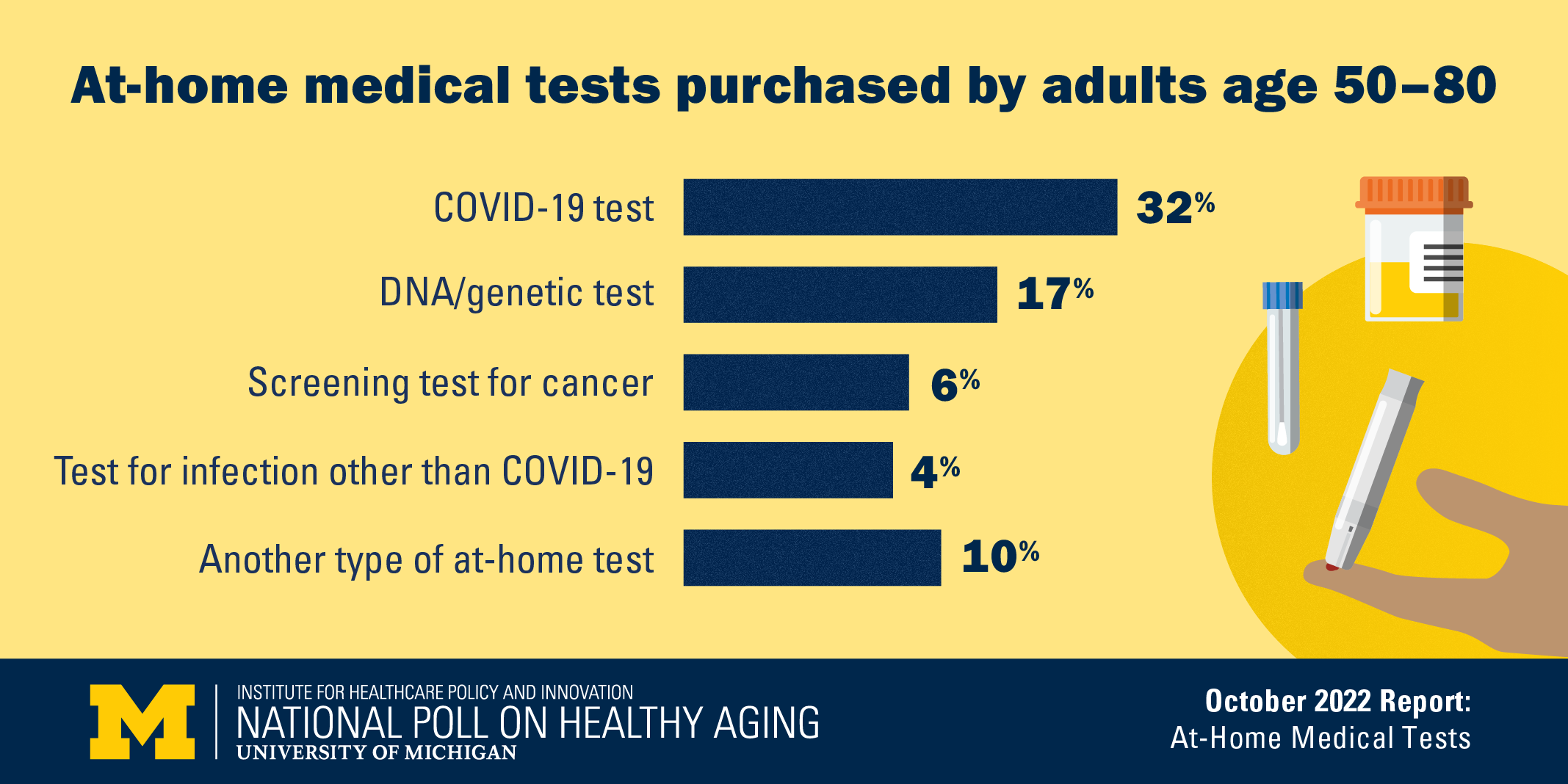
A new poll shows that 48% of people age 50 to 80 have bought at least one kind of at-home health test, including 32% who had bought COVID-19 tests, 17% who had bought a DNA test, and lower percentages who had bought other types of tests. But use of such direct-to-consumer medical tests varies greatly by age, race/ethnicity, marital status, income and years of education.