Researchers describe a newly-observed role for the protein Cyclin Dependent Kinase 9 (CDK9) in regulating DNA repair during cellular division, where errors can become the origin of cancerous tumor growth.
Tag: Cancer Therapy
New treatment for a rare and aggressive cancer improves survival rates in breakthrough clinical trial
An innovative treatment significantly increases the survival of people with malignant mesothelioma, a rare but rapidly fatal type of cancer with few effective treatment options, according to results from a clinical trial led by Queen Mary University of London.
New technology makes cancer easier for immune system to find and destroy
A new technology to increase visibility of cancer cells to the immune system using CRISPR has been developed, and could lead to a new way to treat cancer.
Department of Energy Announces $16 Million for Particle Accelerators for Science & Society
Today, the U.S. Department of Energy (DOE) announced $16 million in funding for advanced research projects in particle accelerator science and technology.
NUS researchers develop novel approach for predicting resistance against cancer therapy
A team of researchers from the Cancer Science Institute of Singapore (CSI Singapore) at the National University of Singapore (NUS), led by Assistant Professor Anand Jeyasekharan, has discovered a unique combination of oncogenes that could predict treatment resistance, and hence unfavourable outcomes, of patients with Diffuse large B cell lymphoma (DLBCL), the most common type of blood cancer in Singapore and globally.
Chula Makes Progress in “CAR T-Cell Therapy” Innovation: New Hope for Thai Lymphoma Cancer Patients
Chulalongkorn University, Thailand, and Nagoya University, Japan, in their collaboration to develop an immunotherapy method for curing cancer, reported on the progress of CAR T-cell immunotherapy innovation for treating cancer in leukemia and B-cell lymphoma patients, which can increase survival rates and reduce cancer recurrence.
Study Shows Promising Safety, Patient Outcomes Data for MRI-Guided Adaptive Radiation Therapy to Treat Pancreatic Cancer
Findings from a recent prospective study show promising safety and patient outcomes data for locally advanced and borderline resectable pancreatic cancer treatment using ablative Stereotactic MRI-Guided On-table Adaptive Radiation Therapy, also known as SMART.
Department of Energy Announces $14.8 Million for Particle Accelerators for Science & Society
Today, the U.S. Department of Energy (DOE) announced $14.8 million in funding for advanced research projects in particle accelerator science and technology. Particle accelerators provide unique sources of light and particles that support the research of thousands of scientists worldwide, play a direct role in the production of more than $500 billion of goods annually, and treat more than 5 million cancer patients each year.
Prompt Recognition and Treatment Found Effective for Lung Disease in Patients Who Received New Drug for Advanced Cancer
Lung disease caused by a new drug for cancers—including metastatic or advanced breast cancer—can be effectively treated using approaches that focus on early detection and prompt management, according to a study published in ESMO Open on August 11, 2022.
Plant virus plus immune cell-activating antibody clear colon cancer in mice, prevent recurrence
A new combination therapy to combat cancer could one day consist of a plant virus and an antibody that activates the immune system’s “natural killer” cells, shows a study by researchers at the University of California San Diego. In mouse models of colon cancer, the combination therapy eliminated all tumors and prevented their recurrence, which in turn resulted in 100% survival. The therapy also increased survival in mouse models of melanoma.
Computer Simulations of Proteins Help Unravel Why Chemotherapy Resistance Occurs
Understanding why and how chemotherapy resistance occurs is a major step toward optimizing treatments for cancer. A team of scientists including Markus Seeliger, PhD, of the Renaissance School of Medicine at Stony Brook University, believe they have found a new process through which drug resistance happens.
Mount Sinai Researchers Discover Genes That Predict Good Response to Blood Cancer Therapy
Mount Sinai researchers have for the first time identified genes that predict a good response to a vital new therapy for a blood cancer that can have serious side effects for some patients.
Existing Cancer Therapy in Narrow Use Shows Significant Activity against Other Cancers
A drug currently used in just 1% of cancers has significant potential against the remaining 99%, according to a new study from UH Seidman Cancer Center and Case Western Reserve University, published in the prestigious journal Nature Cancer. Ivosidenib, or AG-120, is currently used against cancers that have a mutation in the IDH1 gene. However, study results show that Ivosidenib is also effective against unmutated, or “wild-type” IDH1. The protein coded by the IDH1 gene in cancers helps cancer cells survive in a stressful tumor environment, so any inhibitor medication that could weaken this defense mechanism is considered a promising therapy. UH and CWRU scientists discovered that under conditions present in the tumor microenvironment, drugs previously believed to be selective for the mutant enzyme have activity against the normal protein. Specifically, low glucose and magnesium levels enhance drug activity. The team has now tested Ivosidenib in mouse models of pancreatic, colorectal, o
Penn Medicine and Children’s Hospital of Philadelphia Announce Partnership with Costa Rica for CAR T Cell Therapy
Penn Medicine and Children’s Hospital of Philadelphia (CHOP), who together pioneered the research and development of the world’s first personalized cellular therapy for cancer — also known as CAR T cell therapy — have announced plans with Costa Rica’s CCSS, or the Caja Costarricense de Seguro Social (Social Security Program), to facilitate CAR T research in Costa Rica.
Capturing the Chemistry of Light-Activated Cancer Drugs with Ruomei Gao
Ruomei Gao—an associate professor at SUNY College at Old Westbury—has been using facilities at the Center for Functional Nanomaterials at Brookhaven Lab to investigate two primary processes of photosensitization for cancer therapy and prevention.
LUDWIG CANCER RESEARCH STUDY DISCOVERS HOW TO REVIVE POTENT BUT INERT ANTI-CANCER IMMUNE CELLS FOR THERAPY
A Ludwig Cancer Research study has discovered how to revive a powerful but functionally inert subset of anti-cancer immune cells that are often found within tumors for cancer therapy.
Led by Ludwig Lausanne’s Ping-Chih Ho and Li Tang of the École Polytechnique Fédérale de Lausanne, the study describes how an immune factor known as interleukin-10 orchestrates the functional revival of “terminally exhausted” tumor-infiltrating T lymphocytes (TILs), which have so far proved impervious to stimulation by immunotherapies. It also demonstrates that the factor, when applied in combination with cell therapies, can eliminate tumors in mouse models of melanoma and colon cancer. The findings are reported in the current issue of Nature Immunology.

Do You Know the Way to Berkelium, Californium?
Scientists at Berkeley Lab have demonstrated how to image samples of heavy elements as small as a single nanogram. The new approach will help scientists advance new technologies for medical imaging and cancer therapies.

Memorial Cancer Institute Together with FAU Research Partnership Earn ‘Cancer Center of Excellence’ Designation
A research partnership formed just last year by Memorial Healthcare System and Florida Atlantic University is already being recognized for quality care, results, and advances in research, and that’s great news for patients fighting cancer in South Florida. The alliance between Memorial’s Cancer Institute and FAU (MCIFAU) has been recognized by the state’s Department of Health as a “Florida Cancer Center of Excellence.” It becomes just the fifth in the state, out of more than 80 competitors, to earn the designation.
Scientists develop a cheaper method that might help create fuels from plants
Scientists have figured out a cheaper, more efficient way to conduct a chemical reaction at the heart of many biological processes, which may lead to better ways to create biofuels from plants.

Scientists Recruit New Atomic Heavyweights in Targeted Fight Against Cancer
Researchers from Berkeley Lab and Los Alamos National Laboratory have developed new methods for the large-scale production, purification, and use of the radioisotope cerium-134, which could serve as a PET imaging radiotracer for a highly targeted cancer treatment known as alpha-particle therapy.
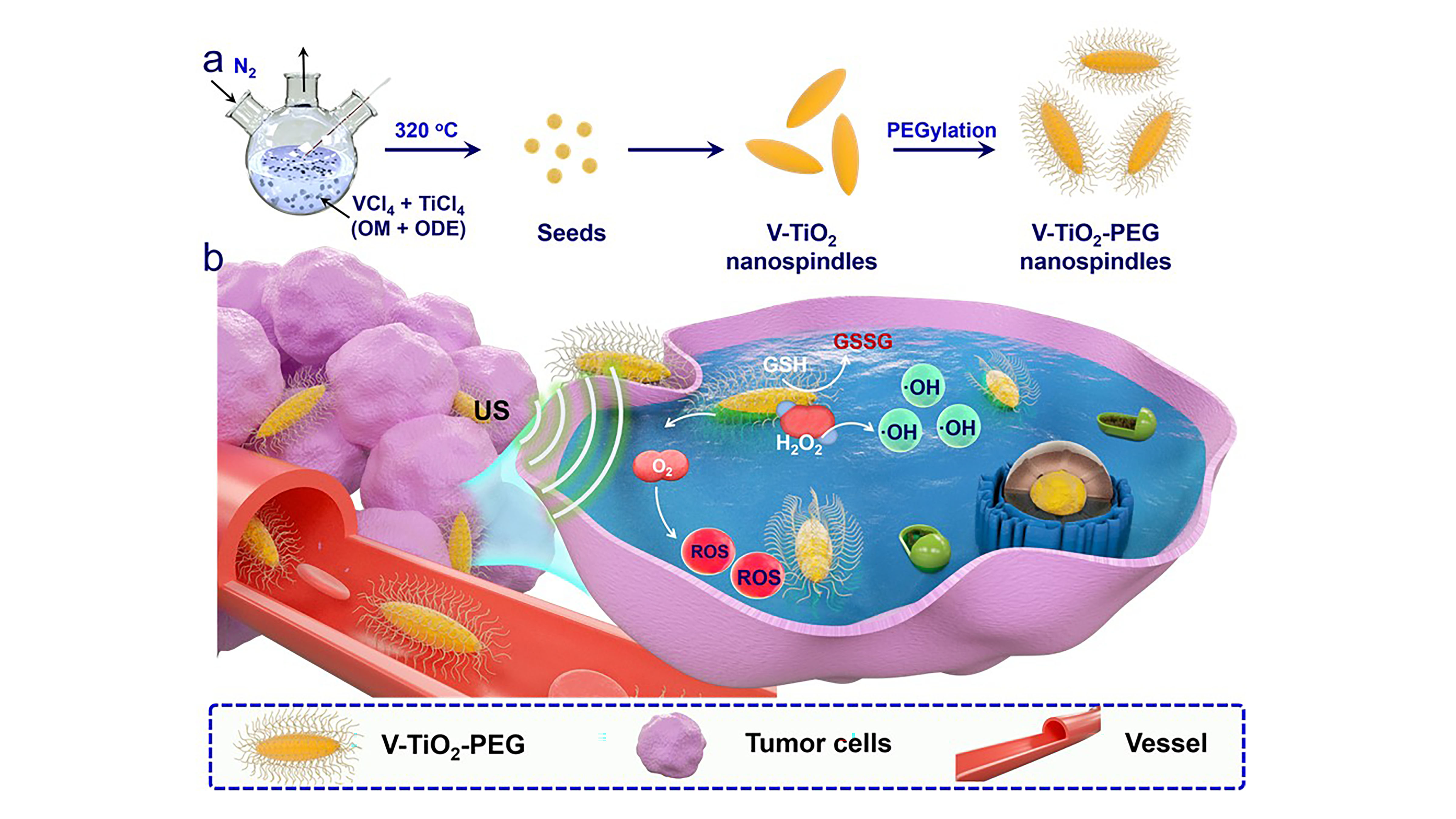
Tiny Nanospindles Enhance Use of Ultrasound to Fight Cancer
Ultrasound can be used to treat cancer when used in combination with molecules that sensitize the system to sound waves. These sonosensitizers generate toxic reactive oxygen species that attack and kill tumor cells. In Applied Physics Review, scientists report a new type of sonosensitizer based on a vanadium-doped titanium dioxide that enhances the amount of damage ultrasound inflicts on tumors. Studies in mice showed that tumor growth was markedly suppressed when compared to a control group.
Ludwig San Diego Study Identifies Deadly Genetic Synergy in Cancer Cells and Shows it Might Be Exploited for Therapy
A Ludwig Cancer Research study has identified a new instance in which the simultaneous mutation of two nonessential genes—neither of which is on its own vital to cell survival—can cause cancer cell death.

MSK: Cancer Isn’t Sheltering in Place
As the height of the COVID-19 outbreak in New York City and the Tri-State area begins to subside, Memorial Sloan Kettering Cancer Center oncologists are urging patients to schedule cancer screenings and treatments now – as the long-term toll of missed diagnoses and delayed treatments could be devastating for patients and their loved ones across the region and the country.
Cancer treatment with immune checkpoint inhibitors may lead to thyroid dysfunction
Thyroid dysfunction following cancer treatment with new treatments called immune checkpoint inhibitors is more common than previously thought, according to research that was accepted for presentation at ENDO 2020, the Endocrine Society’s annual meeting, and will be published in a special supplemental section of the Journal of the Endocrine Society.
Investigational drugs block bone loss in mice receiving chemotherapy
Studying mice, researchers from Washington University School of Medicine in St. Louis have found a driver of bone loss related to cancer treatment — cellular senescence. This process is independent of hormones related to bone health, such as estrogen. Such bone loss can be stopped by treating the mice with either of two investigational drugs already being evaluated in clinical trials.
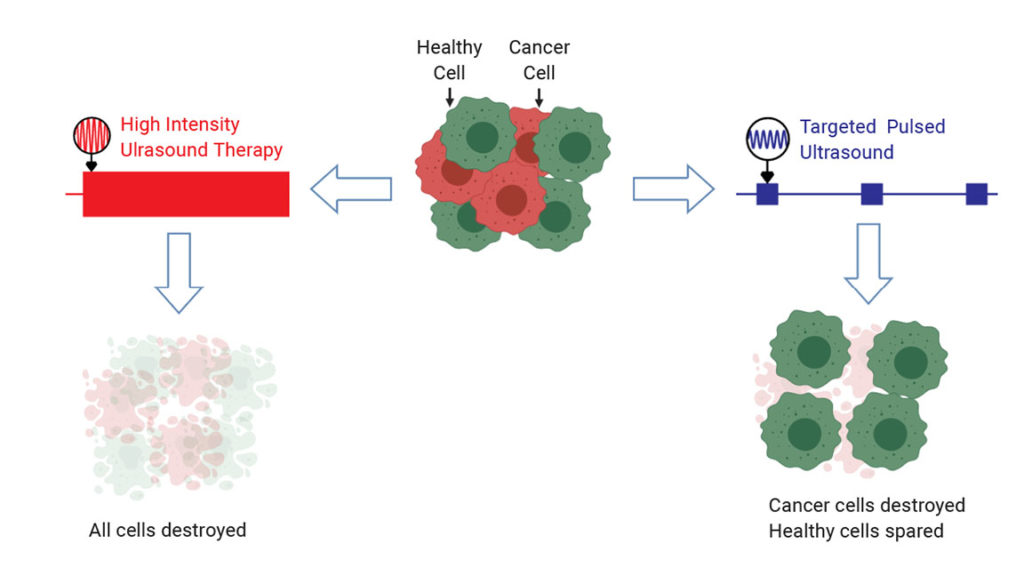
Ultrasound Selectively Damages Cancer Cells When Tuned to Correct Frequencies
Doctors have used focused ultrasound to destroy tumors without invasive surgery for some time. However, the therapeutic ultrasound used in clinics today indiscriminately damages cancer and healthy cells alike. Researchers have now developed a low-intensity ultrasound approach that exploits the properties of tumor cells to target them and provide a safer option. Their findings, reported in Applied Physics Letters, are a new step in oncotripsy, the singling out and killing of cancer cells based on their physical properties.

NUS researchers use light emitted from nanoparticles to intricately control biological processes
Researchers from the National University of Singapore have developed a method to give more control to optogenetics, by using specially designed nanoparticles and nanoclusters (dubbed ‘superballs’). These nanoparticles and superballs can emit different colours of light when excited by lasers at different wavelengths. These different colours of light can then be used to trigger specific biological processes.