MEDIA ADVISORY Paper title: SARS-CoV-2 seropositivity and subsequent infection risk in healthy young adults: a prospective cohort study Corresponding Author: Stuart C. Sealfon, MD, Professor of Neurology, Neuroscience and Pharmacological Sciences, Icahn School of Medicine at Mount Sinai Bottom Line: Although…
Tag: covid 19 testing
Top Sports Medicine Experts to Address COVID-19 and Athlete Care
Competitors at all levels have been impacted by COVID-19. Team physicians, athletic trainers and physical therapists now face unique challenges caring for athletes. Top sports medicine experts will address key issues surrounding competitive and recreational athlete care during COVID-19 at the 2020 Advanced Team Physician Course, a collaborative effort between ACSM, AMSSM and AOSSM.

Rutgers $5 Million NIH Grant Will Improve Access to COVID-19 Testing in Underserved and Vulnerable Communities
A $5 million National Institutes of Health grant awarded to the New Jersey Alliance for Clinical and Translational Science (NJ ACTS) will launch outreach campaigns and expand access to COVID-19 testing for underserved and vulnerable communities in New Jersey.
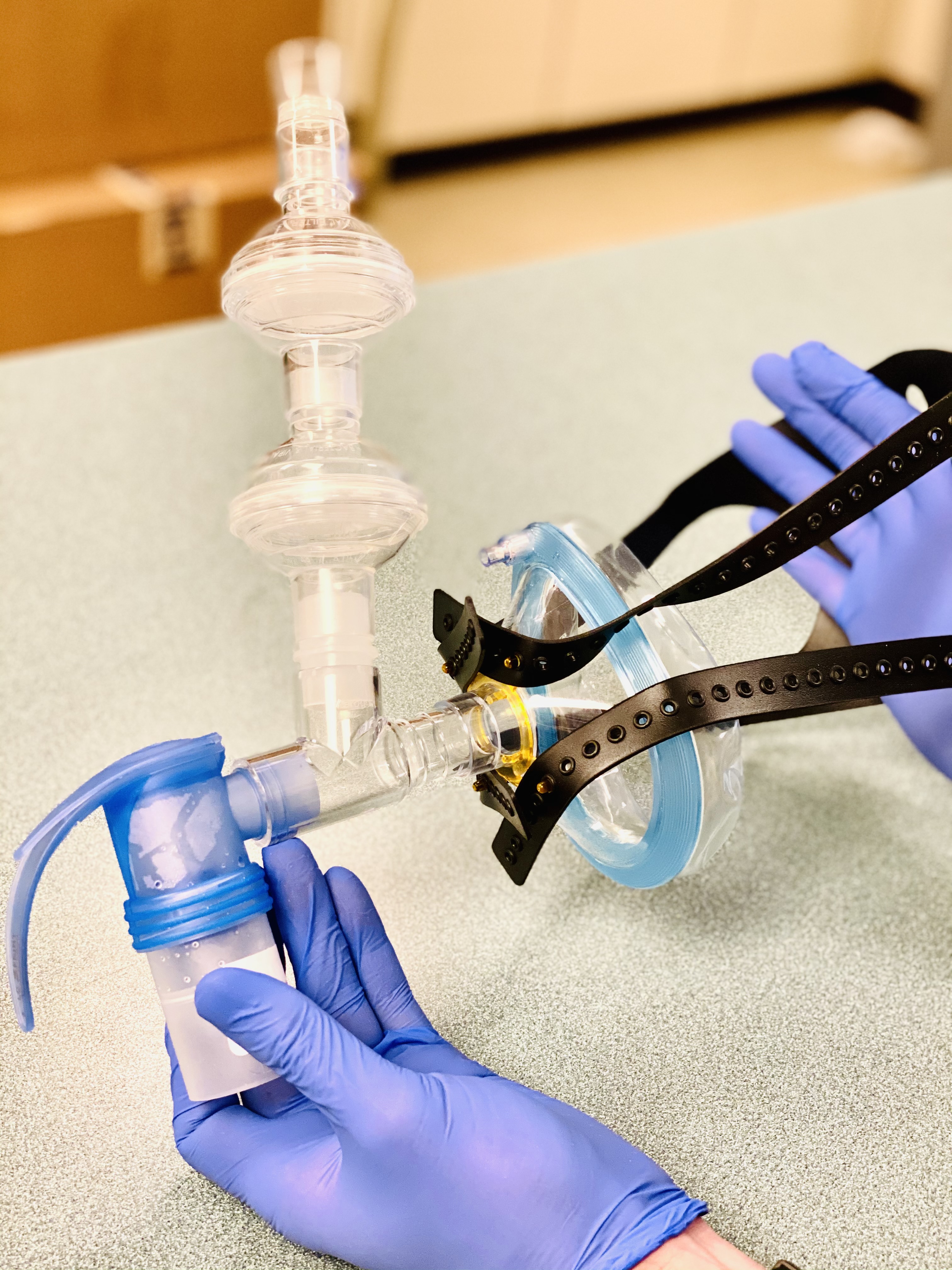
Baylor Scott & White Health Enrolls First Patients in the World in Trial for Inhaled Remdesivir
Earlier this month, Baylor Scott & White Research Institute enrolled the first four patients in the world on Gilead’s new clinical trial involving an investigational inhaled solution of remdesivir (NCT04539262). This is Gilead’s first trial in COVID-19 patients examining the safety and efficacy of an inhaled solution of the drug in an outpatient setting. The study of an inhaled solution asks whether this mode of delivery can help reduce the amount of virus from the airways earlier.

PNNL, UW Broaden Capacity to Test for COVID-19
Researchers from PNNL and UW analyzed kits that could be used as a critical part of a laboratory test used to detect COVID-19 in a sample from an infected person. As a result, two additional kits have been listed as options that labs can consider using, boosting the supply of chemicals and supplies linked to a critical step in the testing process.
ATS Statement on Latest COVID-19 Policy Actions: Convalescent Plasma and Testing of Asymptomatic Patients
The American Thoracic Society is alarmed by two federal agency policy actions that may significantly impact our nation’s COVID-19 response. This week, the FDA issued an emergency use authorization (EUA) for the use of convalescent plasma to treat COVID-19 disease. This action was taken despite a lack of adequate evidence demonstrating a clear benefit for most patients. As pulmonary and critical care clinicians and researchers who treat individuals with COVID-19, we are concerned that an EUA for convalescent plasma has the potential to put patients at risk.

UVA-Designed COVID-19 Swab to Support High-Priority Testing Across Virginia
The Commonwealth of Virginia will receive 60,000 nasopharyngeal swabs weekly to support high-priority COVID-19 testing across the state, thanks to a collaboration led by University of Virginia faculty to design this key testing supply.