A new paper in Biology Methods & Protocols, published by Oxford University Press, shows it may be possible to design vaccines that will induce a stronger immune response to infecting pathogens, such as the virus causing COVID-19.
Tag: Vaccine Development
The potential and challenges of mucosal COVID-19 vaccines
In November 2022, the National Institute of Allergy and Infectious Diseases (NIAID) co-hosted a virtual workshop on the importance and challenges of developing mucosal vaccines for SARS-COV-2. The highlights of this workshop have now been published as a report in npj Vaccines.
Testing vaccine candidates quickly with lab-grown mini-organs
Researchers reporting in ACS Central Science have developed a new testing platform that encapsulates B cells — some of the most important components of the immune system — into miniature “organoids” to make vaccine screening quicker and greatly reduce the number of animals needed for testing.

Memory B cell marker predicts long-lived antibody response to flu vaccine
In a study published in the journal Immunity, researchers describe a distinct and novel subset of memory B cells that predict long-lived antibody responses to influenza vaccination in humans.
CoDe tool makes vaccine development faster and more accurate
A new software tool developed by Texas Biomedical Research Institute and collaborators can help scientists and vaccine developers quickly edit genetic blueprints of pathogens to make them less harmful. The tool, called CoDe – short for Codon Deoptimization – enables users to make precise edits to a genetic code to make genes less functional – in other words, to deoptimize the genes.
Main Line Health researcher releases world’s first complete resource on molecular structure of virus that causes COVID-19
A biomedical researcher at the Lankenau Institute for Medical Research, part of Main Line Health, has created a groundbreaking resource for scientists seeking to develop new and better vaccines in the fight against COVID-19.
National Primate Research Center of Thailand Chulalongkorn University Symposium 2023
As its 11th-anniversary approaches, the National Primate Research Center of Thailand Chulalongkorn University (NPRCT-CU) and the Primates Enterprise Co., Ltd. are pleased to announce two events in February 2023:
Corona vaccine based on new technology tested in clinical study
A new COVID-19 vaccine based on a different platform than current vaccines on the market has been tested in humans for the first time by researchers at Radboud university medical center.
A design of experiments approach to precision vaccine adjuvants
Adjuvants are added to vaccines to improve protection, extend the duration of protection and reduce the dose or number of boosters required.
Rutgers Scientists Produce “DNA Virus Vaccine” to Fight DNA Viruses
Rutgers scientists have developed a new approach to stopping viral infections: a so-called live-attenuated, replication-defective DNA virus vaccine that uses a compound known as centanamycin to generate an altered virus for vaccine development.
Lessons learned from COVID-19 mitigation measures
Researchers analyzed how the expectation of a vaccine influences optimal lockdown measures during a pandemic.
Scientists develop effective intranasal mumps-based COVID-19 vaccine candidate
New research has advanced COVID-19 vaccine work in several ways: using a modified live attenuated mumps virus for delivery, showing that a more stable coronavirus spike protein stimulates a stronger immune response, and suggesting a dose up the nose has an advantage over a shot.
New Genomic Research Shows Why Testing Malaria Vaccines in the Clinic is as Rigorous as Natural Exposure in the Field
Scientists at the University of Maryland School of Medicine’s (UMSOM) Institute for Genome Sciences (IGS) and the UMSOM Center for Vaccine Development and Global Health (CVD), and their collaborators report a new way to test vaccines that may be as rigorous and stringent as exposure to field strains of malaria.
Microparticles could be used to deliver “self-boosting” vaccines
Most vaccines, from measles to Covid-19, require a series of multiple shots before the recipient is considered fully vaccinated.
Nanoparticle vaccine protects against a spectrum of COVID-19-causing variants and related viruses
A new type of vaccine provides protection against a variety of SARS-like betacoronaviruses, including SARS-CoV-2 variants, in mice and monkeys, according to a study led by researchers in the laboratory of Caltech’s Pamela Bjorkman, the David Baltimore Professor of Biology and Bioengineering.
Cleveland Clinic Appoints Ted Ross, Ph.D., as Global Director of Vaccine Development
Ted Ross, Ph.D., has been appointed Global Director of Vaccine Development at Cleveland Clinic.
In this newly created role, he will lead the development of novel vaccine platforms for a variety of infectious diseases, including influenza, HIV and COVID-19. A highly renowned scientist with expertise in virology, vaccines, immunology and microbiology, Dr. Ross’ research focuses on the design of new vaccines and the implementation of new vaccine trials.
Engineering Next-Gen Vaccines
The University of Delaware’s Aditya Kunjapur has been awarded the 2021 American Institute of Chemical Engineers (AIChE) Langer Prize for Innovation and Entrepreneurial Excellence, a highly competitive award that comes with a stipend to pursue “blue sky” ideas.
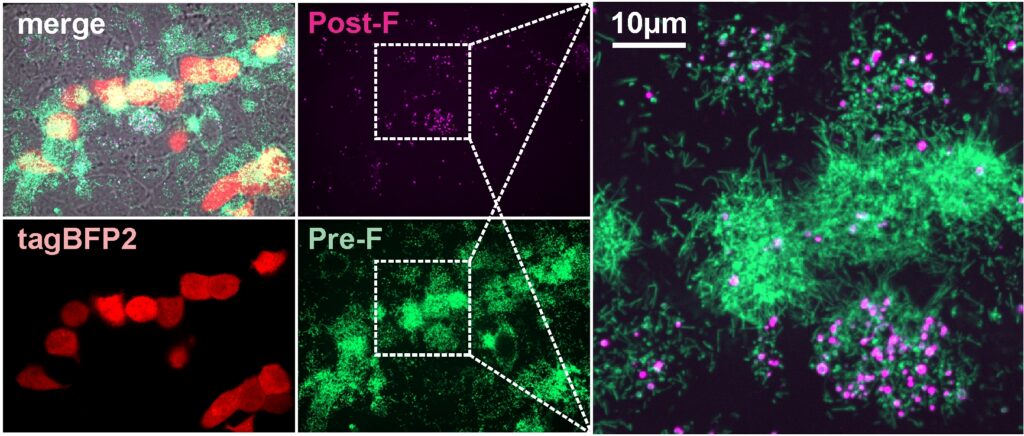
Shape of virus may determine RSV infection outcomes
Using a novel technology, the lab of Michael Vahey at the McKelvey School of Engineering uncovered shape-shifting properties of a common respiratory virus.
These fridge-free COVID-19 vaccines are grown in plants and bacteria
Nanoengineers at the University of California San Diego have developed COVID-19 vaccine candidates that can take the heat. Their key ingredients? Viruses from plants or bacteria.
Children’s Hospital of Philadelphia Researchers Identify Approach for Potential Nontypeable Haemophilus Influenzae Vaccine
Scientists at Children’s Hospital of Philadelphia (CHOP) have identified two proteins that could be used for a potential vaccine against nontypeable Haemophilus influenzae (NTHi). Working in a mouse model, the investigators found that administering two bacterial adhesive proteins that play a key role in helping the bacteria to latch on to respiratory cells and initiate respiratory tract infection stimulated protective immunity against diverse NTHi strains, highlighting the vaccine potential.
UAB’s new Immunology Institute provides new avenues for discovery
UAB has established an interdisciplinary hub for research and patient care in the study of immunity.
Dengue immune function discovery could benefit much-needed vaccine development
The discovery of new possible biomarkers to predict clinical and immune responses to dengue virus infection could be critical to informing future vaccines for the mosquito-borne virus, which saw a record number of over 400 million cases in 2019.
Technion to Award Honorary Doctorate to Pfizer CEO Dr. Albert Bourla
Israel’s Technion will award an honorary doctorate to Pfizer CEO and Chairman Dr. Albert Bourla, for leading the development of the novel vaccine against SARS-CoV-2, the virus that causes COVID-19. The honorary doctorate will be conferred at the Technion Board of Governors meeting in November 2021.
George Washington University Public Health/Medical Experts Available for Media Interviews on COVID-19 Vaccine
On Dec. 17, a U.S. Food and Drug Administration panel is expected to review the data on the Moderna COVID-19 vaccine, paving the way for approval. Researchers at the George Washington University led one of the 100 clinical sites testing…

GenScript Granted Authorization for cPass™ SARS-CoV-2 Neutralization Antibody Detection Test in Brazil
GenScript USA Inc., the world’s leading research reagent provider, announced today that Brazil’s National Health Surveillance Agency (ANVISA) (Agência Nacional de Vigilância Sanitária) has authorized the use of the cPass™ SARS-CoV-2 Neutralization Antibody Detection Kit for detecting neutralizing antibodies. The cPass test is the first and only ANVISA authorized test for detecting neutralizing antibodies to SARS-CoV-2. Neutralizing antibodies specifically block the ability of a virus to infect a cell and are well-recognized to confer immunity.
Rutgers Infectious Disease Expert Available to Discuss Moderna’s Coronavirus Vaccine
Rutgers’ principal investigator for the Moderna phase 3 coronavirus vaccine clinical trial at Rutgers New Jersey Medical School is available to discuss the drugmaker’s vaccine, which the company has announced is 94.5 percent effective. Moderna intends to seek an emergency…

New Model of Tuberculosis Will Speed Search for New Vaccines, Therapies
A team from Seattle Children’s Research Institute has developed an improved mouse model for tuberculosis that replicates characteristics of the human disease not possible in other available models. The ultra-low dose model, named that because mice are infected with 1-3…
Key details about broadly neutralizing antibodies provide insights for universal flu vaccine
New research from an immunology team at the University of Chicago may shed light on the challenges of developing a universal flu vaccine that would provide long-lasting and broad protection against influenza viruses.
Building “ToxAll” — a smart, self-assembling nano-vaccine to prevent toxoplasmosis
A team of researchers at the University of Chicago have developed a self-assembling nanoparticle to create a toolbox for treating infections such as Toxoplasma gondii, a serious parasitic infection.
Something old, something new combine for effective vaccine against parasitic skin disease
Scientists are planning for Phase 1 human trials of a vaccine they developed by using CRISPR gene-editing technology to mutate the parasite that causes leishmaniasis, a skin disease common in tropical regions of the world and gaining ground in the United States.
Experimental vaccine that boosts antigen production shows promise against COVID-19
A bioengineering technique to boost production of specific proteins could be the basis of an effective vaccine against the novel coronavirus that causes COVID-19, new research suggests.
Bouncing, Sticking, Exploding Viruses: Understanding the Surface Chemistry of SARS-CoV-2
Better understanding of the surface chemistry of the SARS-CoV-2 virus is needed to reduce transmission and accelerate vaccine design.

New intranasal vaccine platform provides potential for more effective vaccines with fewer side effects
A new study at the University of Chicago and Duke University finds that a new type of intranasal vaccine induces a strong immune response in lungs, with possible implications for COVID-19. The system uses nanofibers tagged with antigens to prime the immune system against a potential invasion.

Viruses Can Steal Our Genetic Code to Create New Human-Virus Genes
Like a scene out of “Invasion of the Body Snatchers,” a virus infects a host and converts it into a factory for making more copies of itself.
Soup to Nuts
The COVID-19 pandemic demands action on many fronts, from prevention to testing to treatment. Not content to focus its research efforts on just one, the laboratory of George Church in the Blavatnik Institute at Harvard Medical School and the Wyss Institute for Biologically Inspired Engineering at Harvard University is tackling the problem from seven different angles.
COVID-19 and the Ethical Questions it Poses
The coronavirus pandemic has been unprecedented in its impact, leaving no aspect of life unaffected from its arrival in late 2019. From day-to-day impacts on work, school, social gatherings, and travel, to larger shockwaves to the world’s economy and health…
A Coronavirus Vaccine Is in the Works But Won’t Just Emerge Overnight
Supriya Munshaw, a senior lecturer at Johns Hopkins Carey Business School, offers insights on the likely time frame for a coronavirus vaccine, the steps involved in developing one, the most promising candidates currently in the labs of biotech companies, and why, years after the MERS and SARS outbreaks, a coronavirus vaccine still has not been produced.

Tulane University awarded $10.3 million to test therapeutics, vaccines for novel coronavirus
The National Institutes of Health has awarded Tulane National Primate Research Center a contract of up to $10.3 million to evaluate vaccines and treatments to combat coronavirus disease 2019.
University of Kentucky Researchers Unite to Fight COVID-19
Researchers and faculty from multiple disciplines across the University of Kentucky are coming together as part of the global effort to treat, understand and eradicate COVID-19. A new workgroup within UK’s College of Medicine is bringing together experts from across the campus to focus on advising COVID-19 patient care and clinical trials based on emerging research and potential treatment options.
The Department of Energy Tackling the Challenge of Coronavirus
The Department of Energy has a vital role to play in the national response to COVID-19. Researchers have already used tools at national laboratories to make major inroads to analyzing the virus and its spread.

Tulane University launches new coronavirus research program to develop a vaccine and advanced diagnostics
From working to develop one of the first nonhuman primate models for the 2019 novel coronavirus disease (COVID-19) to designing new nanotechnology-based tests to rapidly diagnose infections, researchers at Tulane University are responding across disciplines to the emerging coronavirus epidemic.