Prof. Dr. Thanyavee expressed her appreciation and honor for receiving the Outstanding Researcher Award.
Tag: Antiviral
Researchers Use a Novel Approach to Design a COVID-19 Antiviral Drug
Researchers combined the features of clinical drugs to treat hepatitis C and viruses similar to COVID-19. This allowed them to synthesize BBH-1, a promising inhibitor that targets the breakdown of the SARS-CoV-2 virus. The researchers characterized samples using X-ray and neutron diffraction techniques to provide atomic-level insights on the structure of the BBH-1 inhibitor and how it binds to the SARS-CoV-2 protein.
Texas Biomed tapped for national ‘Dream Team’ developing antivirals against COVID-19 and other threats
Texas Biomedical Research Institute Professor Luis Martinez-Sobrido, PhD, an expert in virology, vaccines and antiviral research, has been recruited to collaborate with three of the nine Antiviral Drug Discovery (AViDD) Centers for Pathogens of Pandemic Concern announced by NIH this spring.

Rensselaer Researchers Make Virus-Fighting Face Masks
Rensselaer Polytechnic Institute researchers have developed an accessible way to make N95 face masks not only effective barriers to germs, but on-contact germ killers. The antiviral, antibacterial masks can potentially be worn longer, causing less plastic waste as the masks do not need to be replaced as frequently.
UCSF Awarded $67.5 Million to Develop New Antiviral Therapies
Scientists at the UC San Francisco (UCSF) Quantitative Biosciences Institute (QBI) and the QBI Coronavirus Research Group (QCRG) have been awarded $67.5 million from the National Institute of Allergy and Infectious Diseases (NIAID) to support its mission of pandemic preparedness.
Peginterferon-lambda shows strong antiviral action to accelerate clearance of COVID-19
A clinical study led by Dr. Jordan Feld, a liver specialist at Toronto Centre for Liver Disease, University Health Network (UHN), showed an experimental antiviral drug can significantly speed up recovery for COVID-19 outpatients – patients who do not need to be hospitalized. This could become an important intervention to treat infected patients and help curb community spread, while COVID-19 vaccines are rolled out this year.
CARES Act funds major upgrade to Corona supercomputer for COVID-19 work
With funding from the Coronavirus Aid, Relief and Economic Security (CARES) Act, Lawrence Livermore National Laboratory, chipmaker AMD and information technology company Supermicro have upgraded the supercomputing cluster Corona, providing additional resources to scientists for COVID-19 drug discovery and vaccine research
Fred Hutch opens COVID-19 Clinical Research Center
SEATTLE — Oct. 5, 2020 — Fred Hutchinson Cancer Research Center announced the opening of the COVID-19 Clinical Research Center, or CCRC. Funded by philanthropic donations and public/private partnerships, the CCRC is one of the first stand-alone facilities in the nation designed to test novel interventions to treat and prevent COVID-19.
Baylor Scott & White Health Enrolls First Patients in the World in Trial for Inhaled Remdesivir
Earlier this month, Baylor Scott & White Research Institute enrolled the first four patients in the world on Gilead’s new clinical trial involving an investigational inhaled solution of remdesivir (NCT04539262). This is Gilead’s first trial in COVID-19 patients examining the safety and efficacy of an inhaled solution of the drug in an outpatient setting. The study of an inhaled solution asks whether this mode of delivery can help reduce the amount of virus from the airways earlier.
Scientists at Texas Biomed develop new tool to aid in the development of SARS-CoV-2 antivirals and vaccines
Researchers apply a novel reverse genetics approach to create recombinant SARS-CoV-2San Antonio, Texas (October 1, 2020) – Researchers at Texas Biomedical Research Institute (Texas Biomed) recently published findings from an innovative SARS-CoV-2 study that will assist in the development of new vaccines and antivirals for COVID-19.
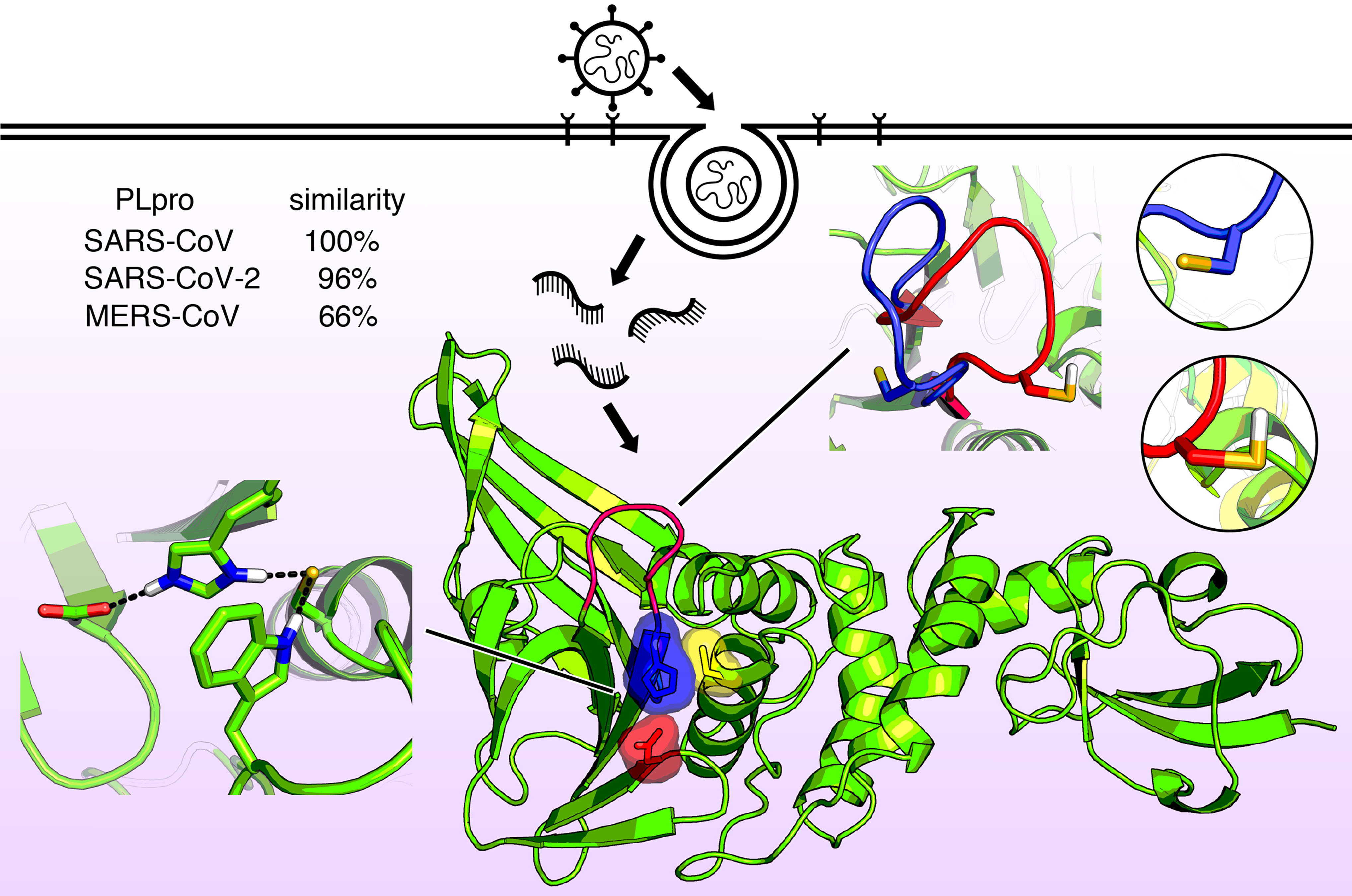
Progress Toward Antiviral Treatments for COVID-19
COVID-19 is caused by the virus SARS-CoV-2, which is structurally similar to the viruses that cause SARS-CoV and MERS-CoV. In The Journal of Chemical Physics, scientists report molecular-level investigations of these viruses, providing a possible pathway to antiviral drugs to fight the diseases. They looked at a viral protein that plays a role in the virus’s ability to replicate and in defeating the host’s immune system, making it an attractive target for potential drug treatments.
New study: Hydroxychloroquine ineffective as a preventive antiviral against COVID-19
esearchers at Case Western Reserve University have added to the growing body of understanding about how hydroxychloroquine (HCQ) is not a possible defense against COVID-19.
Specifically, they found that HCQ is not effective in preventing COVID-19 in patients with lupus and rheumatoid arthritis (RA), suggesting a broader interpretation of HCQ as ineffective preventive medicine for the general population. Their findings were recently published in the Annals of the Rheumatic Diseases.
In Cell Studies, Seaweed Extract Outperforms Remdesivir in Blocking COVID-19 Virus
In a test of antiviral effectiveness against the virus that causes COVID-19, an extract from edible seaweeds substantially outperformed remdesivir, the current standard antiviral used to combat the disease. The research is the latest example of a decoy strategy researchers at Rensselear Polytechnic Institute are developing against viruses like the novel coronavirus that spawned the current global health crisis.
Common FDA-Approved Drug May Effectively Neutralize Virus That Causes COVID-19
A common drug, already approved by the Food and Drug Administration (FDA), may also be a powerful tool in fighting COVID-19, according to research published this week in Antiviral Research.
Next-gen nano technologies to tackle infection and diagnose disease
Next-gen nano technologies that can prevent infection and diagnose disease are set to transform the medical industry as this important UniSA research is awarded more than $2 million dollars under the National Health and Medical Research Council (NHMRC) 2021 Investigator Grants.
Roswell Park to Assess Investigational Immunotherapy Combination in Cancer Patients With COVID-19
A two-drug immunotherapy combination first proposed by a Roswell Park team as an approach for treating cancer will soon be available to cancer patients with COVID-19 through a clinical trial at the Buffalo, N.Y., cancer center.

Sanford Burnham Prebys to develop broad-spectrum antivirals with $10 million Department of Defense grant
Sanford Burnham Prebys Medical Discovery Institute is pleased to announce that Sumit Chanda, Ph.D., has received a $10.2 million, four-year grant from the Department of Defense to develop and advance broad-spectrum antivirals for respiratory diseases. The award aims to provide U.S. military forces and the nation with safe, effective and innovative therapies that combat multiple types of respiratory viruses.