SEATTLE — April 3, 2024 — Below are summaries of recent Fred Hutchinson Cancer Center research findings, patient stories and other news. If you’re covering the American Association for Cancer Research (AACR) Annual Meeting, please see our list of and contact to set up interviews.
Tag: Metastasis
MD Anderson Research Highlights for March 20, 2024
The University of Texas MD Anderson Cancer Center’s Research Highlights showcases the latest breakthroughs in cancer care, research and prevention.
Launch of a pioneering translational research programme in Europe
– The TRIP initiative establishes three new joint laboratories that bring together fundamental biomedical research and clinical research with the aim of accelerating the delivery of results to patients.
– TRIP will focus on emerging fields such as immuno-oncology, inflammation, and cellular senescence.
Chronic stress spreads cancer … here’s how
Stress is inevitable. But too much of it can be terrible for our health. Chronic stress can increase our risk for heart disease and strokes. It may also help cancer spread. How this works has remained a mystery—a challenge for cancer care.
Nanoparticle vaccine could curb cancer metastasis to lungs by targeting a protein
UC San Diego engineers have developed an experimental vaccine that could prevent the spread of metastatic cancers to the lungs. Its success lies in targeting a protein known to play a central role in cancer growth and spread, rather than targeting the primary tumor itself.
Palex and Inbiomotion introduce pioneering test to aid oncologists in predicting recurrence and survival rates in breast cancer patients
Spain is the first country in the world to have this technology
• Results showing the clinical utility of the test were published in
Lancet Oncology and the Journal of National Cancer Institute
• The test is now available to oncologists and pathologists and will
benefit an estimated 24,000 patients each year in Spain
Chromosomal instability in cancer cells causes DNA damage and promotes invasiveness
Chromosomal instability is a phenomenon characterised by rapid changes in the number and structure of chromosomes during cell division. It is very common in solid tumours and it is linked to the aggressive spread of cancer, that is to say, metastasis.
When cancer takes a deadly turn: NUS study sheds a new light on cancer metastasis
The ability of cancer cells to metastasise, or spread from one part of the body to another, is one of the reasons why cancer can be extremely challenging to treat. However, the process that drives metastasis is complex and not fully understood.
Scientists make promising discovery in fight against breast cancer
Researchers from the University of Liverpool have created a biomedical compound that has the potential to stop the spread of breast cancer. A recently published paper details these early findings.

New understanding of why kidney cancers become metastatic discovered by MD Anderson researchers
Researchers at The University of Texas MD Anderson Cancer have engineered a new model of aggressive renal cell carcinoma (RCC), highlighting molecular targets and genomic events that trigger chromosomal instability and drive metastatic progression.
The study, published today in Nature Cancer, demonstrates that the loss of a cluster of interferon receptor (IFNR) genes plays a pivotal role in allowing cancer cells to become tolerant of chromosomal instability. This genomic feature may be used to help clinicians predict a tumor’s potential to become metastatic and treatment resistant.
Researchers discovered how melanoma changes its environment to support metastasis
A new study conducted at Tel Aviv University and the Sheba Medical Center reveals how melanoma cancer cells affect their close environment to support their needs – by forming new lymph vessels in the dermis in order to go deeper into the skin and spread through the body.
Rethinking the Protein Inhibitor Approach to Cancer Therapy
A new method than enables researchers to dial up or tone down the amount of a certain metastatic protein inhibitor (BACH1) within a cell could provide a new path in cancer research that reassesses the effectiveness of protein inhibitors to treat disease.
Study of Cancer Metastasis, Most Common Cause of Cancer Death, Gets $35 Million Boost at Johns Hopkins Medicine
With a $35 million gift from researcher, philanthropist and race car driver Theodore Giovanis, scientists at Johns Hopkins Medicine will study the biological roots of the most fatal aspect of cancer: how it metastasizes, or spreads, through the body.
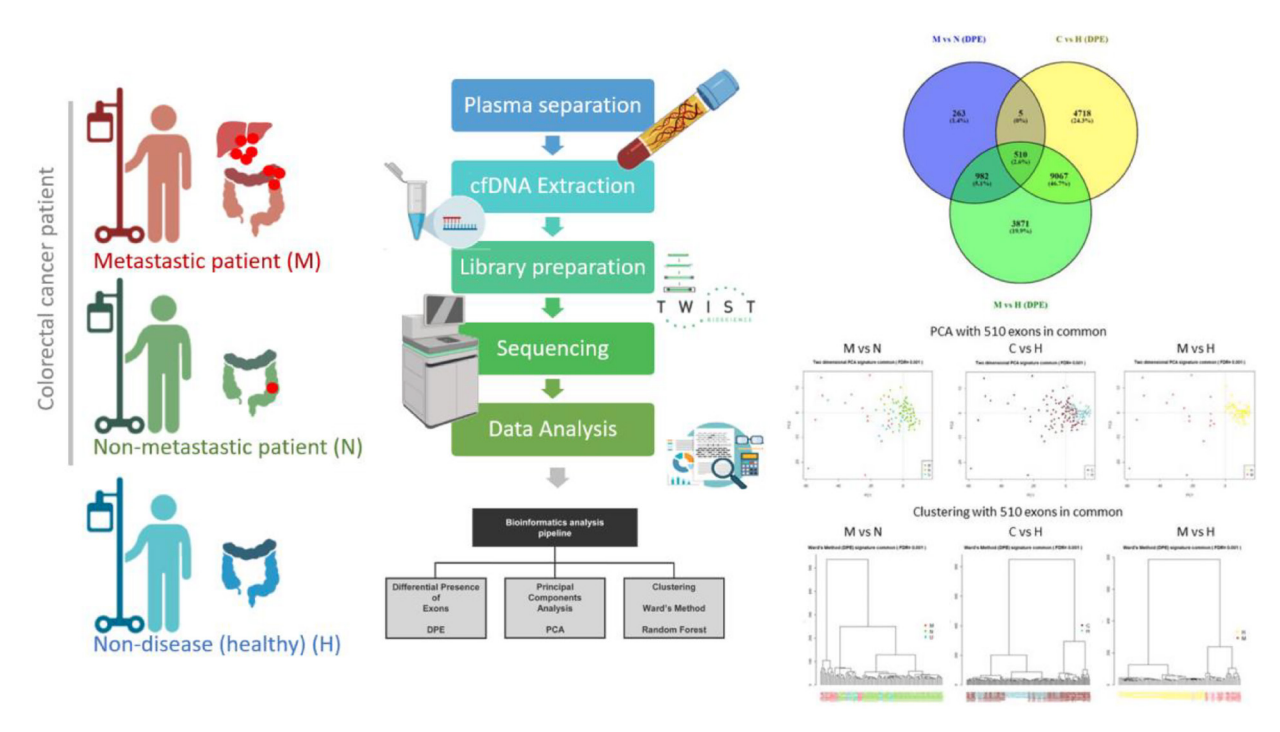
New method of clustering colorectal cancer patients using DPE sequencing
A new editorial paper was published in Oncoscience (Volume 10) on March 23, 2023, entitled, “New method of clustering colorectal cancer patients using differential presence of exons (DPE) sequencing.”
A step towards precision oncology for patients with metastatic urothelial carcinoma
Dr. Niklas Klümper, resident at the Clinic for Urology and working group leader at the Institute for Experimental Oncology at the University Hospital Bonn (UKB), was awarded the C. E. Alken Prize in recognition of his outstanding scientific uro-oncological work.
How Metastatic Cancer Causes Leaky Blood Vessels
In APL Bioengineering, researchers examine the local communication between endothelial cells and tumors cells and its effects on endothelial cell orientation. The approach uses co-cultured human umbilical vein endothelial cells and breast epithelial tumor cell lines to simulate the tumor-endothelial interaction. The group found the clockwise chirality of the hUVECs was less affected by local hormone signaling and more so by direct physical contact with tumor cells. Specific proteins on the tumor cell binding to others on endothelial cells appeared to play a role in changing the clockwise chirality of hUVECs.
Extracellular Viscosity Linked to Cancer Spread
New research findings show how higher viscosity, or resistance to flow, of the extracellular fluid that surrounds cells enables cancer cells to migrate more rapidly from a primary tumor to other sites in the body.
Breast cancer cells use forces to open up channels through tissue
New method uses nanospheres to measure forces that cancer cells use to spread through tissue
MD Anderson Research Highlights for July 27, 2022
Clinical advances include treating hematologic cancers with effective targeted therapies, circulating tumor DNA as a biomarker for recurrence with colorectal liver metastases, and using magnetic resonance imaging (MRI) to guide surgical decisions for patients with lateral pelvic lymph node metastases in rectal cancer. Laboratory findings offer new understanding of the pancreatic cancer immune microenvironment, melanoma cell states, TP53 mutation status in acute myeloid leukemia (AML), and potential targets for metastatic prostate cancer and GNAS-mutant colorectal cancer.
What the mechanical forces behind protein folding can tell us about metastatic cancer
Cells communicate with themselves and their cellular environment through mechanical bonds. This work advances understanding of the role of these forces on proteins as they interact to accomplish their biological functions, including the control of cancer.
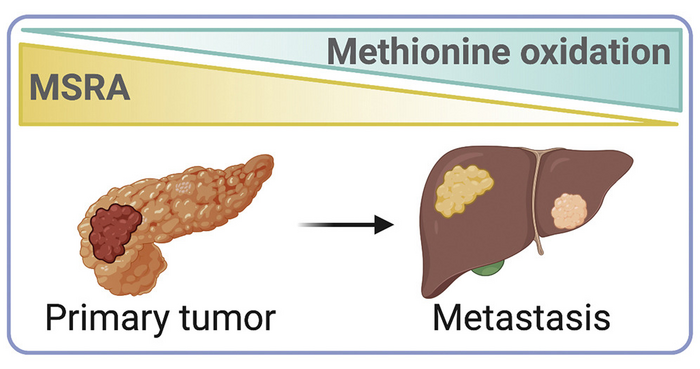
Scientists find trigger that sets off metastasis in pancreatic cancer
Suppressing enzyme that removes oxygen from methionine sparks metastatic spread of cancer.
MD Anderson Research Highlights for November 3, 2021
The University of Texas MD Anderson Cancer Center’s Research Highlights provides a glimpse into recently published studies in basic, translational and clinical cancer research from MD Anderson experts. Current advances include the cost-effectiveness of expanded lung cancer screening criteria, a novel biomarker for predicting immunotherapy responses, development of a technique for multiplex single-cell chromatin profiling, combination immunotherapy for bone metastatic prostate cancer, understanding drivers of lung cancer metastasis, and enabling new T cell therapies for treating COVID-19.
Novel Assay Finds New Mechanism Underlying Red Blood Cell Aging
A multifaceted microfluidic in vitro assay is helping to identify the role of hypoxia on red blood cell aging via the biomechanical pathways. It holds promise for investigating hypoxic effects on the metastatic potential and relevant drug resistance of cancer cells.
Experimental Model of Ovarian Cancer Shows Effect of Healthy Cell Arrangement in Metastasis
A key element to slowing metastasis in ovarian cancer is understanding the mechanisms of how tumor cells invade tissues. In APL Bioengineering, biophysics researchers explain how microscopic defects in how healthy cells line up can alter how easily ovarian cancer cells invade tissue. Using an experimental model, the group found that disruptions in the normal cellular layout, called topological defects, affect the rate of tumor cell invasion.
Small Numbers of Cells in a Tumor Could Be Key Enablers of Cancer Metastasis
Just a small number of cells found in tumors can enable and recruit other types of cells nearby, allowing the cancer to spread to other parts of the body, report Georgetown Lombardi Comprehensive Cancer Center scientists. Working with their research collaborators, the scientists found that ‘enabler cells’ comprise about 20 percent or less of the cells in an aggressive tumor; their small numbers may account for why they are often missed when bulk tissue analyses are used to inform therapeutic decisions.
Ludwig Cancer Research Study Shows Pancreatic Cancer Cells Hit Reverse to Advance in Malignancy
A Ludwig Cancer Research study has identified a previously unrecognized mechanism by which cancer cells of a relatively benign subtype of pancreatic tumors methodically revert—or “de-differentiate”—to a progenitor, or immature, state of cellular development to spawn highly aggressive tumors that are capable of metastasis to the liver and lymph nodes.

Protein rewires metabolism to block cancer cell death, may allow cancer spread
One specific protein may be a master regulator for changing how cancer cells consume nutrients from their environments, preventing cell death and increasing the likelihood the cancer could spread, a study from the University of Notre Dame has shown.

Genomic Test Helps Estimate Risk of Prostate Cancer Metastasis, Death
A commercially available genomic test may help oncologists better determine which patients with recurrent prostate cancer may benefit from hormone therapy, according to new research from the Johns Hopkins Kimmel Cancer Center and 15 other medical centers.
Mount Sinai Researcher Receives NCI Grant to Study New Strategies to Prevent Cancer Metastasis
The Tisch Cancer Institute at Mount Sinai has been awarded a $1.9 million grant by the National Cancer Institute for an innovative study that will explore the mechanisms that enable tumor cells to spread from their original site but remain dormant for some time before becoming metastatic and threatening the lives of patients.
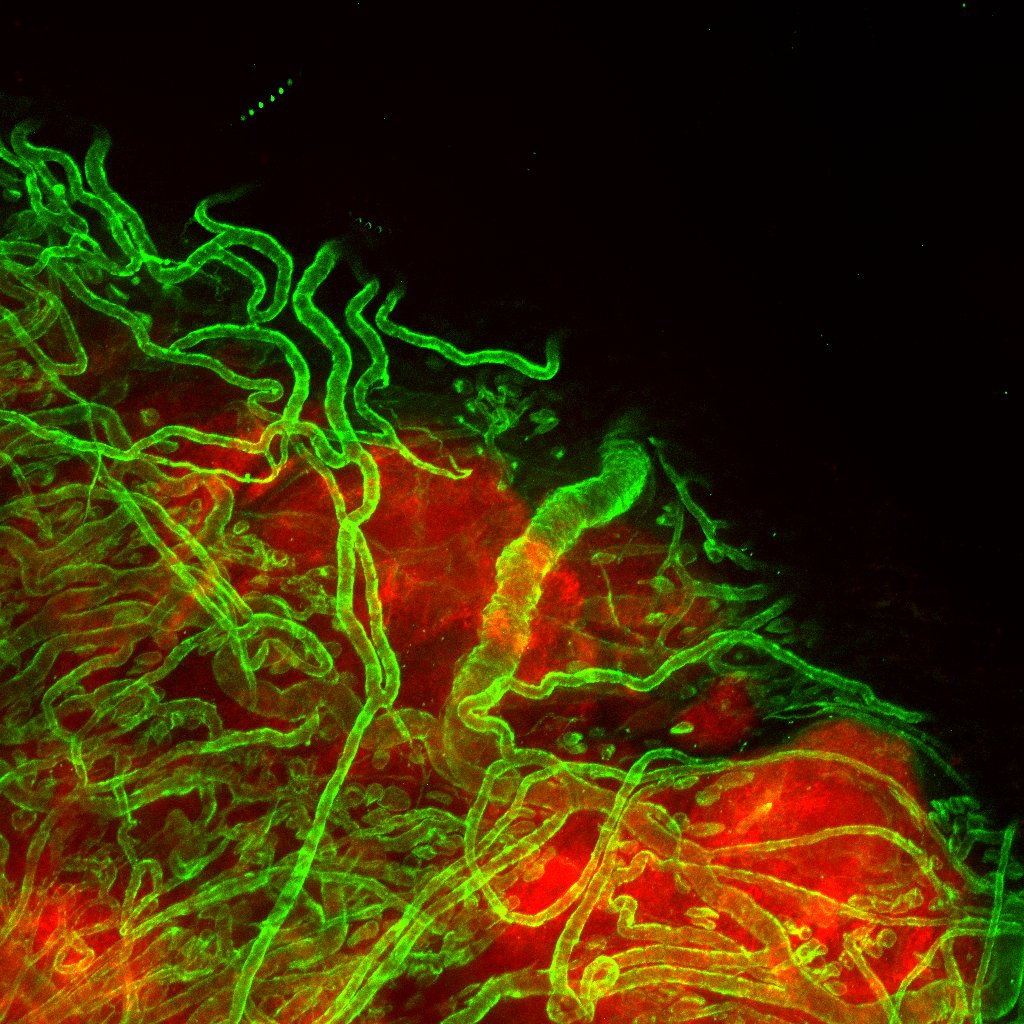
Cancer Cells Take Over Blood Vessels to Spread
In laboratory studies, Johns Hopkins Kimmel Cancer Center and Johns Hopkins University researchers observed a key step in how cancer cells may spread from a primary tumor to a distant site within the body, a process known as metastasis.
Ludwig Chicago Study Identifies a Novel Drug Target for the Control of Cancer Metastasis
Researchers led by Ludwig Chicago Co-director Ralph Weichselbaum and Ronald Rock of the University of Chicago have identified in preclinical studies a potential drug target for curtailing cancer metastasis.
Small Molecule Treatment Reduces Colon Cancer Metastasis
University of Chicago Medicine investigators have found a new way to slow the metastasis of colon cancer: by treating it with a small molecule that essentially locks up cancer cells’ ability to change shape and move throughout the body.

Scientists discover novel drug target for pancreatic cancer
Scientists at Sanford Burnham Prebys Medical Discovery Institute have uncovered a novel drug target, a protein called PPP1R1B, that stops the deadly spread of pancreatic cancer, called metastasis, when inhibited in mice. Published in Gastroenterology, the findings are a first step toward a potential treatment for one of the deadliest cancers known today.

The “M” Word
Danny Welch, PhD, researcher at The University of Kansas Cancer Center, studies metastasis, which is responsible for more than 90% of cancer-related deaths.
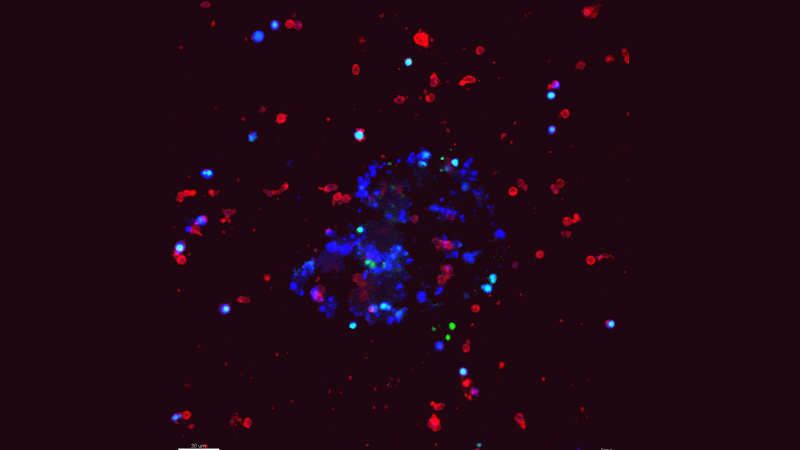
Breast Cancer Cells Can Reprogram Immune Cells to Assist in Metastasis
Johns Hopkins Kimmel Cancer Center investigators report they have uncovered a new mechanism by which invasive breast cancer cells evade the immune system to metastasize, or spread, to other areas of the body. They propose that therapies targeting this process could be developed to halt or prevent metastasis and reduce breast cancer deaths.
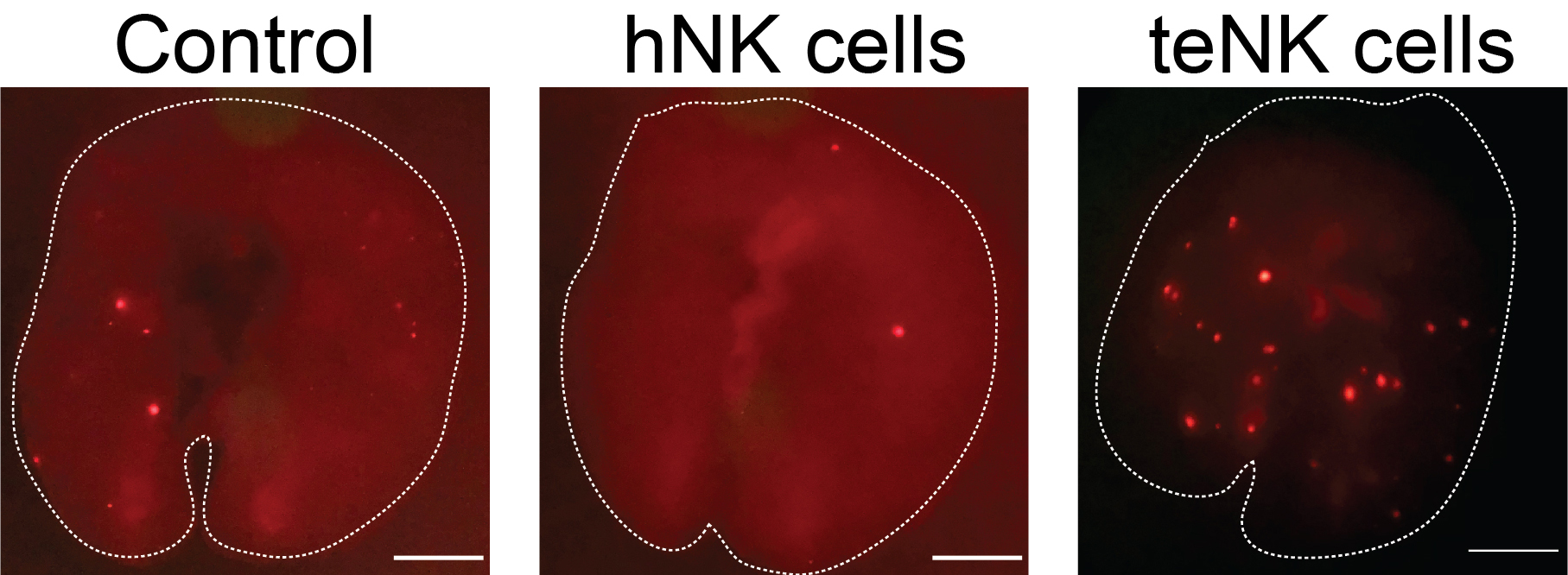
Breast cancer cells turn killer immune cells into allies
Researchers at Johns Hopkins University School of Medicine have discovered that breast cancer cells can alter the function of immune cells known as Natural killer (NK) cells so that instead of killing the cancer cells, they facilitate their spread to other parts of the body. The study, which will be published July 9 in the Journal of Cell Biology (JCB), suggests that preventing this reprogramming might stop breast cancer from metastasizing to other tissues, a major cause of death in breast cancer patients.

Predicting Cancer Behavior Requires Better Understanding of Tumor Cells
Our ability to predict who will get cancer, how patients will respond to treatment, or if patients will relapse is still quite limited, despite advances in the detection of genetic mutations and the establishment of risk factors; recently researchers were inspired to find new ways of looking at the problem. In Biomicrofluidics, they report that using cellular mechanophenotyping, along with traditional methods such as immunostaining and genetic analysis, may provide a more comprehensive view of a tumor.

Continued nicotine use promotes brain tumors in lung cancer patients, Wake Forest study suggests
Researchers at Wake Forest School of Medicine have discovered that nicotine promotes the spread of lung cancer cells into the brain, where they can form deadly metastatic tumors. The study, which will be published June 4 in the Journal of Experimental Medicine (JEM), suggests that nicotine replacement therapies may not be suitable strategies for lung cancer patients attempting to quit smoking. In addition, the researchers show that the naturally occurring drug parthenolide blocks nicotine-induced brain metastasis in mice, suggesting a potential therapeutic option in humans.
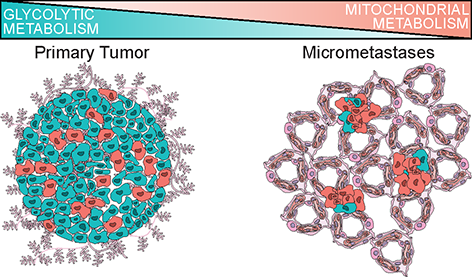
Using new genomic technology, UCI researchers discover breast cancer cells shift their metabolic strategy in order to metastasize
New discovery in breast cancer could lead to better strategies for preventing the spread of cancer cells to other organs in the body, effectively reducing mortality in breast cancer patients.
According to a study, published today in Nature Cell Biology, breast cancer cells shift their metabolic strategy in order to metastasize. Instead of cycling sugar (glucose) for energy, they preferentially use mitochondrial metabolism.

NUS researchers discover breakthrough in cancer diagnosis using big data analytics
A team of researchers from the National University of Singapore has developed a personalised assessment tool which can detect the incidence of cancer, predict patient survivability and determine patient suitability for immunotherapy cancer treatment.
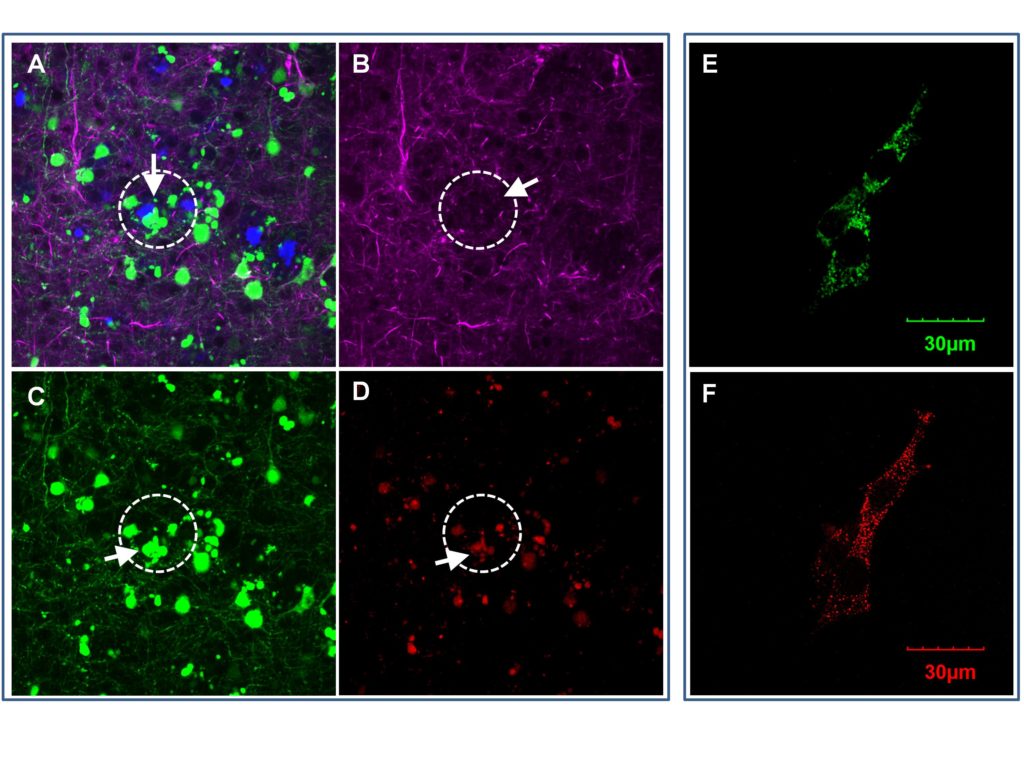
Scientists identify protein associated with ovarian cancer that exacerbates neurodegeneration in Alzheimer’s
Houston Methodist scientists identified a protein found in ovarian cancer that may contribute to declining brain function and Alzheimer’s disease, by combining computational methods and lab research.