Digital Science solutions metaphacts and Dimensions announce the launch of the Dimensions Knowledge Graph, a large ready-made knowledge graph powering AI solutions in the pharmaceutical and life sciences industries.
Tag: Pharmaceutical
Cole-Parmer Re-launches Fluid Handling Product Line
Cole-Parmer, an Antylia Scientific company, is launching an all-new range of industry-leading fluid handling solutions initially comprised of tubing, fittings, parts, and accessories to suit a diverse array of research and production needs.
Positive Results from Diabetic Foot Ulcer Clinical Trial Show Dramatic Healing Rate within 12 Weeks or Less Using Novel Omeza® Platform and Offloading
Omeza®, a regenerative skincare company that develops marine-based therapies for the treatment of chronic wounds, today presented positive interim data from a diabetic foot ulcer (DFU) clinical trial showing that Omeza® OCM™ plus offloading of weight on the affected foot achieved a 91 percent area reduction (PAR) rate in DFUs within twelve weeks, and a 63 percent PAR within four weeks.
Taxpayers should foot the bill if EU demands efficient removal of pharmaceuticals from wastewaters
Public sector should pay if EU demands efficient removal of pharmaceuticals from wastewaters, according to researchers at the Centre for Antibiotic Research, CARe, at the University of Gothenburg.
Machine learning tool simplifies one of the most widely used reactions in the pharmaceutical industry
University of Illinois researchers and a Swiss pharmaceutical company have developed a machine learning model that eliminates the need for extensive experimentation to determine the best conditions for an important carbon-nitrogen bond forming reaction known as the Buchwald-Hartwig reaction.
Medicare drug price news: Johns Hopkins Bloomberg School of Public Health experts available
Two experts from the Johns Hopkins Bloomberg School of Public Health are available for media interviews to discuss today’s announcement identifying the 10 drugs that will be subject to price negotiations under the Medicare program, as directed by the Centers…
Digital Science boosts pharma industry support following OntoChem acquisition
Digital Science is positioning itself to play an even greater role in the pharmaceutical industry’s all-important drug discovery, by helping industry sift through a sea of information and focus on the research that matters.

Digital Science acquires knowledge graph and decision intelligence software company metaphacts
Digital Science has completed the acquisition of metaphacts, which has become the newest member of the Digital Science family.
Scientists invent pioneering technique to construct rare molecules discovered in sediments from the Bahamas with potential to help treat disease and infection
Scientists have created a much faster way to make certain complex molecules, which are widely used by pharmaceuticals for antibiotics and anti-fungal medicines.
New Center for Experimental Therapeutics in Cancer advances promising treatments
UC Davis Comprehensive Cancer Center has launched a Center for Experimental Therapeutics in Cancer to accelerate promising cancer therapies from the lab to the bedside. The idea is to advance precision medicine that leads to more effective, less toxic cancer therapies.
Researchers discover potential treatment for Chagas disease
Researchers from the University of Georgia have discovered a potential treatment for Chagas disease, marking the first medication with promise to successfully and safely target the parasitic infection in more than 50 years. Human clinical trials of the drug, an antiparasitic compound known as AN15368, will hopefully begin in the next few years.
Enhancing the effect of protein-based COVID-19 vaccines
Adding an ingredient called an adjuvant can help vaccines elicit a more robust immune response. In a study in ACS Infectious Diseases, researchers report a substance that boosted the immune response to an experimental COVID-19 shot in mice by 25 times, compared to injection with the vaccine alone.
Media Briefing Schedule for ACS Fall 2022
Media Briefing Schedule for ACS Fall 2022
Evolution Research Group, LLC Acquires Ohio Clinical Trials, LLC Further Expanding Clinical Pharmacology & Early Stage Development Service Capabilities
Evolution Research Group, LLC (ERG), a privately held, independent pharmaceutical services provider focused on neuroscience announced the acquisition of Ohio Clinical Trials (OCT), a 64-bed phase 1 unit in Columbus, OH, that specializes in the execution of human abuse potential (HAP) studies, as well as other highly complex early phase trials including pain modeling, qEEG/EEG, respiratory depression, and alcohol interaction, among others.
New Report Provides Strategies for Managing Contrast Shortage
Recent disruptions in a pharmaceutical supply chain have impacted the global availability of GE Healthcare Omnipaque™ iohexol iodinated contrast media (ICM) for radiologic examinations. A new Special Report published in the journal Radiology provides consensus recommendations for dealing with the shortage of ICM in the near term and discusses long-term issues and potential solutions to supply chain problems.
$1.3 million in NIH grants to enable research into antibody-mediated drug delivery technology
Two National Institute of Health (NIH) grants totaling over $1.3 million will enable research into antibody-mediated drug delivery technology for the treatment of cancer and autoimmune disorders. L. Nathan Tumey, assistant professor of pharmaceutical sciences, is the Principal Investigator on both grants — $1.2 million from the National Institute of General Medical Sciences and $150,000 from the National Institute of Allergy and Infectious Diseases.
Bill Greene, PharmD, of St. Jude receives award for outstanding contributions to the pharmacy profession
William L. “Bill” Greene, PharmD, chief pharmaceutical officer, to receive the 2021 Shelby Rhinehart Public Service Lifetime Achievement Award.
Improved method for generating synthetic data solves major privacy issues in research
Researchers at the Finnish Center for Artificial Intelligence have developed a machine learning-based method that produces synthetic data, making it possible for researchers to share even sensitive data with one other without privacy concerns.
NUS researchers develop novel technique to automate production of pharmaceutical compounds
Giving a new spin to conventional chemical synthesis, a team of researchers from the National University of Singapore (NUS) has developed a way to automate the production of small molecules suitable for pharmaceutical use. The method can potentially be used for molecules that are typically produced via manual processes, thereby reducing the manpower required.
Researchers develop better way to determine safe drug doses for children
To make drugs and their development safer for children, researchers at Aalto University and the pharmaceutical company Novartis have developed a method to help determine safe drug doses more quickly. The new organ maturation model is more data-driven and consequently less prone to bias.

Leveraging Modeling and Simulation in Medicine at VisualizeMED
ASME’s VisualizeMED: Modeling and Simulation in Medicine will take place on April 14-15, 2021. This two-day virtual event is enabling the transformation of modeling and simulation in medicine by bringing together industry experts of technology and masters of technique who are effectively implementing it with the goal to increase the application and adoption on a global scale.
Researchers watch anti-cancer drug release from DNA nanostructures in real time
A team of researchers from Finland and Germany have found a way to study the endonuclease-driven digestion of drug-loaded DNA nanostructures in real time. As the team investigated the binding of anti-cancer drug doxorubicin (Dox) to the DNA structures in great detail, they discovered that the majority of previous studies have vastly overestimated the Dox loading capacity of DNA origami.
Case Western Reserve University teams with Boehringer Ingelheim on artificial intelligence solutions for precision medicine
The Center for Computational Imaging and Personalized Diagnostics (CCIPD) at Case Western Reserve University and Boehringer Ingelheim, one of the world’s largest pharmaceutical companies will leverage the power of CCIPD’s image computing AI solutions to identify patterns and links between cellular response and underlying molecular drivers, with the goal of advancing therapeutics for diseases with no satisfactory treatment option.
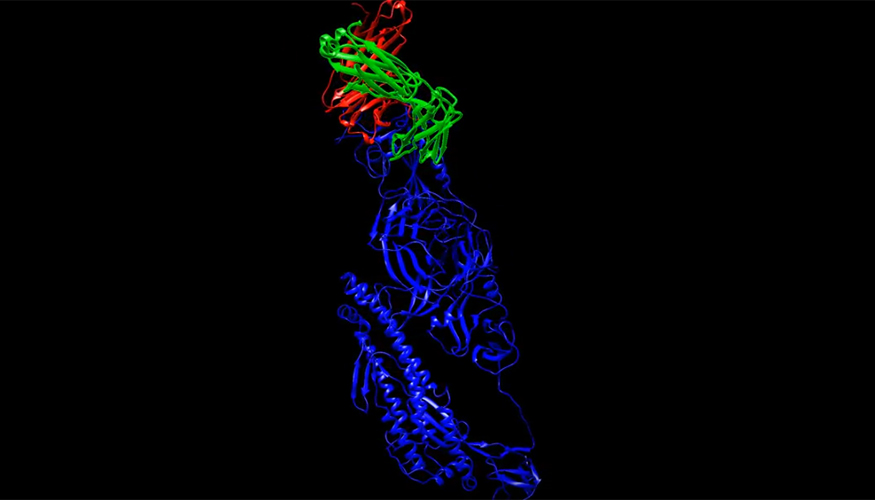
Design and test potential COVID-19 treatments from your phone
Anyone with a smartphone can download the app ViDok, which lets users pick from a library of molecules that might bind to key proteins on the SARS-CoV-2 virus, which causes COVID-19, and then can tweak the molecules to try to find a better fit.
Nephron Pharmaceuticals Corporation Announces Historic Expansion in Lexington County
One of the fastest-growing pharmaceuticals companies in the country, headquartered in Lexington County, today announced several major expansion and investment projects.

One in six medications prescribed to older people causing more harm than good
Millions of people around the world over the age of 70 are taking at least one medication every day that is causing them more harm than good, leading to falls, confusion, hospitalisation and even death.
Optimizing Patient Access to Medications: The American Dermatological Association’s Position Statement on Pharmacy Benefit Managers (PBMs) and Patient Access to Affordable and Appropriate Medications
Access to healthcare has been at the forefront of social and political debate for decades. Reliable and equitable access to provider prescribed medications is tantamount to the delivery of appropriate healthcare, and the lifecycle of medication manufacturing, distribution, pricing and procurement has been shrouded in an incomprehensible array of transactions and involved stakeholders. Among the middlemen interspersed between pharmaceutical manufacturers and patients are pharmacy benefit managers (PBM). Initially tasked with administering drug plans for health insurers,1 the role of PBMs has expanded over time. They currently function in a lightly regulated area,2,3 with few requirements for business transparency. Three PBMs, CVS Caremark, Optum RX, and Express Scripts, control distribution of nearly ¾ of the medications in the United States.
Lab researchers aid COVID-19 response in antibody, anti-viral research
Lawrence Livermore National Laboratory scientists are contributing to the global fight against COVID-19 by combining artificial intelligence/machine learning, bioinformatics and supercomputing to help discover candidates for new antibodies and pharmaceutical drugs to combat the disease.

COVID-19 big picture: For many years, Pinar Keskinocak has studied how society and the nation handle pandemics.
For many years, Pinar Keskinocak has studied how pandemics spread through the nation, how they overburden health care systems, and how they diminish the supply of medications, thus worsening the pandemic. All this also spins off additional medical crises. She…
Coronavirus multiple-times worse than SARS: Global supply-chain effect could exceed $400bn, linger up to 2 years — WashU expert
Panos Kouvelis 314-935-4604 [email protected] Please read: https://source.wustl.edu/2020/02/washu-expert-coronavirus-far-greater-threat-than-sars-to-global-supply-chain/ Please watch: https://youtu.be/ATzgs67Dnx8 Original post https://alertarticles.info