Researchers reporting in ACS’ Nano Letters have developed ultra-absorptive nanofiber swabs that could reduce the number of false-negative SARS-CoV-2 tests by improving sample collection and test sensitivity.

news, journals and articles from all over the world.

Researchers reporting in ACS’ Nano Letters have developed ultra-absorptive nanofiber swabs that could reduce the number of false-negative SARS-CoV-2 tests by improving sample collection and test sensitivity.
AnteoTech marks its entry into the Assay Development market through the design of a high sensitivity COVID-19 Antigen Rapid Test. The test uses an AnteoBind™ activated Europium particle to enable the detection of low viral loads. AnteoTech is pleased to announce the expansion of its business to include Assay Development in addition to its existing Raw Material Supply business.
Results suggest the need for widespread surveillance testing to reduce COVID-19 transmission in group settings
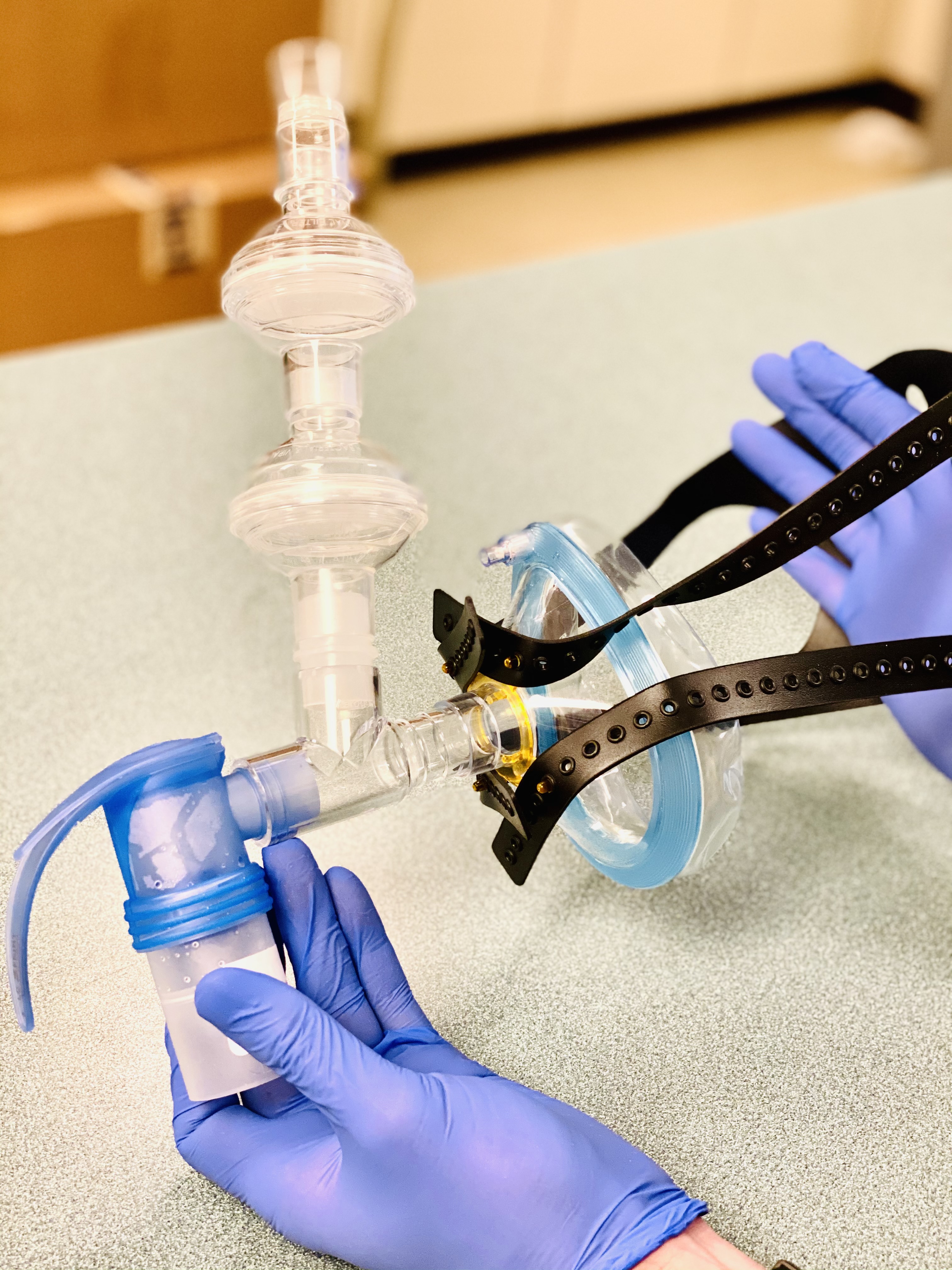
Earlier this month, Baylor Scott & White Research Institute enrolled the first four patients in the world on Gilead’s new clinical trial involving an investigational inhaled solution of remdesivir (NCT04539262). This is Gilead’s first trial in COVID-19 patients examining the safety and efficacy of an inhaled solution of the drug in an outpatient setting. The study of an inhaled solution asks whether this mode of delivery can help reduce the amount of virus from the airways earlier.
Scientists from the Vienna BioCenter have pushed SARS-CoV-2 detection to a new level. Their approach is as sensitive and robust, yet cheaper, simpler and faster to implement than conventional tests. ‘bead-LAMP’ and ‘HomeDip-LAMP’ could be game-changers for population-wide screening, especially in disadvantaged environments, such as developing countries.
Learn how NIH’s new Rapid Acceleration of Diagnostics initiative, or RADx Tech, has mobilized engineers and innovators across the country to bring accurate, rapid, and easy-to-use COVID diagnostic tests to all Americans.
The Association for Molecular Pathology (AMP), the premier global, molecular diagnostic professional society, today released the preliminary results of its April 2020 SARS-CoV-2 Testing Survey for clinical laboratories. The anonymous survey was created and administered to document clinical laboratory efforts and experiences. The results will be used to help inform future advocacy and clinical practice programs related to pandemic responses.
A technology spun from carbon nanotube sensors discovered 20 years ago by Lawrence Berkeley National Laboratory (Berkeley Lab) scientists could one day help healthcare providers test patients for COVID-19, the disease caused by the coronavirus SARS-CoV-2.

Preliminary results from a scientific study aimed at measuring the spread of the novel coronavirus in Indiana show a general population prevalence of about 2.8 percent of the state’s population.
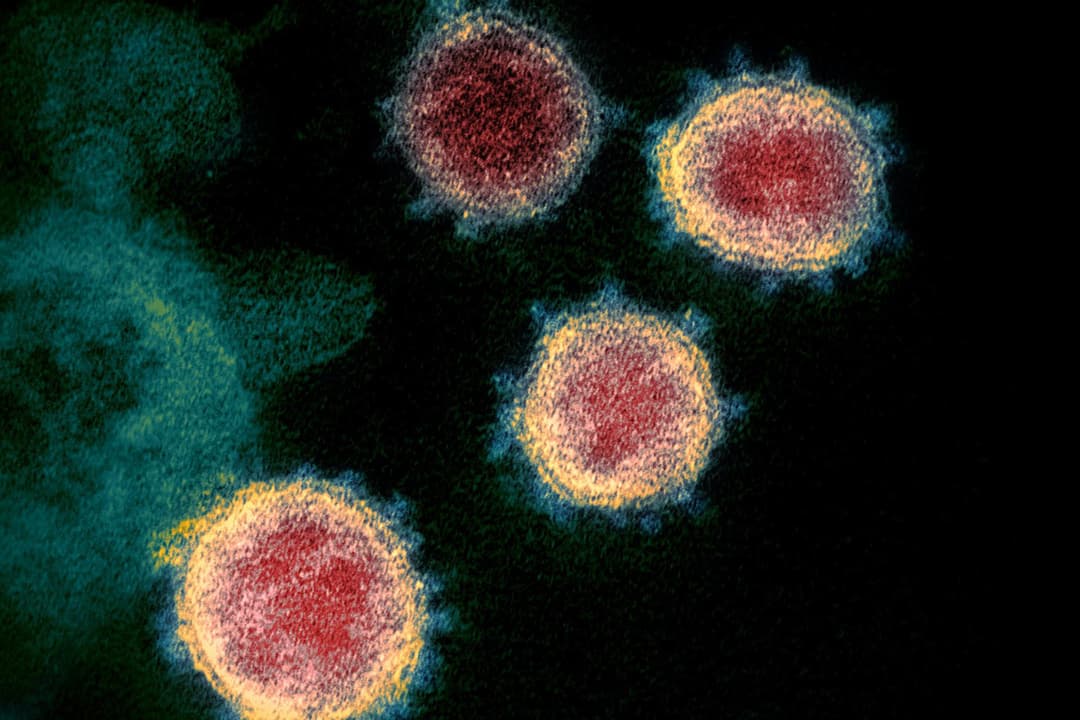
Program makes COVID-19 testing available to thousands of students in effort to track the novel coronavirus and better position the campus to resume in-person activities in the fall.

The Indiana University Richard M. Fairbanks School of Public Health at IUPUI is collaborating with the state health department to conduct a scientific study to measure the spread of COVID-19 throughout Indiana.
Researchers at the McKelvey School of Engineering at Washington University in St. Louis received funding for a new COVID-19 test that is based on brand new technology that won’t require brand new tools, making it easy for clinicians to use.
Physicians and scientists at UC San Diego Health have launched a pair of serological tests that will look for novel coronavirus antibodies—evidence in persons tested that they have previously been infected by the viral cause of COVID-19, even if they never experienced tell-tale symptoms.
Answering the growing need for additional testing, and in direct response to shortages encountered in testing kit supply pipelines, the Coriell Institute for Medical Research is now offering services to create viral testing collection tubes containing different media and/or buffer formulations, for use in COVID-19 testing kits. Coriell is also exploring the feasibility of saliva as a means for testing for the new coronavirus.
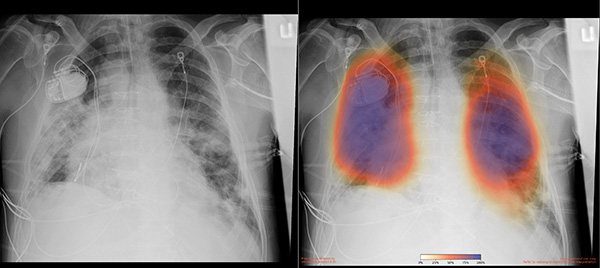
With support from Amazon Web Services, UC San Diego Health physicians are using AI in a clinical research study aimed at speeding the detection of pneumonia, a condition associated with severe COVID-19.
Rutgers’ RUCDR Infinite Biologics has launched a test for the SARS-CoV-2 coronavirus and is using its automation experience and infrastructure to test as many as tens of thousands of samples daily. RUCDR has also submitted an emergency use authorization request for a saliva collection method that will allow for broader population screening.

Using a research assay called VirScan, scientists plan to study how antibodies from people who have had COVID-19 attack the virus that causes it.
UC Davis Health physicians and medical staff who treated the first case of community transmission of COVID-19 in the U.S. provide a detailed case study of her condition and the medical steps and challenges they experienced before arriving at a diagnosis and treatment.