What if we could identify the earliest warning signs of cardiovascular disease from a simple saliva sample? Scientists think they have found a way to do so. Gum inflammation leads to periodontitis, which is linked with cardiovascular disease.
Tag: Saliva
Children’s dislike of cauliflower, broccoli could be written in their microbiome
Researchers reporting in ACS’ Journal of Agricultural and Food Chemistry have found that levels of volatile, sulfurous compounds are similar in parent-child pairs, suggesting shared oral microbiomes. They also found that high levels cause children to dislike the vegetables.
Study: Dental implant surfaces play major role in tissue attachment, warding off unwanted bacteria
The surface of implants, as well as other medical devices, plays a significant role in the adsorption of oral proteins and the colonization by unwanted microorganisms (a process known as biofouling), according to a new study led by the University at Buffalo and the University of Regensburg.
Normal Breathing Sends Saliva Droplets 7 Feet; Masks Shorten This
The WHO and the CDC recommend keeping a certain distance between people to prevent the spread of COVID-19. These social distancing recommendations are estimated from a variety of studies, but further research about the precise mechanism of virus transport is still needed. In Physics of Fluids, researchers demonstrate normal breathing indoors without a mask can transport saliva droplets capable of carrying virus particles to a distance of 2.2 meters in a matter of 90 seconds.
New wiki on salivary proteins may transform diagnostic testing and personalized medicine
To improve the development of new saliva-based diagnostic tests and personalized medicine, the National Institute of Dental and Craniofacial Research (NIDCR) has supported the development of the Human Salivary Proteome Wiki, the first public platform that catalogs and curates data on each of the thousands of proteins within our saliva.

FDA Approves First At-Home Saliva Collection Test for Coronavirus
Rutgers’ RUCDR Infinite Biologics received an amended emergency use authorization from the FDA late Thursday for the first SARS-CoV-2 coronavirus test that will allow people to collect their own saliva at home and send to a lab for results. The decision follows the FDA’s recent emergency approval to RUCDR Infinite Biologics for the first saliva-based test, which involves health care workers collecting saliva from individuals at testing sites.

New Rutgers Saliva Test for Coronavirus Gets FDA Approval
The FDA has granted emergency use authorization (EUA) to Rutgers’ RUCDR Infinite Biologics and its collaborators for a new collection approach that utilizes saliva as the primary test biomaterial for the SARS-CoV-2 coronavirus, the first such approval granted by the federal agency. The new saliva collection method, which RUCDR developed in partnership with Spectrum Solutions and Accurate Diagnostic Labs (ADL), will allow for broader population screening than the current method of nose and throat swabs.
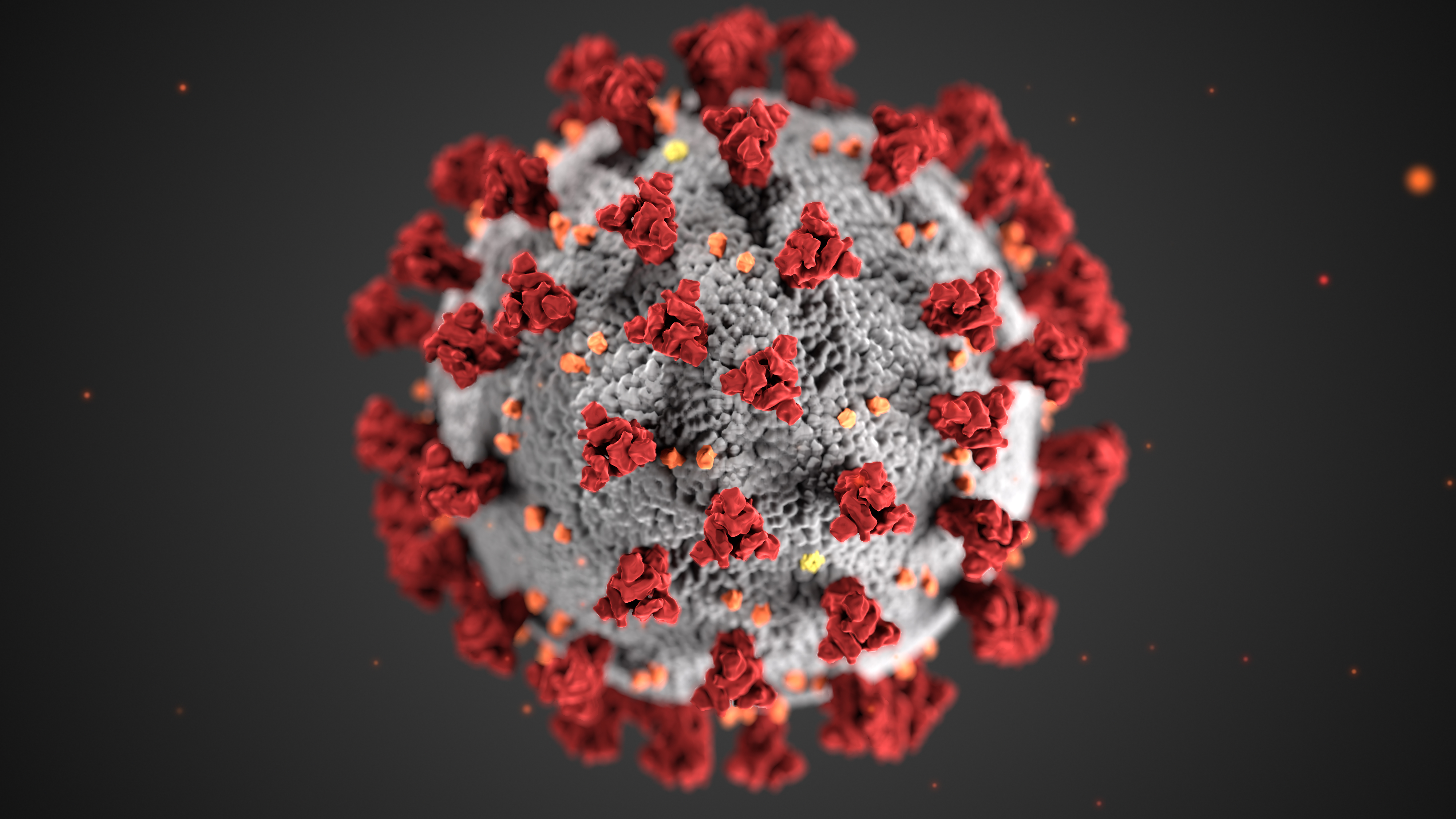
Rutgers Launches Genetic Testing Service for New Coronavirus
Rutgers’ RUCDR Infinite Biologics has launched a test for the SARS-CoV-2 coronavirus and is using its automation experience and infrastructure to test as many as tens of thousands of samples daily. RUCDR has also submitted an emergency use authorization request for a saliva collection method that will allow for broader population screening.