The humanized monoclonal antibody known as hu1F5, which specifically binds to the fusion (F) glycoprotein of both Nipah virus and Hendra virus and prevents virus infection of cells (neutralizes) has now proven effective in protecting against the often fatal Nipah virus in animal studies.
Tag: Monoclonal Antibodies
New Potential Immunotherapies for Pancreatic Cancer
Pancreatic cancer can be a devastating diagnosis due to its extremely aggressive nature and low survival rate.
MD Anderson and Panacea launch Manaolana Oncology to develop antibody-based therapies for cancer
MD Anderson and Panacea Venture announced the launch of Manaolana Oncology, a new company created to develop and advance antibody-based therapies against novel cancer targets.
AAN Issues Guidance on New Treatments for Early Alzheimer’s Disease
New therapies for early Alzheimer’s disease, monoclonal antibodies that remove amyloid-β plaques in the brain, are bringing hope to people whose lives have been affected by the disease. To help neurologists discuss these therapies with patients and caregivers, the American Academy of Neurology has developed an Emerging Issues in Neurology article, published online on July 26, 2023, in Neurology®, the medical journal of the American Academy of Neurology.
Lab-made antibodies offer potential cure for yellow fever
New research from Oregon Health & Science University and collaborators indicates lab-made antibodies may be able to cure people infected with yellow fever, a virus for which there is no treatment.
Small Study Shows Promise for Antimalarial Monoclonal Antibody to Prevent Malaria
monoclonal antibody treatment was found to be safe, well tolerated, and effective in protecting against malaria in a small group of healthy volunteers who were exposed to malaria in a challenge study, according to new research published in The Lancet Infectious Diseases by researchers at the University of Maryland School of Medicine (UMSOM).
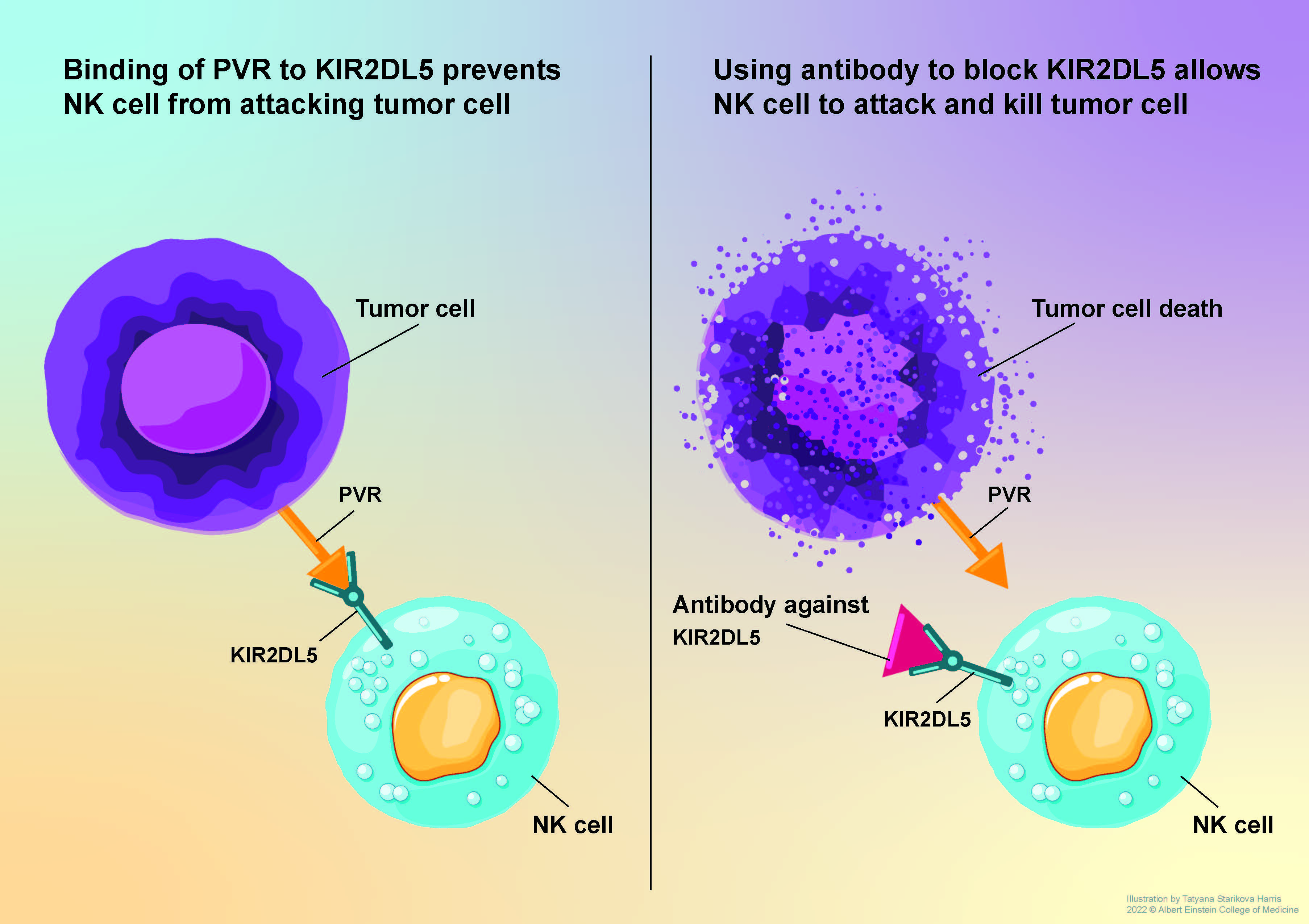
Albert Einstein College of Medicine Researchers Develop Promising New Cancer Therapy
Immune checkpoint inhibitors such as Keytruda and Opdivo work by unleashing the immune system’s T cells to attack tumor cells. Their introduction a decade ago marked a major advance in cancer therapy, but only 10% to 30% of treated patients experience long-term improvement. In a paper published online today in The Journal of Clinical Investigation (JCI), scientists at Albert Einstein College of Medicine describe findings that could bolster the effectiveness of immune-checkpoint therapy.
Could monoclonal antibodies replace opioids for chronic pain?
During the pandemic, physicians used infusions of monoclonal antibodies to help patients fight off COVID-19 infections. Now, in response to the U.S. opioid crisis, researchers at UC Davis are trying to create monoclonal antibodies that can help fight chronic pain. The research is funded with a $1.5 million grant from the National Institutes of Health’s HEAL Initiative, an aggressive effort to speed scientific solutions to stem the national’s opioid crisis.
Good news on blocking a virus considered a global threat
Scientists have reported good news on the pandemic preparedness front: A cocktail of four manufactured antibodies is effective at neutralizing a virus from the Henipavirus family, a group of pathogens considered to be a global biosecurity threat.
Some cases of long COVID-19 may be caused by an abnormally suppressed immune system, UCLA-led research suggests
Researchers studying the effect of the monoclonal antibody Leronlimab on long COVID-19 may have found a surprising clue to the baffling syndrome, one that contradicts their initial hypothesis. An abnormally suppressed immune system may be to blame, not a persistently hyperactive one as they had suspected.
Expert Alert: 5 ways patients who are immunocompromised can protect themselves from COVID-19
As families prepare to gather later this month for Thanksgiving, it is important for patients who are immunocompromised to take extra steps to protect themselves from becoming infected with COVID-19. People who are immunocompromised have weakened immune systems, which means they have a higher risk of becoming severely ill from COVID-19.
Monoclonal antibody treatment highly effective at reducing COVID-19 hospitalizations
Researchers published interim results in The New England Journal of Medicine from a Phase 3 study of the COVID-19 monoclonal antibody treatment sotrovimab, sponsored by Vir Biotechnology and GlaxoSmithKline. The study found that compared to the placebo group, COVID-19 patients who received sotrovimab had a significantly reduced risk of hospitalization or death and that the treatment, which was administered by intravenous infusion on an outpatient basis, was safe.
Researchers show low uptake of only FDA-authorized monoclonal antibody treatments for COVID-19
A new analysis by a team of physician-researchers at Beth Israel Deaconess Medical Center (BIDMC) provides the first evidence that monoclonal antibodies were indeed underutilized in the first six months of FDA authorization.
Monoclonal antibodies help COVID-19 patients avoid hospitalization
In a large observational study, Mayo Clinic researchers have shown that two monoclonal antibodies administered separately helped prevent hospitalization in high-risk patients who became infected with COVID-19. The study also showed more hospitalizations were observed among patients with more comorbidities. The findings appear in The Journal of Infectious Diseases.
COVID-19 dual-antibody therapies effective against variants in animal study
A study from Washington University School of Medicine in St. Louis suggests that many, but not all, COVID-19 therapies made from combinations of two antibodies are effective against a wide range of virus variants, and that combination therapies appear to prevent the emergence of drug resistance.
Monoclonal antibody therapy for COVID-19 safe, effective for transplant patients
Treating transplant patients with mild to moderate cases of COVID-19 with monoclonal antibodies is safe and helps prevent serious illness, according to a Mayo Clinic study recently published in Open Forum Infectious Diseases. These results are especially important because transplant patients who are infected with COVID-19 have a higher risk of severe illness and death.
Houston Methodist and U.S. Department of Health and Human Services partner to expand access to COVID-19 monoclonal antibody treatment
Houston Methodist announced today, in partnership with the U.S. Department of Health and Human Services (HHS), that it has expanded access to COVID-19 monoclonal antibody therapy (mAb) in the Greater Houston area. The promising treatment has been shown to keep high-risk COVID-19 patients out of the hospital and reduce the likelihood of progression to severe disease.
Study Shows Significant Reduction in Triglycerides in Patients at High Risk for Acute Pancreatitis Through Novel Monoclonal Antibody
The investigational drug evinacumab reduced triglycerides in patients with severe hypertriglyceridemia (sHTG) and a history of hospitalizations for acute pancreatitis in a phase 2 global study led by Mount Sinai
Atlantic Health System Physicians Co-Author 5 Studies, Presented at American Association for Cancer Research (AACR) Annual Meeting
Atlantic Health System Cancer Care physicians are co-authors of five original studies presented at this year’s AACR Annual Meeting, held virtually April 10-15 and May 17-21. The AACR meeting is one of the world’s premier scientific gatherings of cancer specialists and researchers.
Leading the Way in Practical Treatment of COVID-19
A Michigan Medicine team’s online guidelines have been viewed more than 30,000 times by providers in 150 countries since the beginning of the pandemic–and their deployment of an effective COVID-19 therapy has been a model for health systems and hospitals statewide.
Monoclonal Antibody “Cocktail” Blocks COVID-19 Variants: Study
A monoclonal antibody “cocktail” developed at Vanderbilt University Medical Center (VUMC) to neutralize the COVID-19 virus is effective against all known strains, or variants, of the virus, according to a report published in the journal Nature Medicine.
Repurposed Arthritis Drug Did Not Significantly Improve Severe COVID-19 Pneumonia
A repurposed drug used to treat arthritis did not significantly improve the outcomes of patients with severe COVID-19 pneumonia. Tocilizumab did not significantly improve clinical status or mortality rate at 28 days for participants who received it compared to a placebo.
Antibody-based COVID-19 treatments work best in concert with immune cells
Antibody-based drugs have been authorized for emergency use in COVID-19 patients by the Food and Drug Administration. Researchers at Washington University School of Medicine in St. Louis have discovered that the ability to interact with other elements of the immune system is an indispensable part of the effectiveness of such antibodies. The findings could help improve the design of the next generation of antibody-based COVID-19 drugs.

COVID-19: Monoclonal Antibody Drugs Available for High-Risk Patients
UVA Health is offering monoclonal antibody drugs for appropriate patients with COVID-19 who are at highest risk for developing severe symptoms and requiring hospitalization.

Free Webinar—Next Steps in Eradicating COVID-19: Emerging Treatments and Vaccines
In this free STS webinar, Next Steps in Eradicating COVID-19: Emerging Treatments and Vaccines, STS President Dr. Joseph Dearani will talk with infectious disease experts from the White House Coronavirus Task Force and Mayo Clinic about promising new treatments and preventive measures.
Principal Investigator for COVID-19 drug with FDA Emergency Use Authorization available to speak on limited government supply of drug and pending clinical trials
COVID-19 and Monoclonal Antibodies – Emergency Use Authorization for Bamlanivimab The FDA’s Emergency Use Authorization to treat high risk COVID-19 patients with the monoclonal antibody bamlanivimab will offer limited availability in Texas, where only 6,000 government doses are expected next…

Keck Medicine of USC is enrolling individuals in a phase 3 clinical trial to test monoclonal antibody treatment for COVID-19
Keck Medicine of USC is enrolling individuals in a phase 3 clinical trial to test monoclonal antibody treatment for COVID-19
UIC opens phase 3 monoclonal antibody clinical trial
Researchers at the University of Illinois Chicago are now enrolling people into a phase 3 clinical trial that will test if a monoclonal antibody treatment — administered as a series of four shots — will help protect uninfected individuals from acquiring or getting sick from COVID-19 after someone in the household tests positive for COVID-19.
Woman recovering from COVID-19 shares experience as monoclonal antibody clinical trial participant
When Christina Loville tested positive for the coronavirus, she was terrified. She decided to channel her fear into researching COVID-19 treatments, where she discovered a local clinical trial led by experts at The University of Texas Health Science Center at Houston (UTHealth).
Key details about broadly neutralizing antibodies provide insights for universal flu vaccine
New research from an immunology team at the University of Chicago may shed light on the challenges of developing a universal flu vaccine that would provide long-lasting and broad protection against influenza viruses.
Fred Hutch opens COVID-19 Clinical Research Center
SEATTLE — Oct. 5, 2020 — Fred Hutchinson Cancer Research Center announced the opening of the COVID-19 Clinical Research Center, or CCRC. Funded by philanthropic donations and public/private partnerships, the CCRC is one of the first stand-alone facilities in the nation designed to test novel interventions to treat and prevent COVID-19.
Team assessing if dual-antibody injection prevents COVID-19 illness
A combination antibody treatment for preventing COVID-19 illness in individuals who have had sustained exposure to someone with the virus is being studied by researchers at The University of Texas Health Science Center at Houston (UTHealth). The clinical trial is enrolling patients at Harris Health System’s Lyndon B. Johnson Hospital.
UTHealth joins NIH trial to test antibodies and other experimental outpatient treatments for mild COVID-19 pneumonia
A study on the effectiveness of multiple treatments, including laboratory-made antibodies, at preventing mild COVID-19 from advancing to severe illness in the outpatient setting is underway by researchers at The University of Texas Health Science Center at Houston (UTHealth). The clinical trial is enrolling patients at Harris Health System’s Lyndon B. Johnson Hospital.

First Patients in NIH ACTIV-3 Clinical Trial Enroll in Dallas
On Wednesday in Dallas, just one day after the initiative was launched by the National Institutes of Health (NIH), Baylor Scott & White Research Institute enrolled the first patient in the world for the ACTIV-3 clinical trial. A second patient was enrolled the following day.
Imitation May Be a Sincere Form of Treatment
The National Institutes of Health will soon launch a phase II clinical trial to evaluate the safety and efficacy of potential new therapeutics for COVID-19, including the use of investigational synthetic monoclonal antibodies. Davey Smith of UC San Diego is the protocol chair and answers questions.

Baylor Scott & White Research Institute Expands Efforts in the Fight Against COVID-19
As the global response to the SARS-COV-2 virus that causes COVID-19 approaches 200 days, Baylor Scott & White Research Institute, the research and development arm of Baylor Scott & White Health, is accelerating its pace of bringing clinical trials online.
Baylor Scott & White Research Institute continues to mobilize staff and resources, including components needed to integrate critical patient-safety measures at every participating site within the Baylor Scott & White system for industry sponsored drug trials, investigator-initiated drug trials and research studies, and observational and data studies designed to help increase knowledge around case trends, viral epidemiology, and care best practices.