David Winter, MD, at Baylor Scott & White Health, answers the most common patient questions and reacts to the latest medical research. Are COVID cases starting to go down? (SOT@ :14, TRT :49) How reliable are at-home COVID tests? Can…
Tag: COVID-19 testing
11 things to know now that COVID-19 isn’t an “emergency” anymore
Life has changed forever because of COVID-19. And the virus is still spreading, and still causing serious illness or significant disruption of ‘normal’ life. But when the clock strikes 11:59 p.m. on May 11, some of the special rules and programs put in place during the past three years will end. Here’s a guide.
Data Analytics Could Prevent Testing Bottlenecks During Future Pandemics
Breaking research demonstrates the efficacy of two data analytics-based strategies that clinical labs employed to meet COVID-19 testing demands during the height of the pandemic. These findings, published in the Data Science Issue of AACC’s The Journal of Applied Laboratory Medicine, give labs a blueprint for using data analytics to ensure patient access to testing during future infectious disease outbreaks.
World Cup Soccer 2022: The Medical Perspective
Bert Mandelbaum, MD, medical director of the FIFA Medical Center of Excellence, Cedars-Sinai Kerlan-Jobe Institute, and associate chief medical officer of Major League Soccer, has been providing medical care to the U.S men’s national soccer team for nearly three decades.
Kids and the COVID-19 Vaccine: Eleven Key Questions Answered
With the vaccine for children ages 6 months and older approved, the experts at Children’s Hospital Los Angeles provide guidance for families. The Centers for Disease Control and Prevention (CDC) recommends that everyone approved to receive a COVID-19 vaccine get one—including children ages 6 months and older.
Breaking Research That Could Advance COVID-19 Treatment and Testing Announced at the 2021 AACC Annual Scientific Meeting
A groundbreaking study shows that a new rapid test identifies COVID-19 patients who will deteriorate with greater accuracy than existing tests, thus helping patients to get life-saving treatment. Findings on this method, as well as a novel study on the performance of coronavirus tests in children, were presented today at the 2021 AACC Annual Scientific Meeting & Clinical Lab Expo.
Connect Chicago Initiative Expands Community COVID-19 Testing
As COVID-19 cases rise in the U.S., Connect Chicago — new initiative between the Chicago Department of Public Health, Rush University Medical Center, and Esperanza Health Centers — is aiming to redouble testing efforts in Chicago communities that need it most.

NIH Chooses University of Miami Miller School of Medicine to Head Project for Its Safe Return to In-Person School Initiative
The University of Miami Miller School of Medicine is helping to lead a National Institutes of Health (NIH) COVID-19 testing initiative to safely return children to in-person school.
Black, Latinx People Confident in COVID-19 Safety Precautions but Skeptical About Vaccines
Black and Latinx people intensely sought information on COVID-19 and engaged in public health measures such as mask-wearing and testing due to devastating experiences during the pandemic but are still skeptical about vaccines, according to a Rutgers study.
Is it a Virus or Bacteria? New Tech Rapidly Tests for COVID-19 and More
S&T is preparing for future outbreaks/pandemics by investing in a new tech that can quickly discriminate between bacterial and viral infections so that the U.S. can triage patients and plan a response without delay.
Mount Sinai Health System and Pershing Square Foundation Announce Expansion of New Saliva-Based COVID-19 Testing Program
Expanded program will be available to NYC schools and other organizations

How X-rays Could Make Reliable, Rapid COVID-19 Tests a Reality
Vaccines are turning the tide of the pandemic, but there’s still a risk of COVID-19 infections. Instant at-home tests would help us return to normal, but current options aren’t very accurate. A new discovery could get reliable tests on the market.
Publication reveals essential ingredients of the NIH RADx groundbreaking national diagnostics effort
One-year into implementation of the NIH RADx initiative, the IEEE Open Journal of Engineering in Medicine and Biology has dedicated a special issue to exploring the innovative structure and operation of the RADx Tech program.
New rapid COVID-19 test the result of university-industry partnership
A partnership between UC Davis and Maurice J. Gallagher, Jr., chairman and CEO of Allegiant Travel Company, has led to a 20-minute COVID-19 test. The method pairs a mass spectrometer with a powerful machine-learning platform to detect SARS-CoV-2 in nasal swabs. A recent study published in Nature Scientific Reports shows the test to be 98.3% accurate for positive COVID-19 tests and 96% for negative tests.

RADx diversifies COVID-19 test portfolio with four new contracts, including one to detect variants
NIH has awarded four additional contracts for the development and scaled-up manufacturing of new COVID-19 diagnostic testing technologies through its Rapid Acceleration of Diagnostics Tech (RADx) initiative. The awards total $29.3 million and will help increase testing capacity for COVID-19.
Hackensack Meridian Health’s Center for Discovery and Innovation Develops Advanced Test to Track and Diagnose COVID-19 Variants
The test can detect the known UK, Brazil, and South African variants, as well as others containing the key E484K mutation, which are gaining prominence as the virus evolves.

D.C. DERMATOLOGIST PLAYS LEADING ROLE IN COVID-19 TESTING FOR VULNERABLE COMMUNITIES
The American Academy of Dermatology (AAD) named board-certified dermatologist Ginette Okoye, MD, FAAD, a Patient Care Hero for establishing a dedicated COVID-19 community testing site in a historically underserved neighborhood in northeast Washington, D.C.
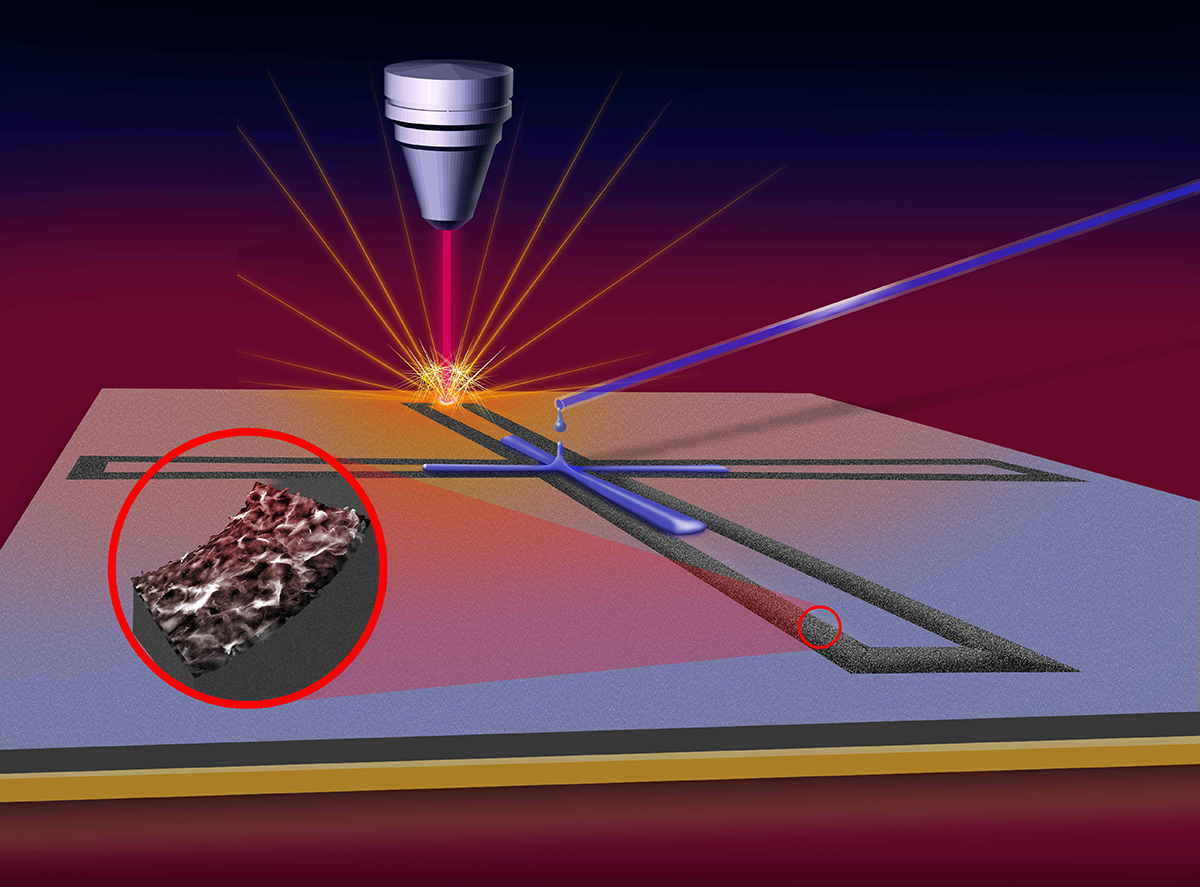
Engineers developing COVID-19 tests for mass production, mass distribution
Iowa State engineers are using their expertise in graphene-based biosensors to develop quick, inexpensive and sensitive saliva tests for the virus that causes COVID-19. Federal agencies are supporting the research with the goal of producing tests that can be easily mass produced and widely distributed.
BIDMC researchers develop model to estimate false-negative rate for COVID-19 tests
A team of researchers at Beth Israel Deaconess Medical Center (BIDMC) has developed a mathematical means of assessing tests’ false-negative rate.

Ultra-absorptive nanofiber swabs could improve SARS-CoV-2 test sensitivity
Researchers reporting in ACS’ Nano Letters have developed ultra-absorptive nanofiber swabs that could reduce the number of false-negative SARS-CoV-2 tests by improving sample collection and test sensitivity.

NIH-funded COVID-19 home test is first to receive over-the-counter authorization from FDA
The FDA granted emergency use authorization today for an innovative COVID-19 viral antigen test developed with support from NIH’s RADx Initiative.
Two New Tests Could Lower the Coronavirus Mortality Rate by Improving Treatment for Severely Ill COVID-19 Patients
A new study shows that a first-of-its-kind rapid test could identify which COVID-19 patients are responding to drug treatments and which patients are still deteriorating and need higher doses of medication. Findings on this method and on a second test that could predict COVID-19 severity were presented today at the virtual 2020 AACC Annual Scientific Meeting & Clinical Lab Expo.
Media Advisory: AnteoTech enters high sensitivity Assay Development market built on proprietary AnteoBind™ technology.
AnteoTech marks its entry into the Assay Development market through the design of a high sensitivity COVID-19 Antigen Rapid Test. The test uses an AnteoBind™ activated Europium particle to enable the detection of low viral loads. AnteoTech is pleased to announce the expansion of its business to include Assay Development in addition to its existing Raw Material Supply business.
George Washington University Public Health/Medical Experts Available for Media Interviews on the COVID-19 Pandemic
Public health experts predict the United States may be headed for thousands of new COVID-19 cases and deaths this winter, a surge that is already straining health care systems around the country. The George Washington University has the following experts…

NIH-funded tool helps organizations plan COVID-19 testing
The COVID-19 Testing Impact Calculator is a free resource that shows how different approaches to testing and other mitigation measures, such as mask use, can curb the spread of the virus in any organization.
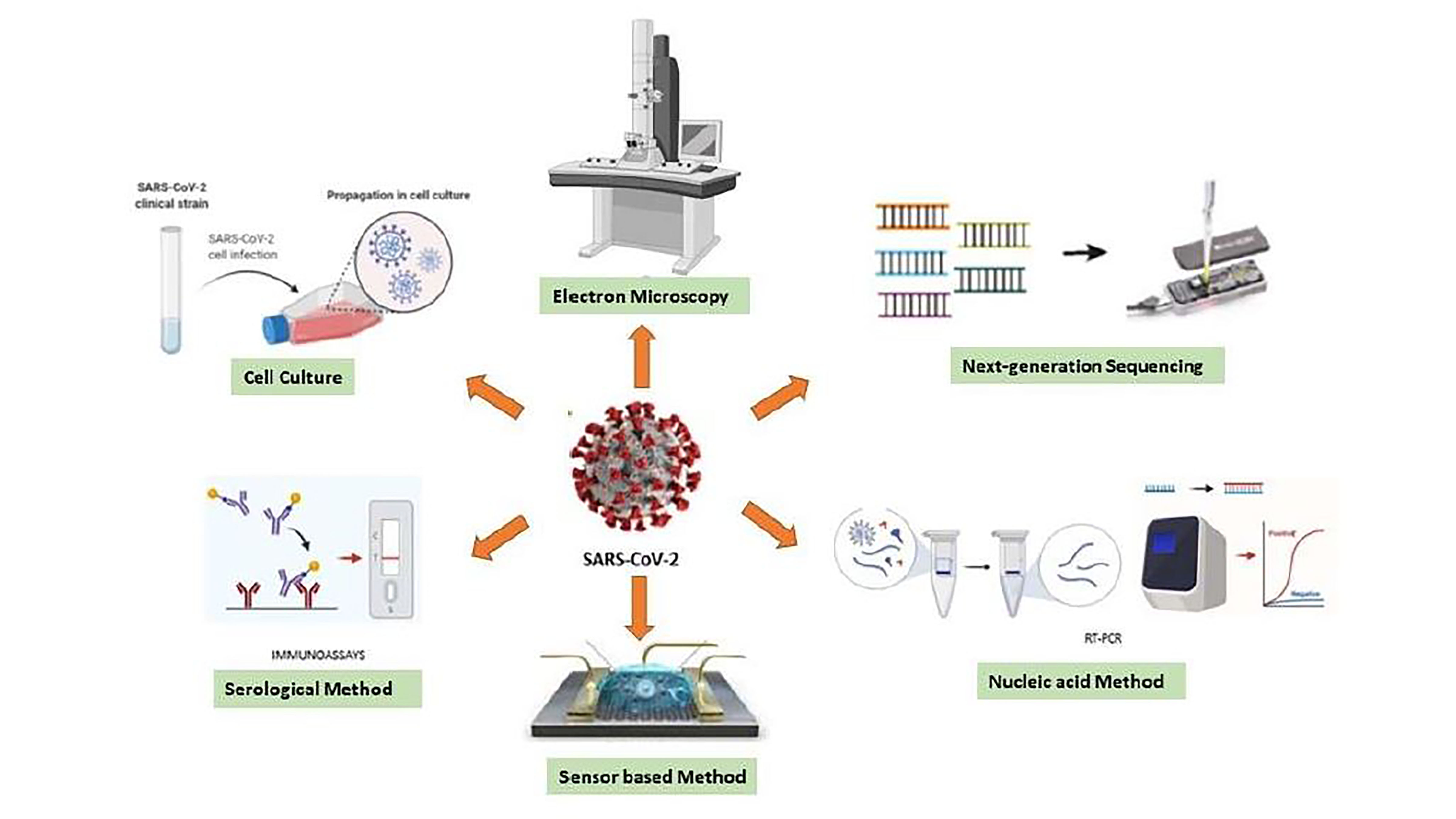
Out of Many COVID-19 Tests, Which One to Choose?
Curbing the coronavirus pandemic relies heavily on how quickly a potentially exposed individual can be tested and quarantined. However, the current diagnostic techniques vary in reliability and relevance, so an understanding of which test is most appropriate for a given circumstance is necessary to avoid false reports. Researchers evaluated the available diagnostic techniques and determined key steps required for better testing moving forward. They present their findings in the journal APL Bioengineering.
Virus-like probes could help make rapid COVID-19 testing more accurate, reliable
Nanoengineers at the University of California San Diego have developed new and improved probes, known as positive controls, that could make it easier to validate rapid, point-of-care diagnostic tests for COVID-19 across the globe. The advance could help expand testing to low-resource, underserved areas.
Rutgers Expert Available to Discuss New Jersey Using New Quick-Result COVID-19 Testing
Rutgers scholar Shawna Hudson is available to discuss the importance of New Jersey being one of the first states to receive a new quick-result coronavirus test from the U.S. Department of Health to help combat the second wave of the…

UNH Digs Deep Testing Wastewater for Early Warning Signs of COVID-19
The University of New Hampshire has gone underground to flush out cases of the coronavirus by testing wastewater on campus. The sewage sampling is being used as a secondary surveillance method to the already required twice a week individual nasal test to track and detect SARS-CoV-2, the virus that causes COVID-19.

The Medical Minute: The three types of COVID-19 tests
Eight months into the COVID-19 pandemic, questions about testing abound. With COVID-19 cases trending upward, it’s important to understand when individuals should seek getting tested and what type of test they should have.
Coronavirus Test Results Are Faster but Still Too Slow For Contract Tracing, National Survey Says
The average test times for coronavirus results have fallen from four days in April to 2.7 days in September, but results are still too slow for effective contact tracing, according to a new nationwide survey led by researchers from Rutgers University–New Brunswick and Northeastern, Harvard and Northwestern universities.
AACC Urges Congress to Fund Laboratory Training Programs to Better Prepare the U.S. for Future Pandemics
In the face of a chronic shortage of professionals who are qualified to perform clinical laboratory tests—including those for COVID-19—AACC released a position statement today calling on Congress to provide federal funding to expand clinical laboratory training programs. This will help to ensure that labs have the staffing they need to deliver timely, accurate test results, particularly during public health emergencies such as the current coronavirus pandemic.
A New Test Could Identify COVID-19 Patients Who Are at Risk for Severe Illness
A novel study in AACC’s Clinical Chemistry journal shows that a new test for SARS-CoV-2 viral proteins known as antigens could determine which COVID-19 patients are most likely to become critically ill. This test could help improve management of COVID-19 by enabling hospitals to ensure that these patients have access to intensive care.

Parents Trust Colleges More than Students for COVID-19 Safety, New Survey by TimelyMD Finds
Parents of college students indicate many concerns about their students’ return to the classroom (on campus or online), including their health, the quality of their education, and the likelihood of their following public health guidance when administrators aren’t looking. Fielded last week, this survey by TimelyMD has the latest data available as campus reopening plans change daily.

Engineers developing no-touch, mail-in, fast-scan test for COVID-19, other outbreaks
Engineers are developing a no-touch, mail-in, fast-scan diagnostic sensing system that could be used to quickly test for COVID-19 or other outbreaks. The system would also produce a real-time outbreak map with demographic details.
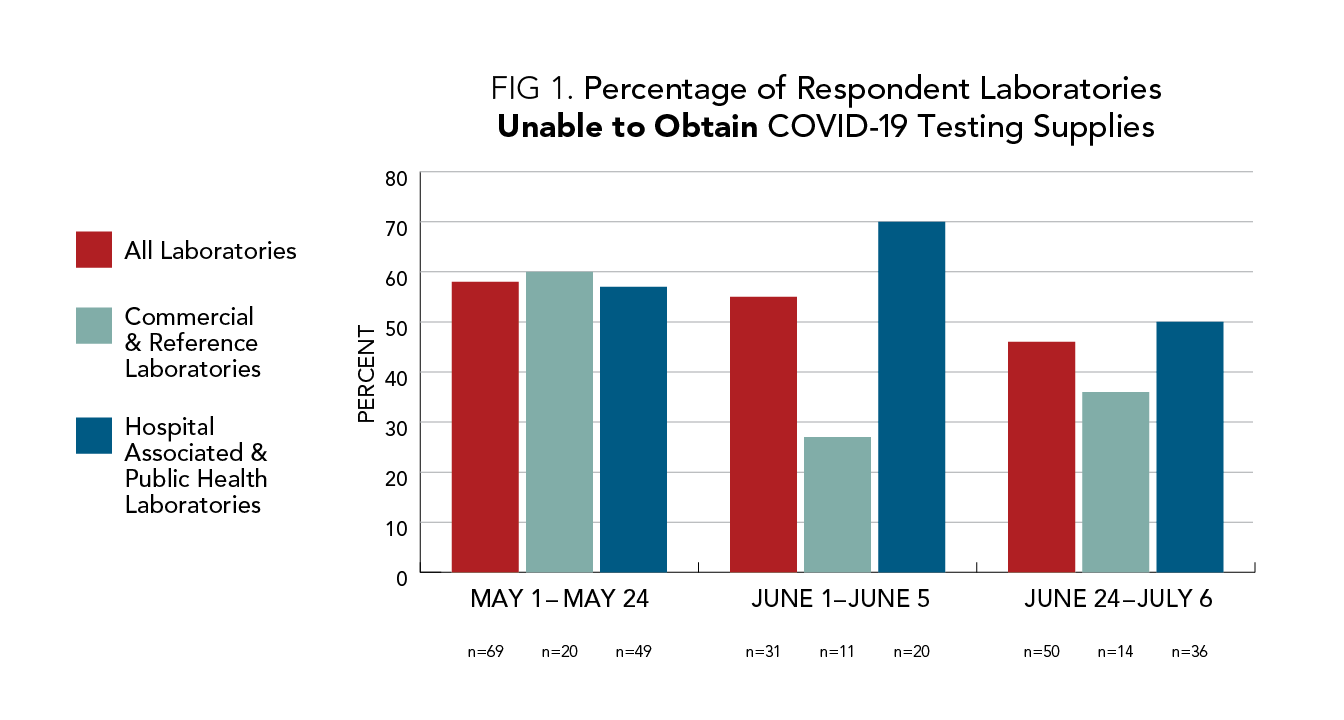
Labs Still Do Not Have Supplies for COVID-19 Testing, AACC Survey Finds
A new survey of U.S. clinical laboratories conducted by AACC has found that nearly half of all responding labs still do not have the supplies they need to run COVID-19 tests. AACC presented these findings to the White House Coronavirus Task Force today in a letter that calls on the federal government to take a more active role in alleviating this problem, so that labs can increase their testing capacity in the midst of the virus’s latest surge.
AACC Is Honored to Participate in a New Federal Initiative to Improve COVID-19 Testing Across the U.S.
We at AACC would like to thank Assistant Secretary for Health Admiral Brett P. Giroir, MD, for his leadership in establishing the National Testing Implementation Forum to address the persistent challenges that the U.S. is facing with COVID-19 testing and for inviting us to participate in this initiative.
How Would Pooled Testing Work for Covid-19?
As supply shortages continue to hamper COVID-19 testing, AACC President Dr. Carmen Wiley explains how pooled testing could enable clinical labs to process more tests while saving the materials needed to run them.

Asymptomatic Testing Central to UC San Diego’s Return to Learn for Fall Quarter
The University of California San Diego today announced the next step in its Return to Learn program, which will guide an incremental repopulation of the campus while offering broad, asymptomatic testing for faculty, staff and students on a recurring basis to detect the presence of SARS-CoV-2, the novel coronavirus that causes COVID-19.
To Prevent a Second Wave of COVID-19, AACC Calls on Senate to Include 5 Recommendations in Upcoming Coronavirus Bill
Now that the latest coronavirus relief package, known as the Heroes Act, has moved forward to the U.S. Senate, AACC has sent a letter to Senate leadership outlining five key recommendations that will improve COVID-19 testing capacity across the U.S. AACC urges the Senate to ensure these recommendations are addressed within the Heroes Act, as they are critical to preventing a second wave of the pandemic.
AACC Issues Recommendations on the Use of COVID-19 Antibody Tests
To ensure the proper use of antibody testing for the novel coronavirus, AACC today issued a public statement detailing the role these tests should play in the management of COVID-19 patients and in the development of public health policy. In particular, the organization emphasizes that healthcare professionals and policymakers should work closely with laboratory experts on antibody testing to ensure that these tests are validated, used appropriately, and interpreted correctly.
Not All FDA-Authorized COVID-19 Antibody Tests Are Equally Reliable
A first-of-its-kind study published today in AACC’s Clinical Chemistry journal compared the performance of two COVID-19 antibody tests and found that—even though both tests are FDA authorized—one produced more incorrect results than the other. These findings could help healthcare professionals and researchers to better select antibody tests, which are essential to both treating COVID-19 patients and determining the full extent of the outbreak.
Introducing the UC San Diego Return to Learn Program
Program makes COVID-19 testing available to thousands of students in effort to track the novel coronavirus and better position the campus to resume in-person activities in the fall.
AACI Calls for Coordinated National COVID-19 Testing Strategy
AACI urges the federal government to take the lead in deploying personal protective equipment to hospitals, establishing a consistent national COVID-19 testing strategy, and managing the COVID-19 testing supply chain.
The Nation Needs More Evidence Before Relying on Home Test Collection Kits for COVID-19
With the first home collection test kit for COVID-19 now authorized by the FDA, AACC is warning that more evidence is needed before the country can rely on home-based kits.
Online Map Tracks COVID-19 Testing Stations Throughout U.S.
Johns Hopkins Carey Business School Associate Professor Jim Kyung-Soo Liew leads a team that has created an online map showing the locations of coronavirus testing stations throughout the United States.
AACC Launches Free Video Series to Help Explain COVID-19 Testing
To address some of the confusion surrounding COVID-19 testing, AACC today launched a video series in which leading laboratory experts answer common questions about tests for the pandemic. From persistent supply shortages to the emergence of antibody tests, these videos quickly answer the questions that patients and the general public have been asking.
Lab Experts Can Fill Gaps in FDA Regulation by Validating COVID-19 Antibody Tests
Now that diagnostic companies can sell COVID-19 antibody tests without FDA authorization, healthcare teams should work closely with clinical laboratory experts to ensure that these tests are thoroughly validated and used appropriately. A new opinion piece in AACC’s Clinical Chemistry journal emphasizes that this is critical to minimizing the risk of inaccurate results from these tests, which could have potentially life-threatening consequences.

Working behind the scenes: UAB pathologists play key role in fighting coronavirus pandemic
Testing has been a major cause for concern worldwide ever since the pandemic began, but clinicians and researchers with UAB’s Department of Pathology have been working around the clock to make testing available for as many people as possible, making sure accurate results are available in a timely manner.

Indiana State Department of Health, IU partner on COVID-19 study
The Indiana University Richard M. Fairbanks School of Public Health at IUPUI is collaborating with the state health department to conduct a scientific study to measure the spread of COVID-19 throughout Indiana.