Oak Ridge National Laboratory joins four other national laboratories — Idaho, Los Alamos, Lawrence Livermore and Lawrence Berkeley — that have institutes named after nuclear chemist and Nobel Prize winner Glenn T. Seaborg.
news, journals and articles from all over the world.

Oak Ridge National Laboratory joins four other national laboratories — Idaho, Los Alamos, Lawrence Livermore and Lawrence Berkeley — that have institutes named after nuclear chemist and Nobel Prize winner Glenn T. Seaborg.
The science of radiation chemistry flourished from the 1940s through the 1960s as the United States weighed the benefits of several different reactor technologies to power an energy-hungry planet.
Now, as a new generation of nuclear reactor designers develop advanced molten salt reactor concepts as an alternative for providing reliable, sustainable, carbon-free power, the need for radiation chemistry has never been greater.
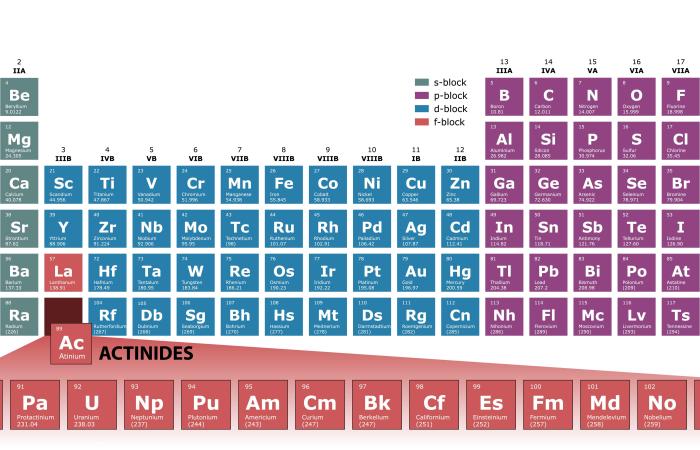
Researchers have for the first time examined in detail a compound of einsteinium (Es). Einsteinium is one of the synthetic elements and is also the heaviest element currently available for classical chemistry studies. These experimental results chart the path to exploring the fundamental behavior of rare heavy elements and could lead to a new understanding of chemistry across the Periodic Table.

Scientists at Berkeley Lab have demonstrated how to image samples of heavy elements as small as a single nanogram. The new approach will help scientists advance new technologies for medical imaging and cancer therapies.
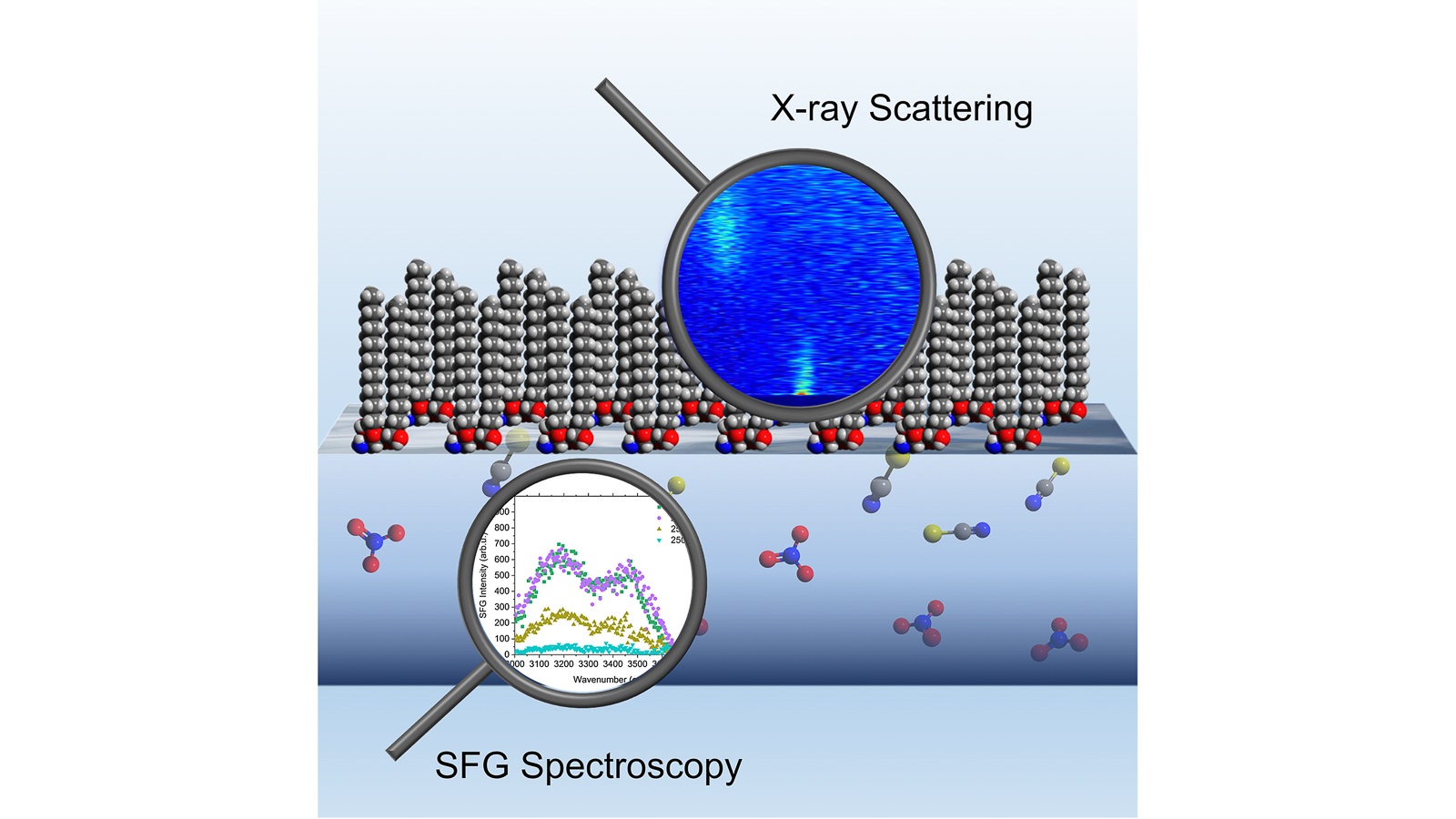
Scientists investigate a process that recycles nuclear and electronic waste materials to extend their lifetime and reduce expensive and invasive mining.

Since element 99 – einsteinium – was discovered in 1952 at the Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) from the debris of the first hydrogen bomb, scientists have performed very few experiments with it because it is so hard to create and is exceptionally radioactive. A team of Berkeley Lab chemists has overcome these obstacles to report the first study characterizing some of its properties, opening the door to a better understanding of the remaining transuranic elements of the actinide series.

An international team of researchers has demonstrated how curium — element 96 in the periodic table and one of the last that can be seen with the naked eye — responds to the application of high pressure created by squeezing a sample between two diamonds.
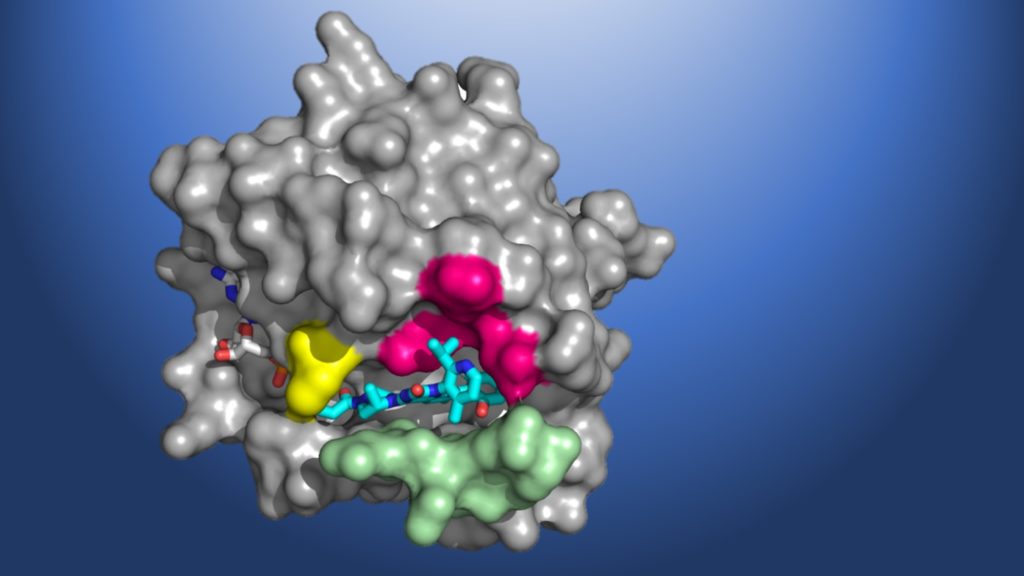
This edition of Science Snapshots highlights the discovery of an investigational cancer drug that targets tumors caused by mutations in the KRAS gene, the development of a new library of artificial proteins that could accelerate the design of new materials, and new insight into the natural toughening mechanism behind adult tooth enamel.

For the first time, Argonne scientists have printed 3D parts that pave the way to recycling up to 97 percent of the waste produced by nuclear reactors. From left to right: Peter Kozak, Andrew Breshears, M Alex Brown, co-authors of a recent Scientific Reports article detailing their breakthrough. (Image by Argonne National Laboratory.)