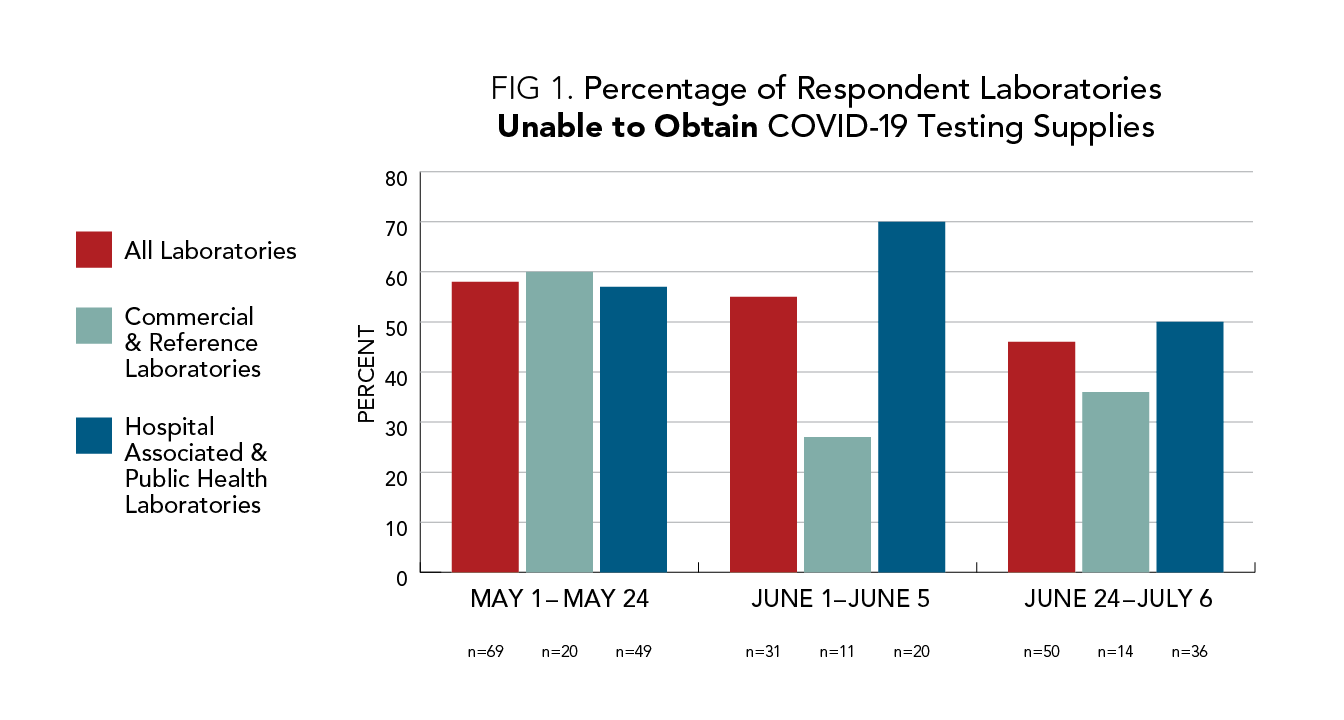
Labs Still Do Not Have Supplies for COVID-19 Testing, AACC Survey Finds
A new survey of U.S. clinical laboratories conducted by AACC has found that nearly half of all responding labs still do not have the supplies they need to run COVID-19 tests. AACC presented these findings to the White House Coronavirus Task Force today in a letter that calls on the federal government to take a more active role in alleviating this problem, so that labs can increase their testing capacity in the midst of the virus’s latest surge.
AACC Is Honored to Participate in a New Federal Initiative to Improve COVID-19 Testing Across the U.S.
We at AACC would like to thank Assistant Secretary for Health Admiral Brett P. Giroir, MD, for his leadership in establishing the National Testing Implementation Forum to address the persistent challenges that the U.S. is facing with COVID-19 testing and for inviting us to participate in this initiative.
The Impact Factor of AACC’s Clinical Chemistry Journal Increases to 7.292
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce that the impact factor of its journal, Clinical Chemistry, has risen to 7.292 in the 2019 Clarivate Analytics Journal Citation Reports. This impact factor places Clinical Chemistry in the top 4.2% of 12,838 ranked academic journals and speaks to the significant influence of the science it publishes on laboratory medicine and patient care.
To Prepare the U.S. for Future Pandemics, AACC Calls on Congress to Enact 4 Recommendations
In response to the Senate health committee’s white paper on preparing for future pandemics, AACC sent a letter to committee leadership detailing four key steps the government should take to ready the U.S. for the next outbreak. AACC urges the health committee to address these recommendations in future pandemic legislation, as they are crucial to preventing another public health crisis like the one COVID-19 has caused.
AACC Elects New Leaders to Serve Terms Starting in August 2020
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce that Stephen R. Master, MD, PhD, FAACC, has been elected to serve on the AACC Board of Directors as president-elect starting in August 2020.

AACC Welcomes Mark J. Golden as CEO
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce that its Board of Directors has appointed Mark J. Golden as CEO effective August 17.
AACC Launches a New Competition to Support Innovative Research in Diagnostic Testing
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce a new competition that will support cutting-edge research that could significantly improve diagnostic testing and patient care. Winners of the competition will each receive a sample set from AACC’s Universal Sample Bank, which includes blood samples from hundreds of healthy individuals that were collected to aid medical studies.
AACC Releases Guidance Document on Using Point-of-Care Tests to Improve Patient Care
AACC has issued a new guidance document detailing best practices that hospitals and other healthcare institutions should follow when running a point-of-care testing program. As point-of-care tests emerge for more and more conditions—including COVID-19—the guidance emphasizes that it is essential for laboratory professionals and clinicians to collaborate on point-of-care testing programs to ensure this testing benefits patients.
AACC Statement for Racial Equality
Laboratory professionals cannot be mute bystanders to inequality. Our legacy is one of service and AACC calls upon our community to be part of the dialogue to promote racial equality.
To Prevent a Second Wave of COVID-19, AACC Calls on Senate to Include 5 Recommendations in Upcoming Coronavirus Bill
Now that the latest coronavirus relief package, known as the Heroes Act, has moved forward to the U.S. Senate, AACC has sent a letter to Senate leadership outlining five key recommendations that will improve COVID-19 testing capacity across the U.S. AACC urges the Senate to ensure these recommendations are addressed within the Heroes Act, as they are critical to preventing a second wave of the pandemic.
AACC Issues Recommendations on the Use of COVID-19 Antibody Tests
To ensure the proper use of antibody testing for the novel coronavirus, AACC today issued a public statement detailing the role these tests should play in the management of COVID-19 patients and in the development of public health policy. In particular, the organization emphasizes that healthcare professionals and policymakers should work closely with laboratory experts on antibody testing to ensure that these tests are validated, used appropriately, and interpreted correctly.
Not All FDA-Authorized COVID-19 Antibody Tests Are Equally Reliable
A first-of-its-kind study published today in AACC’s Clinical Chemistry journal compared the performance of two COVID-19 antibody tests and found that—even though both tests are FDA authorized—one produced more incorrect results than the other. These findings could help healthcare professionals and researchers to better select antibody tests, which are essential to both treating COVID-19 patients and determining the full extent of the outbreak.
The Nation Needs More Evidence Before Relying on Home Test Collection Kits for COVID-19
With the first home collection test kit for COVID-19 now authorized by the FDA, AACC is warning that more evidence is needed before the country can rely on home-based kits.
AACC Launches Free Video Series to Help Explain COVID-19 Testing
To address some of the confusion surrounding COVID-19 testing, AACC today launched a video series in which leading laboratory experts answer common questions about tests for the pandemic. From persistent supply shortages to the emergence of antibody tests, these videos quickly answer the questions that patients and the general public have been asking.
Lab Experts Can Fill Gaps in FDA Regulation by Validating COVID-19 Antibody Tests
Now that diagnostic companies can sell COVID-19 antibody tests without FDA authorization, healthcare teams should work closely with clinical laboratory experts to ensure that these tests are thoroughly validated and used appropriately. A new opinion piece in AACC’s Clinical Chemistry journal emphasizes that this is critical to minimizing the risk of inaccurate results from these tests, which could have potentially life-threatening consequences.
AACC Commends Congress for Including Funding for Testing Improvements in the Latest Coronavirus Relief Package
AACC commends Congress for responding to the concerns of the clinical laboratory community and including $25 billion in the Paycheck Protection Program and Health Care Enhancement Act that is broadly marked for expanding the nation’s coronavirus testing capacity.
AACC Announces 2020 Award Winners; Celebrates Achievements in Laboratory Medicine
AACC, a global scientific and medical professional organization dedicated to better health through laboratory medicine, is pleased to announce the recipients of the 2020 AACC and AACC Academy Awards. Through this annual awards program, AACC and its academy recognize individuals worldwide for exceptional research and service in the field of laboratory medicine, and strive to raise awareness that clinical laboratory testing is vital to quality patient care.
The Federal Government Must Act on Supply Chains to Enable COVID-19 Testing in High Volumes
With shortages of test components and swabs, labs still face huge obstacles to COVID-19 testing. With new White House guidelines reliant on more testing, AACC is urging the administration to find and coordinate resources so lab experts can do their jobs.
The Need for FDA to Regulate Poorly Performing Commercial COVID-19 Tests Should Not Hinder Laboratory-Developed Tests
In the wake of FDA’s decision to loosen its emergency use authorization criteria for COVID-19 tests, reports have now been surfacing about unreliable commercial COVID-19 serological tests. While this is a major concern, AACC wants to emphasize that these problematic commercial tests are not the same as laboratory-developed tests–and that we still strongly support FDA’s decision to step back from regulating lab-developed tests for COVID-19.
Novel Research in AACC’s Clinical Chemistry Journal Shows That Vitamin D Supplements Do Not Prevent Osteoporotic Fractures
A first-of-its-kind study published in AACC’s Clinical Chemistry journal has found that low vitamin D levels alone do not cause osteoporotic fractures. This research could resolve the longstanding debate over whether vitamin D supplements prevent these fractures, and indicates that members of the general population should not rely on vitamin D by itself for this purpose.
AACC Reschedules 2020 AACC Annual Scientific Meeting & Clinical Lab Expo to December to Ensure a Healthy and Safe Meeting Experience for Attendees
AACC is pleased to announce that the organization will be able to preserve the complete 2020 AACC Annual Scientific Meeting & Clinical Lab Expo experience for its attendees by moving the meeting to December 13-17, 2020. After carefully monitoring the ongoing global crisis caused by the COVID-19 outbreak, the association’s leadership determined that rescheduling is the best way to safeguard the health of meeting attendees, partners, and staff, which is AACC’s number one priority.
AACC Applauds the Senate for Updating Its Legislation to Ensure that All Patients Have Access to COVID-19 Testing
AACC applauds the U.S. Senate for responding to the concerns of the clinical laboratory community and revising the Coronavirus Aid, Relief, and Economic Security (CARES) Act to ensure that it provides insurance coverage for all COVID-19 tests, regardless of whether or not they are performed under an FDA emergency use authorization (EUA).
When Mothers Receive Fentanyl Epidurals During Labor, the Fentanyl Gets Passed on to Their Babies
Breaking research in AACC’s The Journal of Applied Laboratory Medicine shows that the fentanyl in epidurals can pass on to babies during labor. While the infants in this study did not experience adverse effects from this fentanyl transfer, this information is crucial to ensuring that new mothers don’t get falsely accused of fentanyl abuse, which can have dire social repercussions for mother and child.
AACC Launches Directory of U.S. Labs That Are Performing COVID-19 Testing
To facilitate rapid identification of COVID-19 cases, AACC has launched a directory of U.S. clinical laboratories that are, or will be, performing testing for SARS-CoV-2, the virus that causes COVID-19. This directory is designed to help healthcare providers quickly find laboratories that can diagnose patients suspected to have COVID-19.