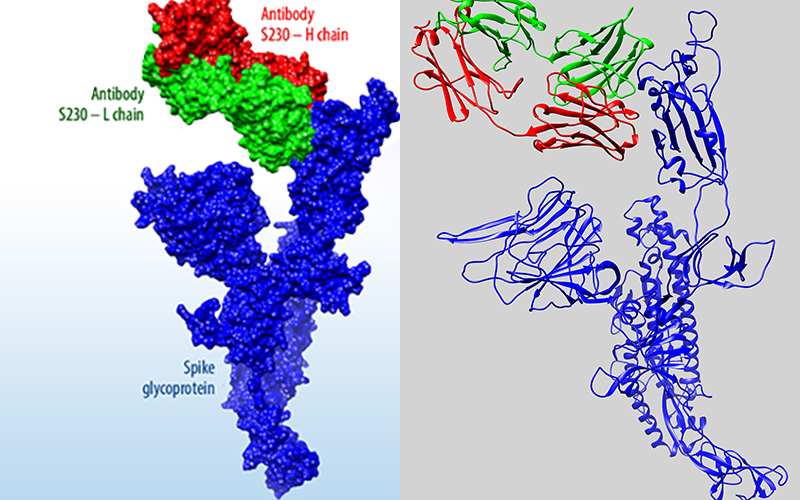
In a groundbreaking development for addressing future viral pandemics, a multi-institutional team involving Lawrence Livermore National Laboratory researchers has successfully combined an artificial intelligence-backed platform with supercomputing to redesign and restore the effectiveness of antibodies whose ability to fight viruses has been compromised by viral evolution. The work was published in the journal Nature.
Tag: Antibody
Signs of Multiple Sclerosis Show Up in Blood Years Before Symptoms
In a discovery that could hasten treatment for patients with multiple sclerosis (MS), UC San Francisco scientists have discovered a harbinger in the blood of some people who later went on to develop the disease.
Targeting inflammatory protein could help treat severe asthma
Australian scientists have revealed a promising new treatment for severe asthma, targeting a protein that controls inflammation and scarring of the airways.
Snaking toward a universal antivenom
Scripps Research scientists discovered antibodies that protect against a host of lethal snake venoms.
Chula Researcher’s Innovative Wireless Hepatitis B Test Kit for Complete Screening and Data Collection in One Step
Chula researchers have developed a remarkable wireless hepatitis B virus test kit to screen for infection and collect data for an online database that’s fast and complete in one step.
‘Tipping The Balance’ Of Immune Cells from Bad to Good Reverses Multiple Sclerosis Symptoms in Mice
According to the federal government’s National Institute of Neurological Disorders and Stroke, nearly 3 million people worldwide — with almost a third in the United States — are living with multiple sclerosis (MS), a disabling neurological disease in which the body’s immune system mistakenly attacks nerves feeding information to the central nervous system (the brain and spinal cord). Although rarely fatal, MS can lead to long-term disabilities, and impair movement, muscle control, vision and cognition.
Artificial Intelligence Aids Discovery of Super Tight-Binding Antibodies
UC San Diego scientists developed an artificial intelligence tool that could accelerate the development of new high affinity antibody drugs.
MD Anderson Research Highlights for November 30, 2022
The University of Texas MD Anderson Cancer Center’s Research Highlights provides a glimpse into recent basic, translational and clinical cancer research from MD Anderson experts.
Finding the answers hidden in our antibodies
An innovative protocol called PepSeq is changing the way researchers test for contagious diseases—and this knowledge should change the way humanity responds to future pandemics. NAU researcher Jason Ladner and a team of collaborators from TGen published a comprehensive study about PepSeq that lays out the process, the tool and how to interpret the results.
Johns Hopkins Medicine Scientists Create Nanobody That Can Punch Through Tough Brain Cells and Potentially Treat Parkinson’s Disease
Johns Hopkins Medicine researchers have helped develop a nanobody capable of getting through the tough exterior of brain cells and untangling misshapen proteins that lead to Parkinson’s disease, Lewy body dementia, and other neurocognitive disorders caused by the damaging protein.
Plant virus plus immune cell-activating antibody clear colon cancer in mice, prevent recurrence
A new combination therapy to combat cancer could one day consist of a plant virus and an antibody that activates the immune system’s “natural killer” cells, shows a study by researchers at the University of California San Diego. In mouse models of colon cancer, the combination therapy eliminated all tumors and prevented their recurrence, which in turn resulted in 100% survival. The therapy also increased survival in mouse models of melanoma.
COVID-19 Vaccine Gets Strong Response in Some With Weak Immunity
Researchers at Cedars-Sinai found that patients with inflammatory bowel disease (IBD)─whose treatment can weaken the immune system─produced a strong antibody response to COVID-19 vaccination. The study findings have been published in the Annals of Internal Medicine.
In Covid-19 Vaccinated People, Those with Prior Infection Likely to Have More Antibodies
In what is believed to be one of the largest studies of its kind, Johns Hopkins Medicine researchers have shown that antibody levels against SARS-CoV-2 (the COVID-19 virus) stay more durable — that is, remain higher over an extended period of time — in people who were infected by the virus and then received protection from two doses of messenger RNA (mRNA) vaccine compared with those who only got immunized.
UAMS Research Team Finds Potential Cause of COVID-19 ‘Long-haulers’
A research team has identified a potential cause of long-lasting symptoms experienced by COVID-19 patients, often referred to as long-haulers. The findings were published in the journal, The Public Library of Science ONE (PLOS ONE).
Vaccines grown in eggs induce antibody response against an egg-associated glycan
Researchers have found that viral vaccines grown in eggs, such as the H1N1 flu vaccine, produce an antibody response against a sugar molecule found in eggs, which could have implications for the effectiveness of these vaccines.
Response to COVID-19 Vaccines Varies Widely in Blood Cancer Patients
Patients with a type of blood cancer called multiple myeloma had a widely variable response to COVID-19 vaccines—in some cases, no detectable response—pointing to the need for antibody testing and precautions for these patients after vaccination, according to a study published in Cancer Cell in June.
For Transplant Recipients, Third Time May Be the Charm for Better COVID Vaccine Protection
In a study published today in the Annals of Internal Medicine, Johns Hopkins Medicine researchers say they believe that, for the first time, there is evidence to show that three doses of vaccine increase antibody levels against SARS-CoV-2 — the virus that causes COVID 19 — more than the standard two-dose regimen for people who have received solid organ transplants.
Immune Response to SARS-CoV-2 in an Asymptomatic Pediatric Allergic Cohort
Abstract: Disease-specific COVID-19 pediatric comorbidity has not been studied effectively todate. Atopy and food anaphylaxis disease states require improved characterization of SARS-CoV-2 infection risk. To provide the first such characterization, we assessed serum samples of a highlyatopic, food anaphylactic, asymptomatic…
Organ Transplant Recipients Remain Vulnerable to Covid-19 Even After Second Vaccine Dose
In a study published today in the Journal of the American Medical Association (JAMA), Johns Hopkins Medicine researchers show that although two doses of a vaccine against SARS-CoV-2 — the virus that causes COVID 19 — confers some protection for people who have received solid organ transplants, it’s still not enough to enable them to dispense with masks, physical distancing and other safety measures.

No more needles?
Blood draws are no fun.They hurt. Veins can burst, or even roll — like they’re trying to avoid the needle, too.Oftentimes, doctors use blood samples to check for biomarkers of disease: antibodies that signal a viral or bacterial infection, such as SARS-CoV-2, the virus responsible for COVID-19, or cytokines indicative of inflammation seen in conditions such as rheumatoid arthritis and sepsis.
Dynamic, personalized treatment approach may improve outcomes in gastroesophageal cancers
A phase 2 clinical trial providing personalized treatments based on the genetic profile of metastatic tumors in gastroesophageal cancers has found that using customized treatment approaches, and adapting them over time as tumors become resistant, led to higher rates of survival compared to historical controls.
Low Immunoglobulin G Antibody Levels Found in Severe COVID-19
Article title:Immunoglobulin deficiency as an indicator of disease severity in patients with COVID-19 Authors: Faeq Husain-Syed, István Vadász, Jochen Wilhelm, Hans-Dieter Walmrath, Werner Seeger, Horst-Walter Birk, Birgit Jennert, Hartmut Dietrich, Susanne Herold, Janina Trauth, Khodr Tello, Michael Sander, Rory E.…

UCI researchers develop rapid antibody generation technology
Irvine, Calif., Dec. 7, 2020 — Using the same strain of yeast that ferments wine and makes dough rise, a team led by University of California, Irvine and Harvard Medical School researchers has developed an in vitro technology that can rapidly hypermutate antibodies. The new technology generates antibodies faster than animal immune systems and better than current synthetic methods, giving researchers the tools for evolving exceptionally potent agents, including therapeutic candidates that target SARS-CoV-2.

Llama Nanobodies Could be a Powerful Weapon Against COVID-19
Today in Science, researchers at the University of Pittsburgh School of Medicine describe a new method to extract tiny but extremely powerful SARS-CoV-2 antibody fragments from llamas, which could be fashioned into inhalable therapeutics with the potential to prevent and treat COVID-19.
Mount Sinai Researchers Develop a First-in-Class Humanized Antibody Targeting Bone and Fat
Blocking the Follicle-Stimulating Hormone (FSH) whose levels rise at menopause could solve bone loss and weight gain Senior Author: Mone Zaidi, MD, PhD, MACP, Director of the Mount Sinai Bone Program and Professor of Medicine (Endocrinology, Diabetes and Bone Disease) at…
UChicago Medicine begins clinical trial testing the efficacy of antibody against SARS-CoV-2
The University of Chicago Medicine is launching a clinical trial to examine the potential of an antibody against the SARS-CoV-2 spike protein for treating COVID-19, the disease caused by the novel coronavirus.

Baylor Scott & White Research Institute Expands Efforts in the Fight Against COVID-19
As the global response to the SARS-COV-2 virus that causes COVID-19 approaches 200 days, Baylor Scott & White Research Institute, the research and development arm of Baylor Scott & White Health, is accelerating its pace of bringing clinical trials online.
Baylor Scott & White Research Institute continues to mobilize staff and resources, including components needed to integrate critical patient-safety measures at every participating site within the Baylor Scott & White system for industry sponsored drug trials, investigator-initiated drug trials and research studies, and observational and data studies designed to help increase knowledge around case trends, viral epidemiology, and care best practices.
Coronavirus antibodies fall dramatically in first 3 months after mild cases of COVID-19
A study by UCLA researchers shows that in people with mild cases of COVID-19, antibodies against SARS-CoV-2 — the virus that causes the disease — drop sharply over the first three months after infection, decreasing by roughly half every 73 days. If sustained at that rate, the antibodies would disappear within about a year.
High-Accuracy COVID-19 Antibody Testing Begins in Arkansas
The University of Arkansas for Medical Sciences (UAMS) has developed and begun using high-accuracy antibody testing to determine the magnitude of COVID-19 infection in Arkansas and inform the decisions of policymakers.
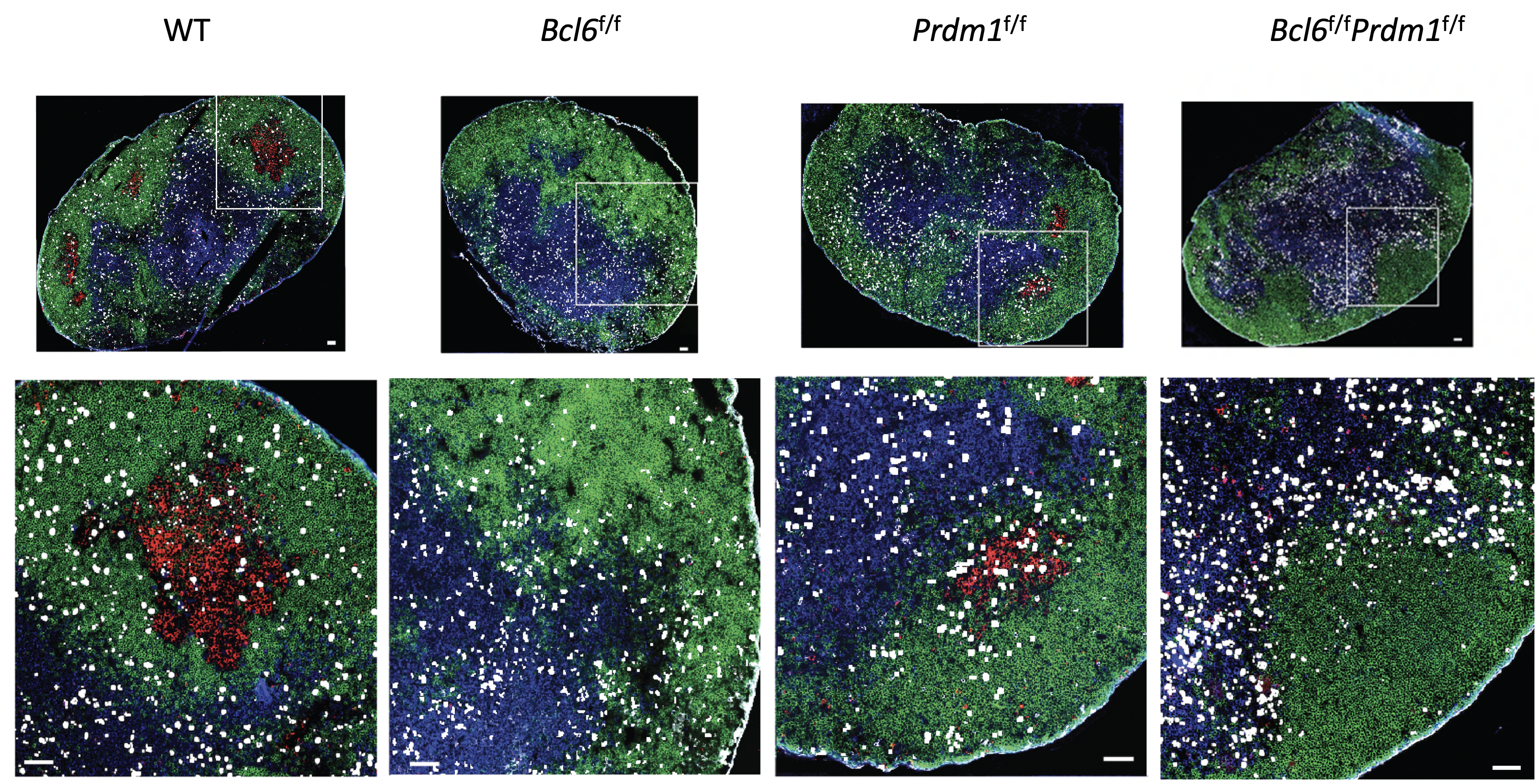
LJI Scientists Investigate a Powerful Protein Behind Antibody Development
Scientists at the La Jolla Institute for Immunology (LJI) have discovered a potential new way to better fight a range of infectious diseases, cancers and even autoimmune diseases. The new study, published recently in Nature Immunology, shows how a protein works as a “master regulator” in the immune system.
COVID-19 Incidence in New York Higher Among Non-White Adults
Recent research by the University at Albany and the New York State Department of Health shows that over 2 million adults in New York were infected with the coronavirus SARS-CoV-2, the virus that causes COVID-19, through late March 2020.
LLNL makes COVID-19 research public through new searchable data portal
To help accelerate discovery of therapeutic antibodies or antiviral drugs for SARS-CoV-2, the virus that causes COVID-19, Lawrence Livermore National Laboratory has launched a searchable data portal to share its COVID-19 research with scientists worldwide and the general public.
Researchers work to quantify the human immune response to COVID-19
Notre Dame’s Merlin Bruening and a team of scientists have begun research to develop point-of-care antibody tests that would help public health officials to better understand how an individual’s immunity to COVID-19 lasts over time.
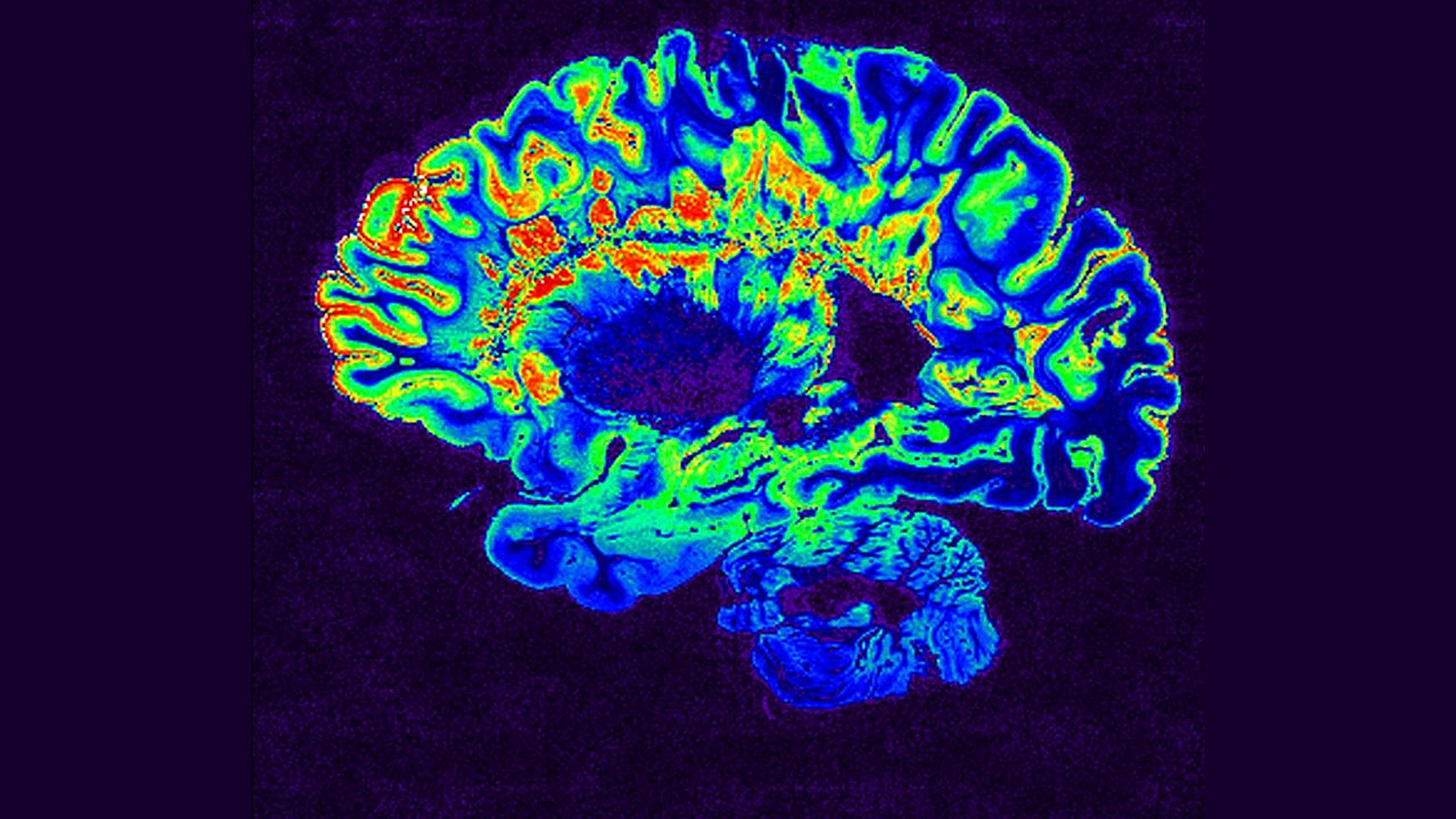
Tip Sheet: Coronavirus biology, Q&A with Joel McHale, brain topographic maps, single cell sequencing and more
Summaries of recent Fred Hutch research findings and other news with links for additional background and media contacts.
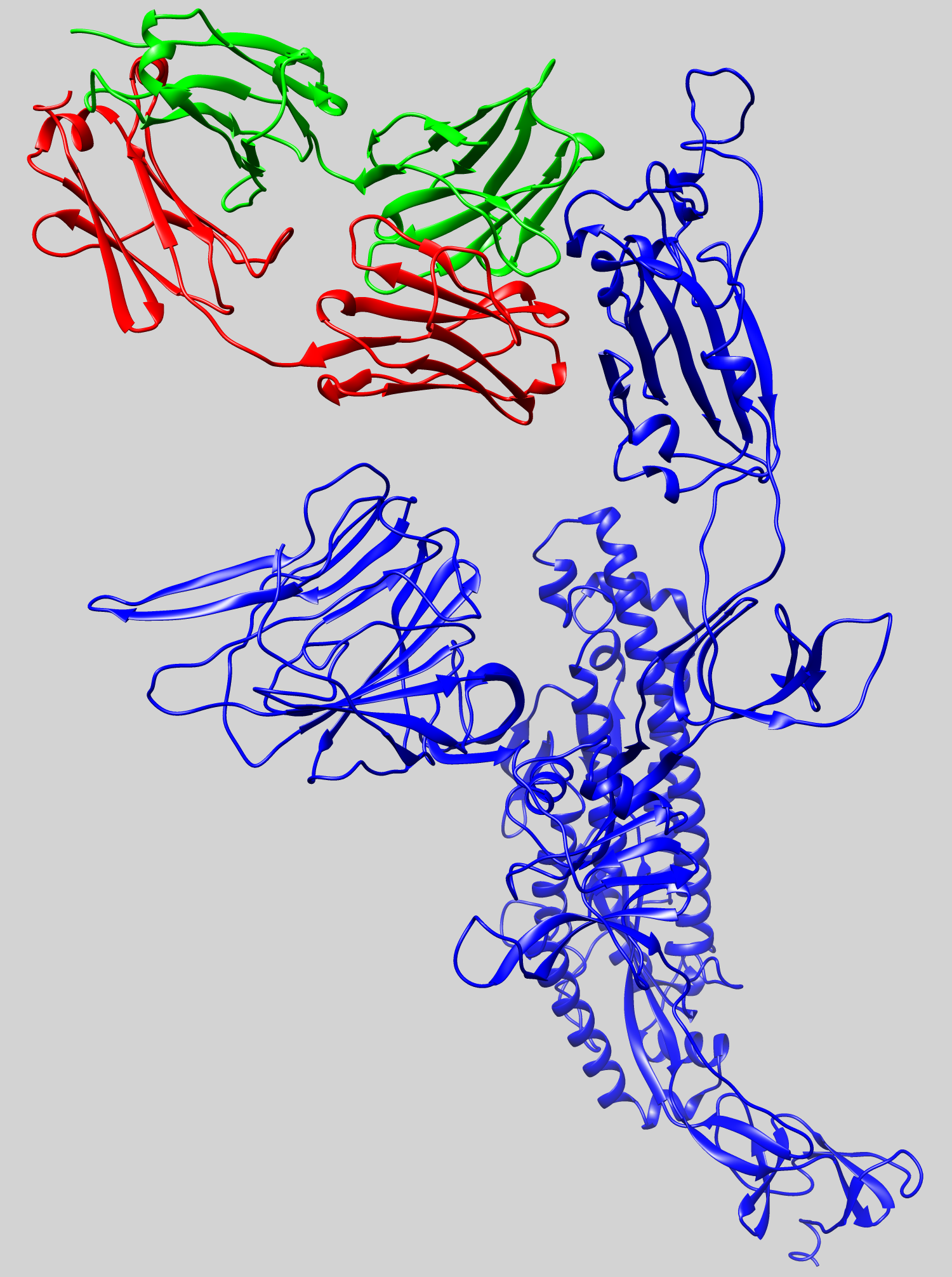
LLNL’s new machine learning platform generates novel COVID-19 antibody sequences for experimental testing
Lawrence Livermore National Laboratory researchers have identified an initial set of therapeutic antibody sequences, designed in a few weeks using machine learning and supercomputing, aimed at binding and neutralizing SARS-CoV-2, the virus that causes COVID-19. The research team is performing experimental testing on the chosen antibody designs.
The search for IgG: What you need to know about antibody testing
As officials consider how to reopen the country safely, researchers say antibody testing will play an important role in navigating those future decisions. But not all antibody tests are created equal, and the presence of antibodies doesn’t necessarily equate to COVID-19 immunity.

Mount Sinai Researchers Collaborate with GenScript to Develop a COVID-19 Antibody to Treat Sick Patients
The highly targeted antibody may block the coronaviruses’ ability to enter human lung cells

Upgrades for LLNL supercomputer from AMD, Penguin Computing aid COVID-19 research
To assist in the COVID-19 research effort, Lawrence Livermore National Laboratory, Penguin Computing and AMD have reached an agreement to upgrade the Lab’s unclassified, Penguin Computing-built Corona high performance computing (HPC) cluster with an in-kind contribution of cutting-edge AMD Instinct™ accelerators, expected to nearly double the peak performance of the machine.
Scientists developing portable viral tests for future pandemics
Iowa State University researchers are developing a portable, inexpensive technology that could allow people to test for the presence of a virus or antibodies without having to go to a medical facility. The technology is still about a year away, but it could come in handy in the event of a resurgence of the coronavirus or for future pandemics.
New Method Detects Toxin Exposure from Harmful Algal Blooms in Human Urine
A newly developed method can detect even low-dose human exposure to microcystins and nodularin in human urine. During harmful algal blooms (HABs), species of cyanobacteria release toxic peptides, including microcystins and nodularin into waterways, impacting wildlife and humans living in these marine environments. These findings are the first to report microcystin concentrations directly from exposed residents impacted by cyanobacteria in Florida, and is a critical step in developing and interpreting clinical diagnostic tests for HABs exposure worldwide.

Mosquitoes Engineered to Repel Dengue Virus
An international team of scientists has synthetically engineered mosquitoes that halt the transmission of the dengue virus. The development marks the first engineered approach in mosquitoes that targets the four known types of dengue, improving upon previous designs that addressed single strains.