Researchers discover that a cytokine called interleukin-12 (IL-12) acts like a switch to direct which antibody response B cells will generate.
Tag: Antibodies

Nanotubes, nanoparticles, and antibodies detect tiny amounts of fentanyl
Using his platform composed of carbon nanotubes and gold nanoparticles, Professor Alexander Star added antibodies to detect the opioid. His sensor can also distinguish fentanyl from several other common opioids.
Moffitt Researchers Discover New Therapeutic Target for Non-Small Cell Lung Cancer
A new Moffitt Cancer Center study published in the journal Immunity offers insight into how lung cancer cells evade the protective immune system, potentially opening a door for novel antibody-based immunotherapies. Their study centers on a molecule called Jagged2, which plays a primary role in fueling the aggressiveness and immune evasion capacity of lung cancer.
New Study Reveals Insights into Lack of Durability in COVID Antibody Response to Infections and Vaccines
Researchers at the Institute of Human Virology (IHV) at the University of Maryland School of Medicine published a new study in the Journal of Infectious Diseases investigating the short-lived antibody response following SARS-CoV-2, the virus that causes COVID.
Breastfeeding after COVID-19 booster can give babies antibodies
A recently published study that shows lactating mothers who get the COVID-19 booster pass along the antibodies to their children via their breast milk – and potentially protect babies too young to receive the vaccine.
New antibody could target breast cancers
An enzyme that may help some breast cancers spread can be stopped with an antibody created in the lab of Cold Spring Harbor Laboratory Professor Nicholas Tonks.

New algorithm may fuel vaccine development
Immune system researchers have designed a computational tool to boost pandemic preparedness. Scientists can use this new algorithm to compare data from vastly different experiments and better predict how individuals may respond to disease.
Immune system changes behavior to promote allergen avoidance
The immune system plays a key role in changing our behaviors in response to environmental dangers, a new Yale School of Medicine study finds. Researchers found ova-allergic mice lost their avoidance behavior to the egg protein when Immunoglobulin E antibodies,…
Antibodies associated with rare disorder may signal future risk of heart attack and stroke
Seemingly healthy people whose blood contained antibodies associated with a condition called antiphospholipid syndrome (APS) were significantly more likely to experience a cardiovascular event such as a heart attack or stroke than those without, a study led by UT Southwestern Medical Center scientists shows
Finger-prick test developed for ‘trich’ a common, undiagnosed STI
A quick, affordable diagnostic test developed by a Washington State University researcher may help curb one of the most prevalent but least discussed sexually transmitted infections.
Boosting anti-cancer antibodies by reducing their grip
New research from the Centre for Cancer Immunology at the University of Southampton, published ahead of World Cancer Day (4 February), has shown that changing how tightly an antibody binds to a target could improve treatments for cancer.

LJI scientists uncover the structure and function of Inmazeb, the first FDA-approved drug for Ebola virus infection
Inmazeb (REGN-EB3), developed by Regeneron, is a three-antibody cocktail designed to target the Ebola virus glycoprotein. The drug was first approved for clinical use in October 2020, but its exact mechanism of action has remained unclear.
COVID-19 booster increases durability of antibody response, research shows
New research from the University of Virginia School of Medicine speaks to the benefits of a COVID-19 booster.
CHOP Researchers Find COVID-19 Vaccination Leads to Higher Antibody Levels than Natural Infection in Both Pregnant People and their Babies
Pregnant people who received one of the mRNA COVID-19 vaccines had 10-fold higher antibody concentrations than those who were naturally infected with SARS-CoV-2, a finding that was also observed in their babies, according to a new study by researchers at Children’s Hospital of Philadelphia (CHOP) and the University of Pennsylvania. The study, published today in JAMA Network Open, also found that vaccine timing played an important role in maximizing the transfer of antibodies, with antibodies detected as early as 15 days after the first vaccine dose and increasing for several weeks after.
Experimental monoclonal antibodies show promise against Epstein-Barr virus
A panel of investigational monoclonal antibodies (mAbs) targeting different sites of the Epstein-Barr virus (EBV) blocked infection when tested in human cells in a laboratory setting.
Vaccinating women infected with COVID during pregnancy prior to delivery provides antibodies to newborns
FINDINGS Women with COVID in pregnancy who are subsequently vaccinated after recovery, but prior to delivery, are more likely to pass antibodies on to the child than similarly infected but unvaccinated mothers are. Researchers who studied a mix of vaccinated and unvaccinated mothers found that 78% of their infants tested at birth had antibodies.

The longer the bootcamp, the better the antibodies
LJI research shows that a “slow delivery, escalating dose” vaccination strategy can prompt B cells to spend months mutating and evolving their pathogen-fighting antibodies.

Fast, efficient COVID-19 biosensor under development
IUPUI researchers are developing a “biosensing platform” for COVID-19 that’s fast, efficient, accurate and highly sensitive, which could help scientists stay on top of shifting virus variants.
Clever COVID-19
As new Omicron subvariants of COVID-19 continue to sweep across the United States, researchers at the University of Missouri have identified specific mutations within the virus’ spike protein that help Omicron subvariants evade existing antibodies humans have from either vaccines or previous COVID-19 infections.
Milk boost: Research shows how breastfeeding offers immune benefits
When infants breastfeed, they receive an immune boost that helps them fight off infectious diseases, according to recent research from Binghamton University, State University of New York.
New Antibody Detection Method for Coronavirus That Does Not Require a Blood Sample
Despite significant and stunning advances in vaccine technology, the COVID-19 global pandemic is not over.
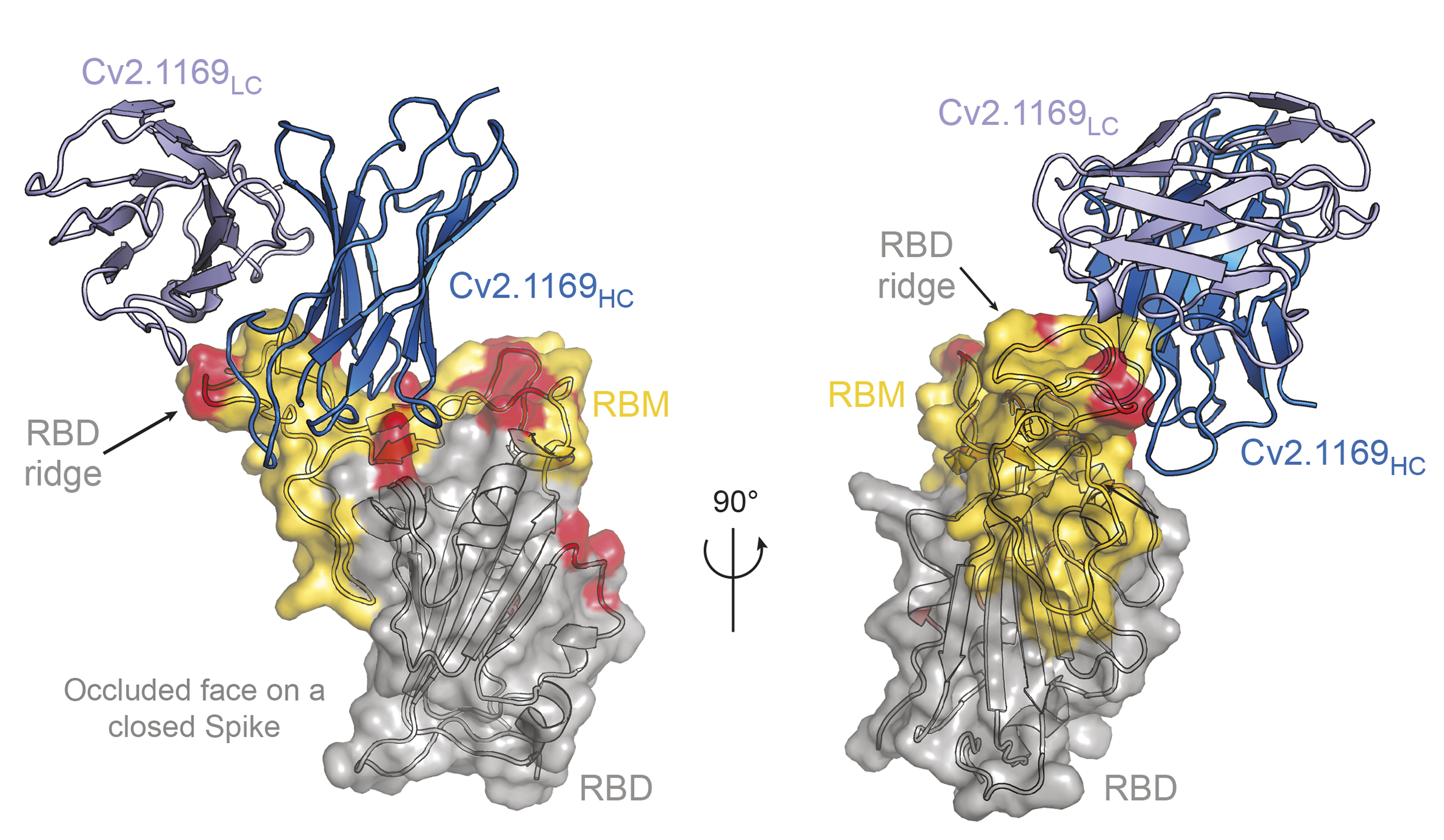
Broadly neutralizing antibodies could provide immunity against SARS-CoV-2 variants
Two broadly neutralizing antibodies show great promise to provide long-acting immunity against COVID-19 in immunocompromised populations according to a paper published June 15 in the Journal of Experimental Medicine (JEM). The antibodies were effective against all SARS-CoV-2 variants of concern tested and could be used alone or in an antibody cocktail to diminish the risk of infection.
Mount Sinai Researchers Develop a Rapid Test to Measure Immunity to COVID-19
Mount Sinai researchers have developed a rapid blood assay that measures the magnitude and duration of someone’s immunity to SARS-CoV-2, the virus that causes COVID-19. This test will allow large-scale monitoring of the population’s immunity and the effectiveness of current vaccines to help design revaccination strategies for vulnerable immunosuppressed individuals, according to a study published in Nature Biotechnology in June.
LJI scientists publish first head-to-head comparison of four COVID-19 vaccines
“Just understanding the immune responses to these vaccines will help us integrate what is successful into vaccine designs going forward.”
‘Nanobodies’ from Llamas Could Yield Cell-Specific Medications for Humans
In “proof of concept” experiments with mouse and human cells and tissues, Johns Hopkins Medicine researchers say they have designed tiny proteins, called nanobodies, derived from llama antibodies, that could potentially be used to deliver targeted medicines to human muscle cells.
COVID-19 vaccine effective in people receiving cancer treatment, study finds
About 95% of participants achieved a measurable immune response after vaccination, according to study published in JAMA Oncology.
Why Breakthrough COVID? Antibodies Fighting Original Virus May Be Weaker Against Omicron
If you’re wondering why after two vaccination doses and a booster shot, you still got sick from the omicron strain of the virus that causes COVID-19, one possible answer may have been found in a recent study by researchers at Johns Hopkins Medicine and the National Institute of Allergy and Infectious Diseases (NIAID) at the National Institutes of Health (NIH).
Some cases of long COVID-19 may be caused by an abnormally suppressed immune system, UCLA-led research suggests
Researchers studying the effect of the monoclonal antibody Leronlimab on long COVID-19 may have found a surprising clue to the baffling syndrome, one that contradicts their initial hypothesis. An abnormally suppressed immune system may be to blame, not a persistently hyperactive one as they had suspected.
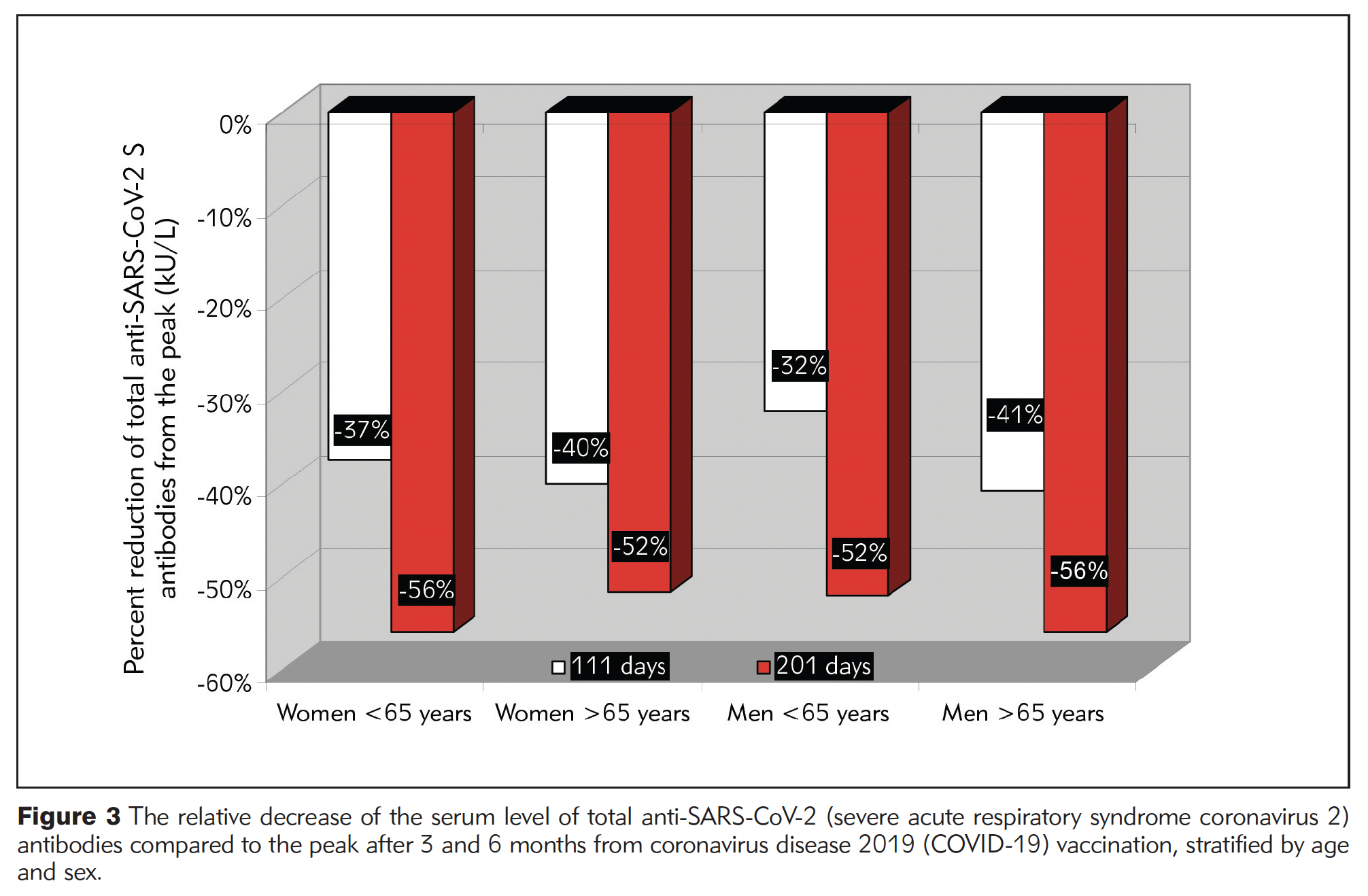
Age, sex and waning COVID-19 antibodies
As widely-anticipated decisions about COVID-19 vaccine boosters roll out from U.S. agencies today, insights from an independent study underscore why boosters are important for all adults.
COVID-19: The older you are, the more antibodies you have
Université de Montréal chemists looked at lab samples of patients who recovered from a mild case of COVID-19 and found that those over 50 produced more antibodies against the SARS-CoV-2 virus.
Duke, UNC-Chapel Hill Scientists Identify New Antibody For COVID-19 and Variants
A research collaboration between scientists at Duke University and the University of North Carolina at Chapel Hill has identified and tested an antibody that limits the severity of infections from a variety of coronaviruses, including those that cause COVID-19 as well as the original SARS illness.
New antibody detection platform offers accelerated multiplex detection of antibodies against pathogens or autoantibodies against biomarkers
Veravas has announced the development of its VeraPrep™ Antibody Detection Kit, a new development platform that provides scientists with a state-of-the-art research tool to detect and measure immunoglobulins against new and emerging pathogens.
From Alpha to Epsilon: Consortium study illuminates surfaces of Spike most resistant to antibody escape
Scientists at La Jolla Institute for Immunology (LJI) have published a detailed map of where human antibodies bind to SARS-CoV-2, a map that was generated by a global collaboration comparing nearly all leading clinical candidates. The new research will guide the development of more effective COVID-19 antibody therapies and help scientists develop effective vaccines to address emerging viral variants.
Llama antibodies have “significant potential” as potent COVID-19 treatment
A unique type of tiny antibody produced by llamas could provide a new frontline treatment against Covid-19 that can be taken by patients as a simple nasal spray.
Pregnant Women Who Receive COVID-19 Vaccination Pass Protection from the Virus to Their Newborns
Women who receive COVID-19 mRNA vaccines during pregnancy pass high levels of antibodies to their babies, a new NYU Langone study finds.
Researchers explain how nanomaterial aids antibody response, study it as antibody factory
Iowa State researchers affiliated with the Nanovaccine Institute have explained how a nanomaterial initiates antibody production by the immune system’s B cells. The technique could be used to turn B cells into factories that provide antibodies for diagnostic tests or treatments.
UTHealth researcher awarded CPRIT grant to test potential large-molecule cancer drugs at preclinical development core facility
A preclinical development core where researchers can test the effectiveness of large molecule drug candidates for novel cancer treatments, led by Qingyun Liu, PhD, has been awarded a nearly $4 million grant from the Cancer Prevention and Research Institute of Texas (CPRIT).
$1.3 million in NIH grants to enable research into antibody-mediated drug delivery technology
Two National Institute of Health (NIH) grants totaling over $1.3 million will enable research into antibody-mediated drug delivery technology for the treatment of cancer and autoimmune disorders. L. Nathan Tumey, assistant professor of pharmaceutical sciences, is the Principal Investigator on both grants — $1.2 million from the National Institute of General Medical Sciences and $150,000 from the National Institute of Allergy and Infectious Diseases.
Breast milk of mothers who received COVID-19 vaccine contains antibodies that fight illness
The breast milk of lactating mothers vaccinated against COVID-19 contains a significant supply of antibodies that may help protect nursing infants from the illness, according to new research from the University of Florida.
Surveillance study finds disparities, high proportion of past COVID-19 infections among adults and children in Santa Ana
In a large-scale, population-based surveillance conducted in partnership with the City of Santa Ana, researchers at the University of California, Irvine’s Program in Public Health found 27% positivity of SARS-CoV-2 antibodies among participating Santa Ana residents. This unique study was one of the first to examine household transmission of COVID-19 and to include a pediatric population (ages 5+).
Antibody protects against broad range of COVID-19 virus variants
Researchers at Washington University School of Medicine in St. Louis have identified an antibody that is highly protective against a broad range of viral variants.
World-first COVID vaccine booster randomized clinical trial in transplant patients proves third shot is very effective
The study enrolled 120 transplant patients between May 25th and June 3rd. None of them had COVID previously and all of them had received two doses of the Moderna vaccine. Half of the participants received a third shot of the vaccine (at the 2-month mark after their second dose) and the other half received placebo.
The primary outcome was based on antibody level greater than 100 U/ml against the spike protein of the virus. In the placebo group – after three doses (where the third dose was placebo), the response rate was only 18% whereas in the Moderna three-dose group, the response rate was 55%.
Hopkins Med News Update
NEWS STORIES IN THIS ISSUE:
– COVID-19 NEWS: Johns Hopkins Medicine Study Shows Vaccine Likely Protects People with HIV
– Johns Hopkins Medicine Documents Stroke Risk in Cardiac Assist Device
– CBD Products May Help People with Epilepsy Better Tolerate Anti-Seizure Medications
COVID-19 infection rate low among rural health care workers
Antibody testing of health care workers in three rural counties in eastern South Dakota and western Minnesota showed 15% had antibodies to the novel coronavirus.
Highly Potent, Stable Nanobodies Stop SARS-CoV-2
Göttingen researchers have developed mini-antibodies that efficiently block the coronavirus SARS-CoV-2 and its dangerous new variants.
Novel autoantibody adds fuel to COVID-19 “firestorm” of inflammation, blood clots
Researchers at Michigan Medicine have discovered another functional autoantibody in COVID-19 patients that contributes to the disease’s development and the “firestorm” of blood clots and inflammation it induces. The autoantibody makes it harder for the body to degrade neutrophil extracellular traps, the toxic webs of DNA and proteins produced by overactive immune cells at heightened levels in COVID patients.
Had COVID-19? One vaccine dose enough; boosters for all, study says
A new study in ACS Nano supports increasing evidence that people who had COVID-19 need only one vaccine dose, and that boosters could be necessary for everyone in the future.
Engineering nanobodies as lifesavers when SARS-CoV-2 variants attack
Scientists are pursuing a new strategy in the protracted fight against the SARS-CoV-2 virus by engineering nanobodies that can neutralize virus variants in two different ways.
Story Tips from Johns Hopkins Experts on COVID-19
NEWS STORIES IN THIS ISSUE:
-Physician and Musician: Johns Hopkins Doctor Brings Passion for Music to Medicine During Pandemic
-Rapid, At-Home Blood Test Could Confirm COVID-19 Vaccination in Minutes
-What to Expect and Prepare for As You Return to Regular Health Care Appointments
-Study Suggests Sudden Hearing Loss Not Associated with COVID-19 Vaccination
-Vaccination May Not Rid COVID-19 Risk for Those with Rheumatic, Musculoskeletal Diseases
Preclinical research reveals that new IgM antibodies administered intranasally to fight COVID-19 more potent than commonly used ones
A nasal therapy, built upon on the application of a new engineered IgM antibody therapy for COVID-19, was more effective than commonly used IgG antibodies at neutralizing the COVID-19 virus in animal models, according to research recently published by The University of Texas Health Science Center at Houston (UTHealth), The University of Texas Medical Branch at Galveston (UTMB Health), the University of Houston, and IGM Biosciences, Inc.