ASCO, the largest event in cancer research, is approaching on May 31st. This year’s meeting promises to be particularly groundbreaking, with new findings and innovations that could revolutionize cancer treatment.
Tag: immunotheraphy
Immunotherapy for Alzheimer’s disease shows promise in mouse study
Scientists at Washington University School of Medicine in St. Louis have shown that treating mice with an antibody that blocks the interaction between APOE proteins (white) sprinkled within Alzheimer’s disease plaques and the LILRB4 receptor on microglia cells (purple) activates them to clean up damaging plaques (blue) in the brain.
Clinical Trial Results Published in Nature Medicine Show Immunotherapy’s Potential in Resectable Esophageal and Gastroesophageal Junction Cancers and the Benefits of Monitoring Circulating Tumor DNA (ctDNA) to Measure Disease Response
The results of a study published today in Nature Medicine show exciting immune responses in patients with operable esophageal or gastroesophageal cancers given neoadjuvant immunotherapy. The study results also show the potential for monitoring circulating tumor DNA as a predictor for future intervention.
New Clinical Practice Guideline Provides Evidence-based Recommendations for Immunotherapy for Inhalant Allergy
The American Academy of Otolaryngology–Head and Neck Surgery Foundation (AAO-HNSF) published the Clinical Practice Guideline: Immunotherapy for Inhalant Allergy today in Otolaryngology–Head and Neck Surgery.
New Potential Immunotherapies for Pancreatic Cancer
Pancreatic cancer can be a devastating diagnosis due to its extremely aggressive nature and low survival rate.
Adding Immunotherapy to Chemotherapy Regimen Improves Survival in Metastatic Bladder Cancer Patients
A clinical trial co-led by Mount Sinai researchers is the first to show that using chemotherapy with immunotherapy resulted in improved survival in patients with an advanced type of bladder cancer. The results were simultaneously reported in The New England Journal of Medicine and at the annual meeting of the European Society for Medical Oncology.
Thailand Hub of Talent for Cancer Immunotherapy International Conference
The Faculty of Medicine, Chulalongkorn University, cordially invites all to attend the “Thailand Hub of Talent for Cancer Immunotherapy International Conference: Portal to Global Collaboration for Next Generation Cell and Gene Therapy Development” on November 16-17, 2023 at Bhumisiri Mangklanusorn Building, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand.

Combination immunotherapy treatment effective before lung cancer surgery
Combination immunotherapy with the anti-PD-L1 monoclonal antibody durvalumab and other novel agents outperforms durvalumab alone in the neoadjuvant (pre-surgical) setting for early-stage non-small-cell lung cancer (NSCLC), according to researchers at The University of Texas MD Anderson Cancer Center.
Trial: Combination Immunotherapy Prior To Lung Cancer Surgery Appears Promising
Patients with stage 1–3 non-small cell lung cancer (NSCLC) given a combination immunotherapy prior to surgery (neoadjuvant) had a better major pathological response (10% or less residual cancer) when the cancer was removed than those who took a single immunotherapy agent alone, according to results of a novel multicenter phase 2 NeoCOAST platform trial that included two investigators from the Johns Hopkins Kimmel Cancer Center and its Bloomberg-Kimmel Institute for Cancer Immunotherapy. The results were published online Sept. 14 in Cancer Discovery, a journal of the American Association for Cancer Research.
CAR-T immune therapy attacks ovarian cancer in mice with a single dose
CAR-T immune therapies could be effective against solid tumors if the right targets are identified, a new study led by University of Illinois Urbana-Champaign researchers suggests. The researchers successfully deployed CAR-T in a mouse model of ovarian cancer, a type of aggressive, solid-tumor cancer that has eluded such therapies until now.
New Computational Tool Identifies Novel Targets for Cancer Immunotherapy
Researchers at Children’s Hospital of Philadelphia (CHOP) and the University of California, Los Angeles (UCLA) have developed a computational platform capable of discovering tumor antigens derived from alternative RNA splicing, expanding the pool of cancer immunotherapy targets. The tool, called “Isoform peptides from RNA splicing for Immunotherapy target Screening” (IRIS), was described in a paper published today in the Proceedings of the National Academy of Sciences.

Targeting uncontrolled inflammation may hold the key to treating therapy-resistant cancers
Van Andel Institute scientists have pinpointed how a specific gene mutation triggers an inflammatory cascade that may drive development of treatment-resistant cancers.
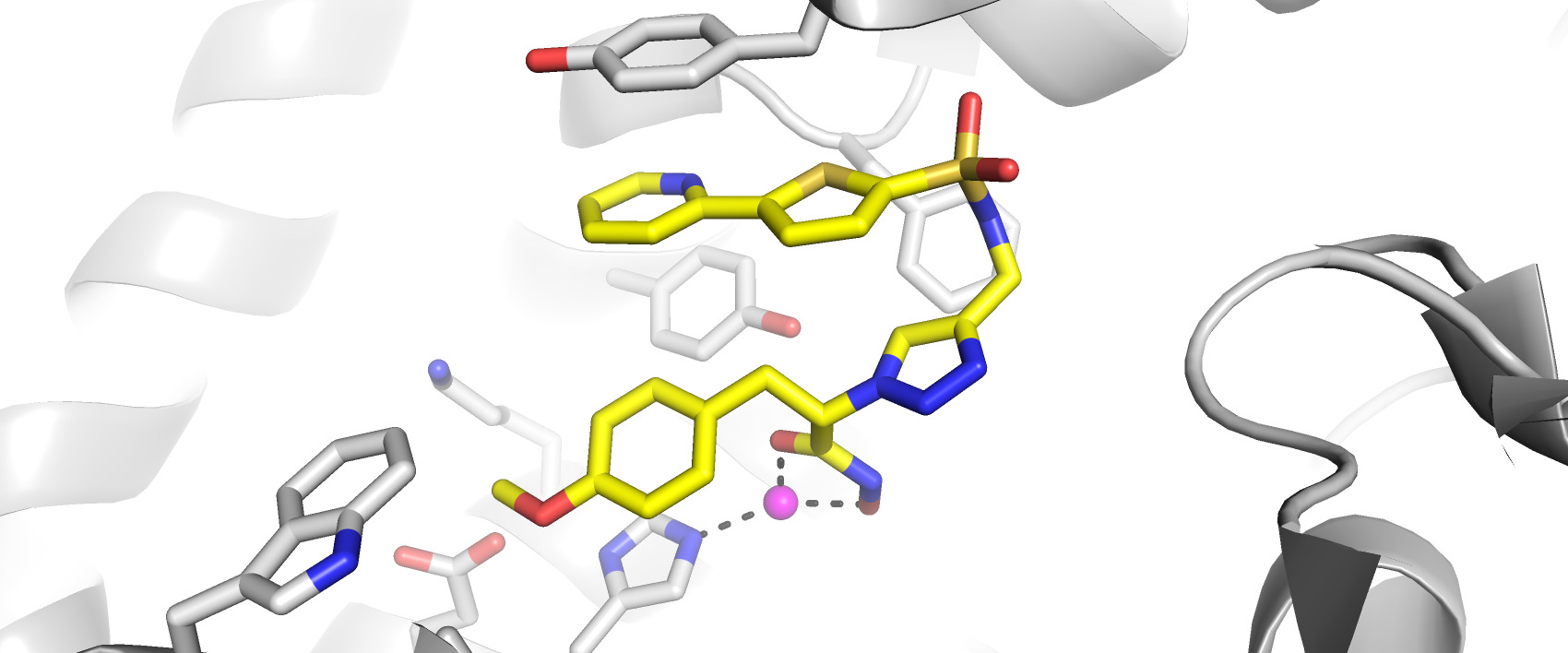
New enzyme inhibitor shows promise for treating cancers, autoimmune diseases
Researchers have found a small molecule enzyme inhibitor capable of manipulating an immune process that plays an important role in cancers and autoimmune diseases.
Combination immunotherapy improves survival for patients with asymptomatic melanoma brain metastases
Combination treatment with immune checkpoint inhibitors nivolumab and ipilimumab demonstrates overall survival for patients with melanoma that has spread to the brain, according to Phase II study results published today in The Lancet Oncology by researchers from The University of Texas MD Anderson Cancer Center.
Studies Find B7-H3 Protein a Novel, Promising Target for Prostate Cancer Treatments
The immune checkpoint protein B7-H3 may be a promising new target for immunotherapy in treatment-resistant prostate cancers, according to two new studies led by researchers at the Johns Hopkins Kimmel Cancer Center. The studies were presented recently at the European Society for Medical Oncology (ESMO) 2021 Conference.
Immunotherapy may be effective for subset of prostate cancer
Researchers report that about a quarter of localized prostate cancers may demonstrate immunologic traits that suggest a substantial number of patients with prostate cancer may benefit from immunotherapies.
Roswell Park, Wilmot Cancer Institute Collaborate on First Large Study of Immunotherapy in Black Cancer Patients
A new collaboration between two Western New York cancer research leaders will help oncologists learn whether Black and white cancer patients respond differently to a game-changing immunotherapy treatment, and seeks to improve the safety and effectiveness of these newer drugs in diverse populations.
Atlantic Health System Physicians Co-Author 5 Studies, Presented at American Association for Cancer Research (AACR) Annual Meeting
Atlantic Health System Cancer Care physicians are co-authors of five original studies presented at this year’s AACR Annual Meeting, held virtually April 10-15 and May 17-21. The AACR meeting is one of the world’s premier scientific gatherings of cancer specialists and researchers.
A clue to how some fast-growing tumors hide in plain sight
Viruses churn out genetic material in parts of the cell where it’s not supposed to be. Cancer cells do too. A new study shows that a tumor-suppressor enzyme called DAPK3 is an essential component of a multi-protein system that senses misplaced genetic material in tumor cells, and slows tumor growth by activating the fierce-sounding STING pathway.
Ludwig Cancer Research Study Uncovers Metabolic Vulnerability in Suppressive Immune Cells That May Be Targeted for Cancer Immunotherapy
A Ludwig Cancer Research study has identified a novel mechanism by which a type of cancer immunotherapy known as CTLA-4 blockade can disable suppressive immune cells to aid the destruction of certain tumors.

New immunotherapy target discovered for malignant brain tumors
Scientists say they have discovered a potential new target for immunotherapy of malignant brain tumors, which so far have resisted the ground-breaking cancer treatment based on harnessing the body’s immune system. The discovery, reported in the journal CELL, emerged from laboratory experiments and has no immediate implications for treating patients.
CRISPR-edited CAR T Cells Enhance Fight Against Blood Cancers
Knocking out a protein known to stifle T cell activation on CAR T cells using the CRISPR/Cas9 technology enhanced the engineered T cells’ ability to eliminate blood cancers.
Activating “Inflammasome” May Improve Cancer’s Response To Immunotherapy And Parp Inhibitors
The inflammasome—a protein signaling network that is activated to rid the body of virus or bacteria-infected cells—may play an important role in triggering an immune response to cancer and causing an existing class of drugs to work better against cancers.
Houston Methodist using 3D technology, artificial intelligence and more in new breast cancer research studies
Trials include a model to create custom breast implants, a smarter method to recommend biopsy, a novel approach to preserve sensation in implant-based breast reconstruction, and a new clinical trial investigating a modified herpes virus as a tactic to trigger immune response.
Effective Cancer Immunotherapy Further Linked To Regulating A Cell ‘Suicide’ Gene
Johns Hopkins Medicine researchers have added to evidence that a gene responsible for turning off a cell’s natural “suicide” signals may also be the culprit in making breast cancer and melanoma cells resistant to therapies that use the immune system to fight cancer. A summary of the research, conducted with mice and human cells, appeared Aug. 25 in Cell Reports.
Three Women Scientists at Johns Hopkins Tapped to Join Exclusive Research Network
Three Johns Hopkins Kimmel Cancer Center scientists are among the first 45 members selected to join the 10x Genomics Visium Clinical Translational Research Network (CTRN), aimed at advancing translational research in some of the world’s leading health problems, including oncology, immuno-oncology, neuroscience, infectious disease, inflammation and fibrosis, and COVID-19.

Combination biomarker predicts response to immune checkpoint therapy in patients with advanced bladder cancer
A combination of two biomarkers was predictive of improved clinical responses and prolonged survival following treatment with immune checkpoint inhibitors in patients with advanced bladder cancers.

Immunotherapy effective in metastatic prostate cancers with specific markers of immune activation
A subset of patients with metastatic prostate cancer and specific markers of immune activity responded well to treatment with immune checkpoint inhibitors, according to results of a Phase II trial.

Roswell Park’s Dr. Pawel Kalinski to Lead $14.5M NCI-Funded Immunotherapy Effort
A team led by Pawel Kalinski, MD, PhD, of Roswell Park Comprehensive Cancer Center has earned a five-year, $14.54 million award from the National Cancer Institute (NCI) to expand a promising immunotherapy platform. Funded through the NCI’s Program Project Grant program, this prestigious five-year grant will fund five clinical trials, all focused on a strategy for making some of the most common immunotherapies work for more cancer patients.
Immunotherapy using ‘young cells’ offers promising option against cancer
A new study from Washington University School of Medicine in St. Louis suggests that the age of certain immune cells used in immunotherapy plays a role in how effective it is. These cells — natural killer (NK) cells — appear to be more effective the earlier they are in development, opening the door to the possibility of an immunotherapy that would not utilize cells from the patient or a matched donor. Instead, they could be developed from existing supplies of what are called human pluripotent stem cells.

Pembrolizumab shows promise for some advanced, hard-to-treat rare cancers
Pembrolizumab shows promise for some advanced, hard-to-treat rare cancers. Open-label Phase II study at MD Anderson reports on four cancer types.

TARGETING TURNCOAT IMMUNE CELLS TO TREAT CANCER
A Ludwig Cancer Research study has identified a mechanism by which regulatory T cells, which suppress immune responses, adapt their metabolism to thrive in the harsh microenvironment of the tumor.
Tip Sheet: Tracking coronavirus, improving immunotherapies, cancer death rates decline, AAAS meeting and more
Summaries of recent Fred Hutch research studies, plus information on a press event at the upcoming AAAS annual meeting in Seattle.
Building a Safer CAR-T Therapy
A Ludwig Cancer Research study has devised a new type of chimeric antigen-receptor (CAR) T cell—a family of promising immunotherapies for cancer—that can be switched on and off on demand.
GW Cancer Center Expands Clinical Trial Offerings for Patients with High Risk Cutaneous Squamous Cell Carcinoma
The Cutaneous Oncology Program at the GW Cancer Center was selected as the first global site for a clinical trial for patients with high-risk cutaneous squamous cell carcinoma. The study, sponsored by Regeneron, will examine outcomes for patients treated with Libtayo® (cemiplimab) — an immunotherapy treatment — prior to surgery and radiation therapy.

John Theurer Cancer Center Participating in Early-Phase Study of Immunotherapy-Boosting Treatment
Investigators at John Theurer Cancer Center at Hackensack University Medical Center in New Jersey are participating in a first-in-patients clinical trial assessing VE800, a novel bacteria-containing therapy, in combination with the immunotherapy drug nivolumab. Laboratory research has suggested that VE800 may enhance the effectiveness of drugs like nivolumab.
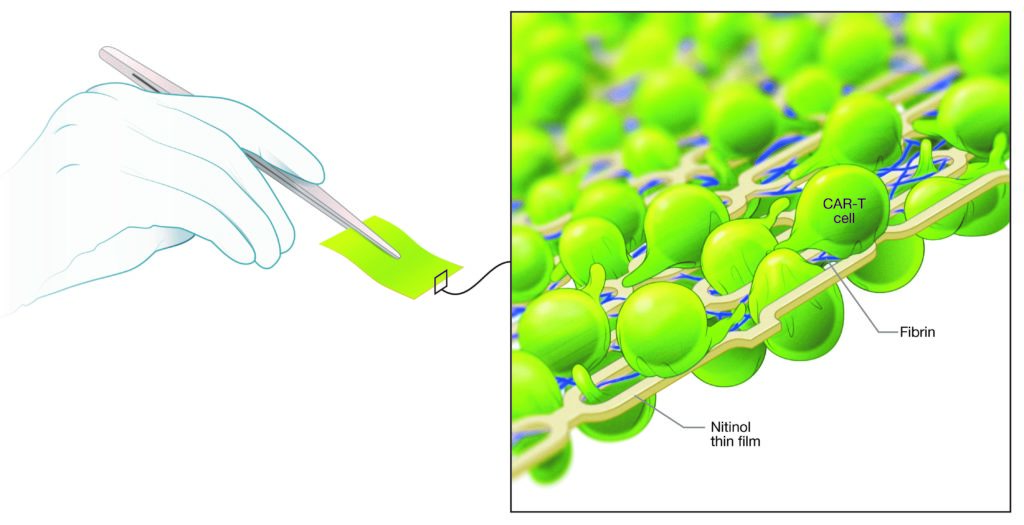
Scientists show thin metal mesh loaded with T cells shrinks solid tumors
Within weeks, CAR T cells targeting ovarian cancer cleared tumors in 70% of treated mice, shows study in Nature Biomedical Engineering. (Note: Images available for media use).
Study Examines Kidney Injury in Patients Taking Immunotherapy Cancer Medications
In patients taking immune checkpoint inhibitors as a treatment for cancer, 17% experienced acute kidney injury (AKI), 8% experienced sustained AKI, and 3% had potential immune checkpoint inhibitor–related AKI.
• Use of proton pump inhibitors, which are commonly used to treat stomach ulcers or acid reflux, was associated with a higher risk of experiencing sustained AKI.

NUS scientist designs ‘express courier service’ for immune cells
Dr Andy Tay, a researcher with the National University of Singapore (NUS) who is currently doing his post-doctoral training at Stanford University, has successfully invented a novel transfection method to deliver DNA into immune cells with minimal stress on these cells. This new technique is expected to boost DNA-based cancer immunotherapy by significantly improving the process of generating high-quality genetically modified immune cells.