These clinical trial results point to the first safe and effective use of immunotherapy for patients with microsatellite stable colorectal cancer, the most common subtype of the disease. The encouraging results of this phase I trial have prompted Dana-Farber investigators to move forward with a phase III trial of this immunotherapy combination.
Tag: cancer immunotherapy
UC San Diego Health First in Region to Provide Novel Therapy for Melanoma
Breakthrough one-time cancer treatment option for select patients with metastatic melanoma and solid tumors now offered at UC San Diego Health.
Altering cancer treatment dosing could reduce climate impact, study finds
Changing how often a popular cancer therapy is delivered would reduce greenhouse gas emissions and improve environmental impact without decreasing cancer survival, according to a new analysis from researchers at the University of Michigan Health Rogel Cancer Center.
Gut bacteria boost immune response to fight tumors
Researchers at Washington University School of Medicine in St. Louis have found that a strain of gut bacteria can boost immune responses and enhance cancer immunotherapy to fight sarcoma tumors in mice. Shown is a rendering of microbes in the intestine.
A gene mutation associated with a rare neurological disorder and increased susceptibility to viral infections may be treatable with oleic acid
A mutation in a protein regulating natural killer (NK) cells’ function is at the root of immune deficiency in some people with a rare genetic condition characterized by cognitive and developmental delay, seizures, and other manifestations. The findings also have broader implications for immunity and cell therapies.
Reimagining Cancer Team Science: Six Hallmarks of Cancer Collaboration
The oncology community has a powerful opportunity to speed the creation of impactful new treatments by adopting a modern playbook for collaboration, according to scientists from Break Through Cancer and other team-oriented cancer organizations working together in a commentary in the journal Cancer Discovery
Nanoparticle vaccine could curb cancer metastasis to lungs by targeting a protein
UC San Diego engineers have developed an experimental vaccine that could prevent the spread of metastatic cancers to the lungs. Its success lies in targeting a protein known to play a central role in cancer growth and spread, rather than targeting the primary tumor itself.
Drug-filled nanocapsule helps make immunotherapy more effective in mice
UCLA researchers have developed a new treatment method using a tiny nanocapsule to help boost the immune response, making it easier for the immune system to fight and kill solid tumors.
Thailand Hub of Talent for Cancer Immunotherapy International Conference
The Faculty of Medicine, Chulalongkorn University, cordially invites all to attend the “Thailand Hub of Talent for Cancer Immunotherapy International Conference: Portal to Global Collaboration for Next Generation Cell and Gene Therapy Development” on November 16-17, 2023 at Bhumisiri Mangklanusorn Building, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand.
Scientists identify new pathway activated by interferon-gamma that leads to tumor cell death
Researchers at the UCLA Jonsson Comprehensive Cancer Center have identified a new role for a protein called extracellular signal-regulated kinase (ERK) in a pathway activated by interferon-gamma that can trigger cells to self-destruct.
LJI scientists harness ‘helper’ T cells to treat tumors
La Jolla Institute for Immunology (LJI) scientists have published a pair of studies that show how we might harness CD4+ T cells while boosting the cancer-fighting power of CD8+ “killer” T cells.
New Computational Tool Identifies Novel Targets for Cancer Immunotherapy
Researchers at Children’s Hospital of Philadelphia (CHOP) and the University of California, Los Angeles (UCLA) have developed a computational platform capable of discovering tumor antigens derived from alternative RNA splicing, expanding the pool of cancer immunotherapy targets. The tool, called “Isoform peptides from RNA splicing for Immunotherapy target Screening” (IRIS), was described in a paper published today in the Proceedings of the National Academy of Sciences.
Immunotherapy plus chemotherapy combination for advanced lung cancer not only prolongs life but also improves its quality
A recent clinical trial showed that the drug combination of cemiplimab plus platinum chemotherapy can prolong survival in patients with advanced lung cancer when compared with placebo plus platinum chemotherapy. Now an analysis published by Wiley online in CANCER, a peer-reviewed journal of the American Cancer Society, indicates that cemiplimab plus platinum chemotherapy also affects quality of life compared to chemotherapy alone.
Gamma delta T cells can fight aggressive breast cancer
Researchers from the University of Frieburg discovered that coordinated differentiation and changes in the metabolism of breast cancer stem cells make them invisible for the immune system.
Reviving exhausted T cells to tackle immunotherapy-resistant cancers
One of the biggest goals of immunotherapy is to reverse T cell exhaustion to boost the immune system’s ability to destroy cancerous cells. Researchers at Sanford Burnham Prebys studying melanoma have found a new way to make this happen.
Nobel Prize-winning immuno-oncology expert receives Block Memorial Lectureship
James P. Allison, PhD, is the recipient of the 25th Herbert and Maxine Block Memorial Lectureship Award for Distinguished Achievement in Cancer. A 2018 Nobel Prize co-recipient in physiology/medicine, Allison serves as the chair of immunology and executive director of the Immunotherapy Platform at The University of Texas MD Anderson Cancer Center.
Chula Makes Progress in “CAR T-Cell Therapy” Innovation: New Hope for Thai Lymphoma Cancer Patients
Chulalongkorn University, Thailand, and Nagoya University, Japan, in their collaboration to develop an immunotherapy method for curing cancer, reported on the progress of CAR T-cell immunotherapy innovation for treating cancer in leukemia and B-cell lymphoma patients, which can increase survival rates and reduce cancer recurrence.
Cancer patients who don’t respond to immunotherapy lack crucial immune cells
Cancer immunotherapy involves using activated T cells to destroy tumors, but it doesn’t work for all patients. Researchers at Washington University School of Medicine in St. Louis have discovered that a kind of dendritic cell is crucial for determining the effectiveness of immunotherapy. The discovery could lead to new ways to extend the benefits of immunotherapy to more patients.
New immunotherapy holds promise for ovarian cancer
CAR T-cell therapy, a certain kind of cancer treatment in which the immune system’s T cells are programmed to attack tumour cells, is effective in mice with ovarian cancer, according to a study published in The Journal for ImmunoTherapy of Cancer.
Drug triggers immune cells to attack prostate cancer
A single drug compound simultaneously attacks hard-to-treat prostate cancer on several fronts, according to a new study in mice and human cells.
Focused ultrasound treatment for essential tremor highly effective after 5 years
A scalpel-free, high-tech form of brain surgery pioneered at UVA Health offers long-term relief for patients with essential tremor, a common movement disorder, a five-year review shows.
Cleveland Clinic Receives $12 Million NIH Grant to Investigate Use of Inflammatory Cytokines in Personalized Cancer Treatments
Cleveland Clinic’s Lerner Research Institute was awarded a five-year $12 million grant by the National Cancer Institute, part of the National Institutes of Health, to define how cytokines – proteins produced during immune response – regulate inflammation and interact with cells and molecules surrounding tumors.
Cleveland Clinic Receives $7.9 Million Grant from National Institutes of Health to Form Radiation Oncology-Biology Integration Network (ROBIN)
Cleveland Clinic received a $7.9 million five-year grant from the National Cancer Institute at the National Institutes of Health to form one of three national centers as part of the newly established Radiation Oncology-Biology Integration Network (ROBIN).
Phase II clinical trial suggests treatment pre-surgery safe and effective option for localized non-small cell lung cancer
New study data shows that the immuno-oncology drug, atezolizumab is a safe and effective treatment for stage IB-IIIB non-small cell lung cancer patients prior to lung cancer surgery, according to a new study led by researchers with The Ohio State University Comprehensive Cancer Center – Arthur G. James Cancer Hospital and Richard J. Solove Research Institute as part of the national Lung Cancer Mutation Consortium 3 study.
Researchers found that the survival of patients in this new study was far better – about 80% at three years post treatment and approximately twice as good as that which would be expected with surgery and chemotherapy alone. Study investigators also report data showing that the presence of high numbers of natural killer (NK) cells – a type of immune cell found in the blood before treatment – were associated with poor immunotherapy treatment effectiveness in this study. Patients with high levels of these cells might benefit from the addition of NK-specific therapy.
Cancer Research Institute Names Newest Class of Lloyd J. Old STARs, Today’s Visionaries in Cancer Immunotherapy Research
Six U.S. scientists have been awarded $1.25 million each over 5 years to conduct high-risk, high-reward cancer immunology research with the potential to transform cancer treatment
Plant virus plus immune cell-activating antibody clear colon cancer in mice, prevent recurrence
A new combination therapy to combat cancer could one day consist of a plant virus and an antibody that activates the immune system’s “natural killer” cells, shows a study by researchers at the University of California San Diego. In mouse models of colon cancer, the combination therapy eliminated all tumors and prevented their recurrence, which in turn resulted in 100% survival. The therapy also increased survival in mouse models of melanoma.
UTSW researchers develop microbubble technology to enhance cancer immunotherapy
Researchers at UT Southwestern have developed a first-of-its-kind ultrasound-guided cancer immunotherapy platform that delivers immune-stimulating agents to cells for the development of systemic anti-tumor immunity. The technology, termed Microbubble-assisted Ultrasound-guided Immunotherapy of Cancer (MUSIC), was described in a study published in Nature Nanotechnology.
Studies reveal new insights into gut microbiome impact on immunotherapy response in multiple cancers, including glioblastoma
Two studies led by The University of Texas MD Anderson Cancer Center that shed new light on the potential of the gut microbiome as a targetable biomarker to improve responses to immunotherapy were presented today at the 2022 American Society for Clinical Oncology (ASCO) Annual Meeting.
Researchers find new mechanism to turn on cancer-killing T cells
In a new study, researchers at the University of Chicago Medicine Comprehensive Cancer Center and the University of Amsterdam have brought insight into one crucial step in the anti-cancer immune response process: T cell priming.
Model predicts early response to cancer immunotherapy
Researchers have developed a model that could predict early on in treatment whether cancer patients will respond to immunotherapy, according to a report published today in eLife.
CHOP Researchers Develop a New Class of CAR-T Cells that Target Previously Untargetable Cancer Drivers
In a breakthrough for the treatment of aggressive solid cancers, researchers at Children’s Hospital of Philadelphia (CHOP) have developed a novel cancer therapy that targets proteins inside cancer cells that are essential for tumor growth and survival but have been historically impossible to reach. Using the power of large data sets and advanced computational approaches, the researchers were able to identify peptides that are presented on the surface of tumor cells and can be targeted with “peptide-centric” chimeric antigen receptors (PC-CARs), a new class of engineered T cells, stimulating an immune response that eradicates tumors.
How a plant virus could protect and save your lungs from metastatic cancer
Using a virus that grows in black-eyed pea plants, researchers developed a new therapy that could keep metastatic cancers from spreading to the lungs, as well as treat established tumors in the lungs.
Ultrasound remotely triggers immune cells to attack tumors in mice without toxic side effects
A new cancer immunotherapy pairs ultrasound with specially engineered CAR T cells to destroy malignant tumors while sparing normal tissue. The new experimental therapy significantly slowed down the growth of solid cancerous tumors in mice.
Cell-based immunotherapy shows promise against melanoma
Researchers at Washington University School of Medicine in St. Louis have shown in preclinical studies conducted in mice and human cells that a type immunotherapy based on natural killer cells could be effective against solid tumors, starting with melanoma, a type of skin cancer that can be deadly if not caught early.
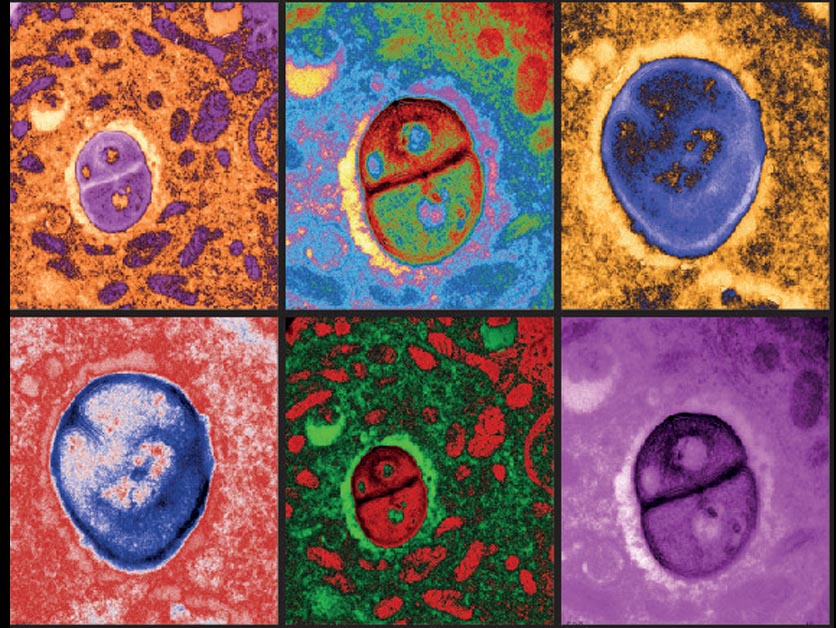
Weizmann Scientists Find That Bacteria May Aid Anti-Cancer Immune Response
The Weizmann Institute’s Prof. Yardena Samuels, Prof. Eran Segal, and Dr. Ravid Straussman, with partners at MD Anderson Cancer Center, the NCI, and elsewhere, have discovered that the bacteria living inside cancer cells can be harnessed to provoke an immune reaction against the tumor. The work could also help explain findings showing that the microbiome affects the success of immunotherapy.
Cancer Experts Call for Reform of Clinical Trials, with an Emphasis on Independent Studies
A recently published paper addresses the threat posed by overstating the significance of inconclusive studies to the public by both the media and the scientific community itself.
Cancer Immunotherapy May Also Treat Certain Autoimmune Diseases
The new approach blocks the interaction between cancer cells and immune receptors, showing promise in mice.
Mutant Gene-Targeted Immunotherapy Approach Developed
Johns Hopkins Kimmel Cancer Center study co-author Bert Vogelstein, M.D., will present the related talk “Targeting genetic alterations in cancers with immunotherapeutic agents” at 11 a.m., March 1, at the Advances in Genome Biology and Technology (AGBT) conference. More information can be found at: https://www.agbt.org/events/general-meeting/agenda/. NOTE: AGBT provides complimentary press registration to staff and working freelance journalists who wish to cover the meeting. https://www.agbt.org/media/guidelines/
An mRNA vaccine for cancer immunotherapy
Researchers reporting in ACS’ Nano Letters have developed a hydrogel that, when injected into mice with melanoma, slowly released RNA nanovaccines that shrank tumors and kept them from metastasizing.
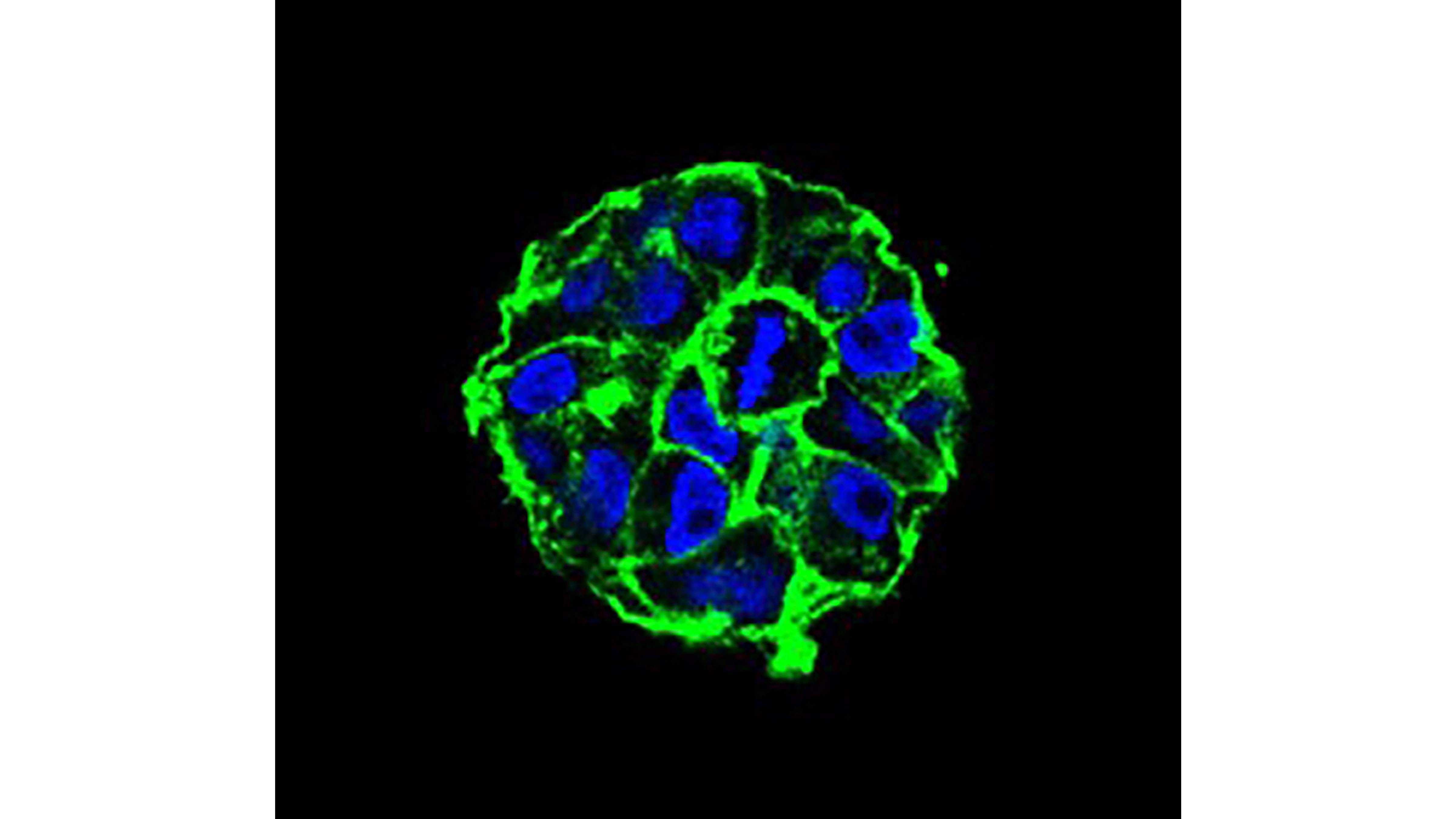
Cancer Research Expands Body’s Own Immune System to Kill Tumors
Scientists are hoping advances in cancer research could lead to a day when a patient’s own immune system could be used to fight and destroy a wide range of tumors. Cancer immunotherapy has some remarkable successes, but its effectiveness has been limited to a relatively small handful of cancers. In APL Bioengineering, researchers describe how advances in engineering models of tumors can greatly expand cancer immunotherapy’s effectiveness to a wider range of cancers.

Research Leader Named to Rutgers Cancer Institute from National Cancer Institute to Build World-Class Cancer Immunology and Immunotherapy Programs
Research Leader Christian Hinrichs, MD, has been named Chief of the Section of Cancer Immunotherapy and Co-Director of the Cancer Immunology and Metabolism Center of Excellence at Rutgers Cancer Institute of New Jersey,
Key Discoveries in Cancer Treatment to Be Presented During the Virtual 2020 AACC Annual Scientific Meeting
During the all-virtual 2020 AACC Annual Scientific Meeting & Clinical Lab Expo, leading scientific experts will dive into the groundbreaking advances that are the future of cancer care. One plenary session will focus on the cutting-edge treatment known as T cell therapy, while another will showcase recent discoveries about tumor metabolism that could lead to new, more effective cancer drugs.

EMBARGOED STUDY: Potential Cancer Therapy May Boost Immune Response
A new approach to cancer therapy shows potential to transform the commonly used chemotherapy drug gemcitabine into a drug that kills cancer cells in a specialized way, activating immune cells to fight the cancer, according to a study led by Cedars-Sinai Cancer investigators.

Rutgers Researcher Receives $400K Grant to Reduce Racial Health Disparities in Cancer Care
Anita Kinney, professor at the Rutgers School of Public Health and associate director for Population Science and Community Outreach at Rutgers Cancer Institute of New Jersey, is one of ten recipients across the nation of an American Cancer Society and Pfizer community grant of $399,892 to support investigation into reducing racial health disparities in cancer care through precision oncology and immunotherapy.

Cancer researchers identify checkpoint target for colorectal cancer immunotherapy
Researchers at the Indiana University Melvin and Bren Simon Comprehensive Cancer Center have identified a target for colorectal cancer immunotherapy.
Study fingers new player in cancer immunity
Study in mice reveals new immune regulatory mechanism involved in cancer, viral infections
Silencing immune-regulating gene in immune cells eradicated colon cancer in mice
Approach also helped some animals clear an aggressive form of melanoma
Findings offer pathway for design of new class of immune-based cancer therapies

Stephan Grupp, MD, PhD, Elected to the National Academy of Medicine
Stephan Grupp, MD, PhD, Chief of the Cell Therapy and Transplant Section in the Division of Oncology and Director of the Cancer Immunotherapy Program at Children’s Hospital of Philadelphia, has been elected into the National Academy of Medicine (NAM), effective Oct. 1, 2019.
Drug-light combo could offer control over CAR T-cell therapy
UC San Diego bioengineers are a step closer to making CAR T-cell therapy safer, more precise and easy to control. They developed a system that allows them to select where and when CAR T cells get turned on so that they destroy cancer cells without harming normal cells. The system requires two “keys”—the drug Tamoxifen and blue light—to activate CAR T cells to bind to their targets. Just one key keeps the cells inactive.