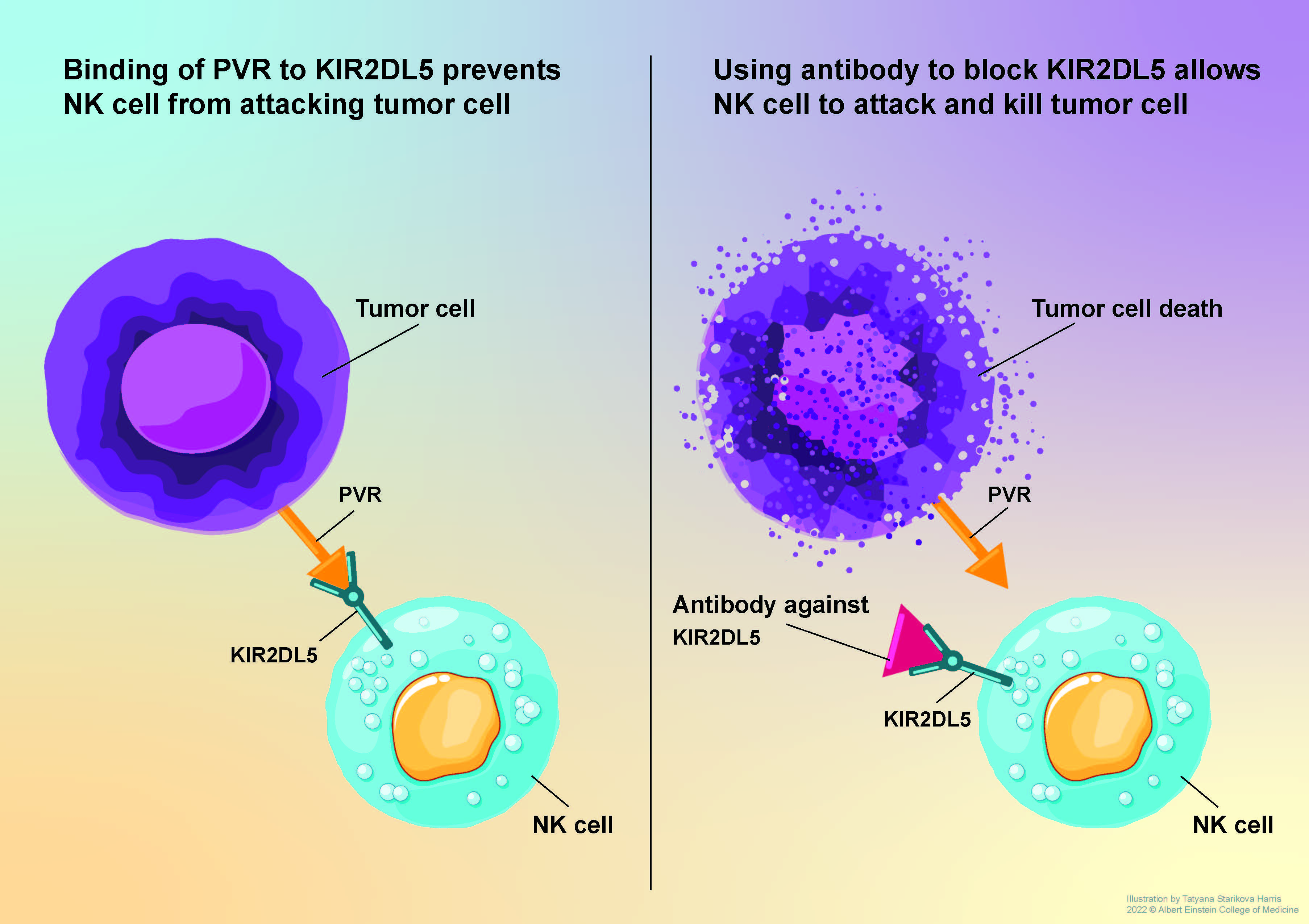
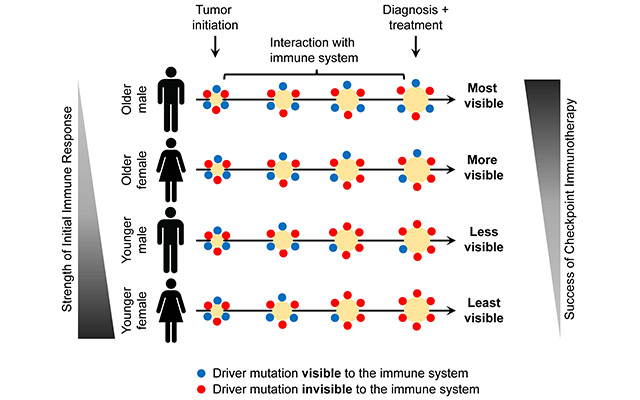
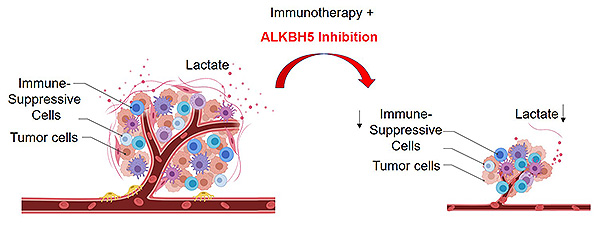
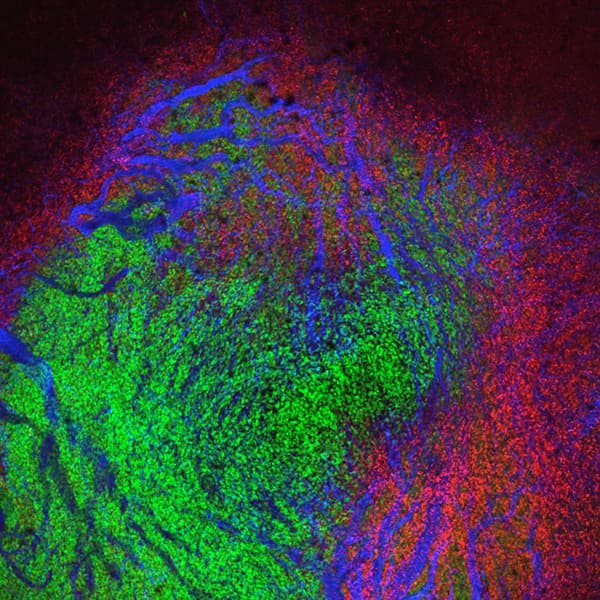
Immune checkpoint inhibitors such as Keytruda and Opdivo work by unleashing the immune system’s T cells to attack tumor cells. Their introduction a decade ago marked a major advance in cancer therapy, but only 10% to 30% of treated patients experience long-term improvement. In a paper published online today in The Journal of Clinical Investigation (JCI), scientists at Albert Einstein College of Medicine describe findings that could bolster the effectiveness of immune-checkpoint therapy.