In recognition of their significant achievements in the realm of cancer care and research, four researchers at The University of Texas MD Anderson Cancer Center have been elected fellows of the American Association for the Advancement of Science (AAAS). This prestigious distinction stands as one of the highest accolades within the scientific research community.
Tag: Lymphoma
Scientists find core regulatory circuit controlling identity of aggressive leukemia
Scientists at St. Jude Children’s Research Hospital and Dana-Farber Cancer Institute found a small set of proteins that maintain anaplastic large cell lymphoma (ALCL) identity, representing potential future therapeutic targets.
CSI Singapore researchers uncover potential novel therapeutic targets against natural killer/T-cell lymphoma
A team of researchers from the Cancer Science Institute of Singapore (CSI Singapore) at the National University of Singapore (NUS) has discovered that a transcription factor, TOX2, was aberrantly increased in patients with Natural killer/T-cell lymphoma (NKTL). The increased TOX2 level leads to the growth and spread of NKTL, as well as the overproduction of PRL-3 – an oncogenic phosphatase that is a known key player in the survival and metastasis of several other types of cancers. This breakthrough discovery presents a potential novel therapeutic target to treat NKTL.
Patients with IBD at higher risk of lymphoma
The risk of developing lymphoma is slightly elevated in inflammatory bowel disease (IBD) and has increased in recent years in patients with Crohn’s disease, report researchers from Karolinska Institutet in a study published in Clinical Gastroenterology and Hepatology.
Moffitt Researchers Discover Pathway Critical for Lymphoma Development
In a new article in Blood Cancer Discovery, which was published simultaneously with a presentation at the American Association for Cancer Research annual meeting 2023, a team of Moffitt Cancer Center researchers demonstrated a possible alternative approach. They showed that MYC activates a downstream pathway that chemically modifies a protein called eIF5A, and that inhibition of this modification process prevents lymphoma development and progression in mouse models.
Chula Makes Progress in “CAR T-Cell Therapy” Innovation: New Hope for Thai Lymphoma Cancer Patients
Chulalongkorn University, Thailand, and Nagoya University, Japan, in their collaboration to develop an immunotherapy method for curing cancer, reported on the progress of CAR T-cell immunotherapy innovation for treating cancer in leukemia and B-cell lymphoma patients, which can increase survival rates and reduce cancer recurrence.
MD Anderson’s Christopher Flowers, M.D., receives ASH Mentor Award
Christopher Flowers, M.D., division head ad interim of Cancer Medicine and chair of Lymphoma & Myeloma at The University of Texas MD Anderson Cancer Center, has received a 2022 ASH Mentor Award from the American Society of Hematology in acknowledgement of his career-long commitment to mentoring.

AI and Cancer: Study Highlights Automated System to Calculate Metabolic Tumor Volume
AI-based approach could make it easier to incorporate metabolic tumor volume into clinical trials and possibly patient care
NCCN Annual Congress on Hematologic Malignancies™ Returns to In-Person October 14–15, 2022 in New York City
The 2022 NCCN Annual Congress on Hematologic Malignancies™ (#NCCNhem22) is taking place Friday, October 14–Saturday, October 15, 2022 in New York City. All live sessions will also stream remotely through a virtual event platform.
Blood cancer scientists developing powerful new tools to improve diagnosis and treatment for patients
Australian researchers will use new technology to improve the diagnosis and treatment of blood cancers, which affect 1.24 million people globally, including 720,000 who die from leukemia, lymphoma or myeloma each year.
KU Cancer Center researchers discover that people with blood-related cancers have a higher chance of COVID breakthrough infections
Individuals with blood-related cancers are more likely to experience a COVID-19 infection even after being vaccinated, a University of Kansas Cancer Center study has found.
From ASCO 2022: Brentuximab Vedotin and Chemotherapy an Effective Treatment for Hodgkin Lymphoma
New research led by Kara Kelly, MD, of Roswell Park Comprehensive Cancer Center and presented today at the American Society of Clinical Oncology (ASCO) 2022 Annual Meeting in Chicago shows that a combination of brentuximab vedotin (Bv) and standard chemotherapy is safe and more effective than standard chemotherapy in pediatric patients up to age 21 years with newly diagnosed high-risk Hodgkin lymphoma. The findings from a phase 3 National Cancer Institute-supported multicenter Children’s Oncology Group clinical trial (NCT 02166463) were presented by first author Sharon Castellino, MD, of Emory University and Children’s Healthcare of Atlanta in an oral abstract session on Friday, June 3.
MD Anderson researchers present cellular therapy advances at the 2022 ASCO Annual Meeting
Promising clinical results with cellular therapies for patients with blood cancers highlight advances being presented by researchers from The University of Texas MD Anderson Cancer Center at the 2022 American Society of Clinical Oncology (ASCO) Annual Meeting.
These findings include long-term outcomes of patients receiving an infusion of brexucabtagene autoleucel (KTE-X19) for mantle cell lymphoma, efficacy of gamma delta CAR T therapy for aggressive B-cell lymphoma and responses of umbilical cord blood-derived expanded natural killer cells when given together with combination therapy before stem cell transplant.
Penn Medicine Developed CAR T Cell Therapy Wins Third FDA Approval
The U.S. Food and Drug Administration (FDA) has expanded its approval for a personalized cellular therapy developed at the University of Pennsylvania’s Abramson Cancer Center, this time for the treatment of adults with relapsed or refractory follicular lymphoma (FL) after two or more lines of systemic therapy. The accelerated approval was granted today to Novartis for the chimeric antigen receptor (CAR) T-cell therapy Kymriah® (tisagenlecleucel), making it the third indication for the nation’s first personalized cellular therapy for cancer. It remains the only CAR-T cell therapy approved for both adult and pediatric patients.
NK cells combined with bispecific antibody showed strong response for patients with lymphoma
Researchers from The University of Texas MD Anderson Cancer Center showed that natural killer (NK) cells derived from donated umbilical cord blood, combined with a novel bispecific antibody known as AFM13 that targets CD16A and CD30, achieved effective responses in patients with pretreated and refractory CD30+ lymphoma. The study was presented today at the American Association for Cancer Research (AACR) Annual Meeting 2022.
Blood Cancer Cures and Care: Addressing Leukemia and Lymphoma
Kevin David, MD the director of the lymphoma program and a hematologist/oncologist at Rutgers Cancer Institute of New Jersey shares information about leukemias and lymphomas for blood cancer awareness month.
Rare Case of Lactic Acidosis Associated with Small Lymphocytic Lymphoma (SLL) in Elderly Man Published in Journal of Cancer Research and Oncobiology
A team of researchers from Hackensack Meridian Jersey Shore University Medical Center and the Cleveland Clinic, led by Professor of Medicine and Nephrologist Sushil K. Mehandru, M.D., authored a new report.
Antiviral T cells safe and effective for treating debilitating complication common after stem cell transplants
A Phase II trial led by researchers at The University of Texas MD Anderson Cancer Center found that BK virus (BKV)-specific T cells from healthy donors were safe and effective as an off-the-shelf therapy for BKV-associated hemorrhagic cystitis (BKV-HC), a painful complication common after allogeneic stem cell transplants for patients with leukemia or lymphoma. The study was published today in the Journal of Clinical Oncology.

John Theurer Cancer Center Investigators Celebrate Newly Approved Lymphoma Therapies Evaluated in Clinical Trials at Hackensack Meridian Hackensack University Medical Center
New Treatments Expand Options for Patients with Persistent Cancers
Five Years Later: Penn-developed CAR T Therapy Shows Long-lasting Remissions in Non-Hodgkin Lymphomas
A significant number of non-Hodgkin lymphoma (NHL) patients in a Penn Medicine-initiated clinical trial continue to be in remission five years after receiving the chimeric antigen receptor (CAR) T cell therapy Kymriah™, researchers in Penn’s Abramson Cancer Center reported today in the New England Journal of Medicine.

Dr. Mohamad Cherry of Atlantic Health System Presents Studies at Top International Blood Cancer Meeting
Dr. Cherry focused on results from a Juno Therapeutics phase 2 study of liso-cel, an investigational CAR T therapy being tested against aggressive relapsed/refractory B cell non-Hodgkin lymphoma. Early results indicate a good safety profile, with few adverse events reported. Dr. Cherry also presented on the final results of a phase 1 study of targeted therapy gilteritinib in newly diagnosed acute myeloid leukemia (AML). Gilteritinib targets the most common mutation that causes AML, the FLT3 gene. Gilteritinib is sold under the brand name Xospata® by Astellas Pharma.

Circulating tumor DNA indicates increased risk of relapse after stem cell transplant in patients with diffuse large B-cell lymphoma
Many patients with diffuse large B-cell lymphoma (DLBCL) can be cured by a transplant using their own blood-forming stem cells, but as many as half eventually relapse. New research led by Dana-Farber Cancer Institute scientists suggests that patients whose blood or stem cell samples harbor tumor DNA are likely to relapse.

Analyzing Outcomes of Older Patients with Primary Central Nervous System Lymphoma
Older individuals are at an increased risk of developing primary central nervous system lymphoma (PCNSL). In a retrospective study of patients with newly diagnosed PCNSL, researchers at Rutgers Cancer Institute of New Jersey and colleagues used geriatric assessments to analyze detailed characteristics, treatment, and outcomes in patients across 17 academic centers.
CAR T cell therapy effective as first-line treatment for high-risk large B-cell lymphoma
A study led by researchers at The University of Texas MD Anderson Cancer Center found that axi-cel, an autologous anti-CD19 chimeric antigen receptor (CAR) T cell therapy, is a safe and effective first-line therapy for patients with high-risk large B-cell lymphoma (LBCL), a group with an urgent need for new and effective treatments.
Dana-Farber to present more than 40 research studies at 2020 ASH Annual Meeting
Dana-Farber Cancer Institute researchers will present more than 40 research studies at the virtual 62nd American Society of Hematology (ASH) Annual Meeting on December 5-8, including two studies that were selected for inclusion in the official press program.

Hematologist/Stem Cell Biologist to Direct Hematology and Cellular Therapy at Cedars-Sinai
Internationally recognized hematologist John P. Chute, MD, has been selected to direct the Division of Hematology and Cellular Therapy in the Department of Medicine at Cedars-Sinai Cancer. The physician-scientist also will serve as director of the Center for Myelodysplastic Diseases Research and associate director of the Board of Governors Regenerative Medicine Institute in the Department of Biomedical Sciences.
Sherry Adkins receives Brown Foundation Award for Excellence in Oncology Nursing
Sherry Adkins is the recipient of the 2020 Brown Foundation Award for Excellence in Oncology Nursing, MD Anderson’s highest nursing honor.
MD Anderson and Allogene Therapeutics announce strategic collaboration to accelerate advancement of allogeneic CAR T therapy (AlloCAR T)
MD Anderson and Allogene today announced a strategic five-year collaboration for preclinical and clinical investigation of allogeneic CAR T cell therapies.
Seattle Cancer Care Alliance Selected as an Authorized Treatment Center for TecartusTM, a Novel Immunotherapy Treatment
Seattle Cancer Care Alliance (SCCA), the only National Comprehensive Cancer Network (NCCN)-designated cancer center in Washington state, has been selected as an authorized treatment center to offer brexucabtagene autoleucel, also known by the brand name TecartusTM. It is the first and only chimeric antigen receptor (CAR) T-cell therapy approved by the U.S. Food and Drug Administration (FDA) for the treatment of adult patients with relapsed or refractory mantle cell lymphoma (MCL).
Study identifies characteristics of infused CAR T cells associated with efficacy and toxicity in patients with large B-cell lymphoma
Researchers at The University of Texas MD Anderson Cancer Center have identified molecular and cellular characteristics of anti-CD19 CAR T cell infusion products associated with how patients with large B-cell lymphoma (LBCL) respond to treatment and develop side effects.
NCCN Meeting on Blood Cancers Will Be Virtual for the First Time
The NCCN Virtual Nursing Forum and Annual Congress: Hematologic Malignancies (#NCCNhem2020) will provide the latest evidence and expert consensus on emerging practices and debates in blood cancer treatment, online October 8-10.
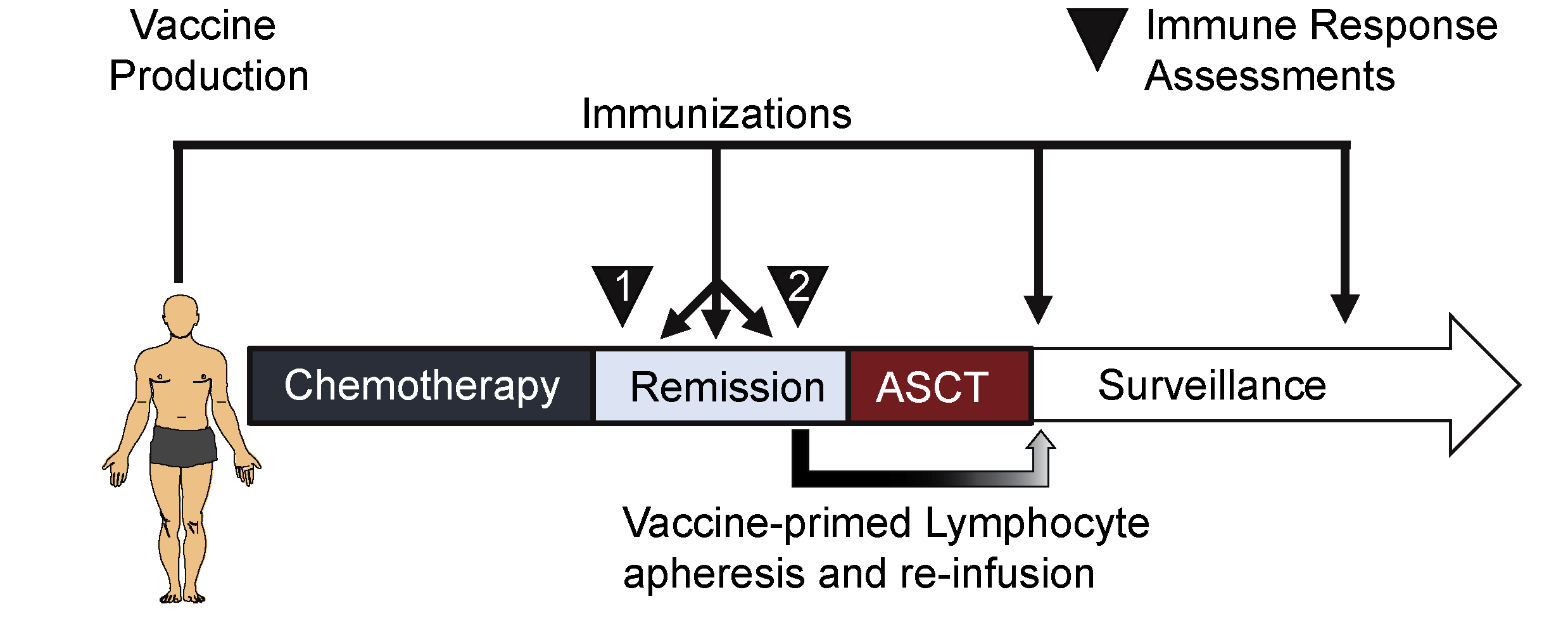
Early clinical trial supports tumor cell–based vaccine for mantle cell lymphoma
A phase I/II clinical trial by researchers at Stanford University suggests that vaccines prepared from a patient’s own tumor cells may prevent the incurable blood cancer mantle cell lymphoma (MCL) from returning after treatment. The study, which will be published June 19 in the Journal of Experimental Medicine (JEM), reveals that the vaccines are a safe and effective way to induce the body’s immune system to attack any tumor cells that could cause disease relapse.

Majority of patients responded in CAR T-cell trial for mantle cell lymphoma
Majority of patients responded in CAR T-cell trial for mantle cell lymphoma. Study showed 93% of patients responded with 67% achieving complete response.

Rutgers Cancer Institute of New Jersey Research Shows Autophagy Impacts Stress Response Pathways Promoting Survival in Laboratory Models
Research from investigators at Rutgers Cancer Institute of New Jersey shows that a cellular process known as autophagy promotes survival in mouse models by suppressing oxidative stress and a tumor suppressor known as p53.

CAR-T immunotherapy for lymphoma studied at UNC Lineberger receives fast-track designation from FDA
Based on proof-of-concept results from clinical trials at University of North Carolina Lineberger Comprehensive Cancer Center and Baylor College of Medicine, an investigational cellular immunotherapy for Hodgkin lymphoma has received a Regenerative Medicine Advanced Therapy designation from the U.S. Food and Drug Administration.

CD19 CAR NK-cell therapy achieves 73% response rate in patients with leukemia and lymphoma
According to results from a Phase I/IIa trial at The University of Texas MD Anderson Cancer Center, treatment with cord blood-derived chimeric antigen receptor (CAR) natural killer (NK)-cell therapy targeting CD19 resulted in clinical responses in a majority of patients with relapsed or refractory non-Hodgkin’s lymphoma (NHL) and chronic lymphocytic leukemia (CLL), with no major toxicities observed.

Study shows protein inhibitor as potential treatment approach for common mutations found in non-Hodgkin lymphomas
Study shows protein inhibitor as potential treatment approach for common mutations found in non-Hodgkin lymphomas. Findings may create immunotherapy options for lymphomas caused by CREBBP mutations.

CAR T-cell therapy effective for relapsed mantle cell lymphoma patients
A one-year follow-up study revealed a majority of patients with mantle cell lymphoma resistant to prior therapies may benefit from treatment with CD19-targeting chimeric antigen receptor (CAR) .

Treatment with PD-1 inhibitor prior to stem cell transplant is safe, effective for patients with classic Hodgkin lymphoma, study finds
A new analysis shows that a donor stem cell transplant following treatment with an immune checkpoint inhibitor is generally safe and produces good outcomes for patients with Hodgkin lymphoma.
Lymphoma Patients May Have New Path to Remission, Even When CAR T Therapy Fails
A new, experimental immunotherapy can put patients with B-cell non-Hodgkin lymphoma (NHL) that is resistant to or has come back after multiple other therapies, including CAR T therapy, into remission.

Retrospective Analysis Identifies Need for Better Classification of Rare Lymphoma
In what is believed to be one of the largest studies of a rare disorder known as primary cutaneous gamma delta T-cell lymphoma, Rutgers Cancer Institute
investigators and other collaborators examined characteristics, treatment patterns and outcomes and determined accurate diagnosis of the disease requires ongoing analysis.