Efforts to vaccinate vulnerable populations against respiratory syncytial virus (RSV) faced an uphill battle this season. However, now a new study by researchers at New York Institute of Technology College of Osteopathic Medicine (NYITCOM) could help to combat RSV vaccine misinformation and quell vaccine…
Tag: Food And Drug Administration
Association for Diagnostics & Laboratory Medicine (formerly AACC) Statement on Proposed FDA Rule on Laboratory Developed Tests
We at the Association for Diagnostics & Laboratory Medicine were disappointed to see the U.S. Food & Drug Administration’s (FDA’s) attempt to circumvent Congress with its new proposed rule to duplicate the regulation of laboratory developed tests by placing these tests under FDA authority, in addition to their current regulation under the Centers for Medicare and Medicaid Services.
Ahead of Lecanemab FDA Decision: University of Michigan #Alzheimers expert available for interview
The U.S. Food and Drug Administration is considering full approval of the new Alzheimer’s disease drug Lecanemab (brand name Leqembi), with a decision expected by July 6. University of Michigan Health, Michigan Medicine, has an expert available to talk about…
Study: Blood Vessel Damage Could Be an Alzheimer’s Driver
Blood vessel abnormalities in the eye are a major factor in the progression of Alzheimer’s disease, according to research from Cedars-Sinai investigators published in the peer-reviewed journal Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association.
Cedars-Sinai Cancer Collaborates on a New Type of Clinical Trial
Investigators from Cedars-Sinai Cancer are collaborating on a streamlined clinical trial design in a study called Pragmatica-Lung.
Cedars-Sinai Experts Available to Explain Breaking News About Alzheimer’s Disease Advances
Experts from the Jona Goldrich Center for Alzheimer’s and Memory Disorders at Cedars-Sinai are available for interviews and to comment on research being presented at the conference.
Research reveals widespread use of ineffective COVID-19 treatments after FDA deauthorized their use
In a paper published in JAMA Network Open, physician-scientists assessed the use of these two monoclonal antibodies for patients with COVID-19 before and after FDA deauthorization.
For Youngsters, Getting Ear Tubes No Longer Involves Surgery
Like a lot of young children, from the time she was a year old, Cassidy Clapp was plagued by one ear infection after another.
Chronic Kidney Disease is Curable if Detected Early – Chula’s User-friendly CKD Screening Strips with Results in 15 minutes!
A Chula research team has developed a screening strip kit to detect the early stage of chronic kidney disease (CKD) that’s easy to use, yields quick results, increasing the chance of being cured for patients, and helping to cut over 10 billion baht of the ever-increasing annual healthcare costs for CKD patients. The CKD screening strip kits are expected to be released early next year.

ACI Urges FDA to Address Hand Sanitizer Safety Concerns
The American Cleaning Institute (ACI) is pressing the U.S. Food and Drug Administration (FDA) to clarify policies on labeling and refilling practices involving hand sanitizers.
In a letter sent to the agency, ACI expressed concerns related to the “new, increasingly widespread practice of providing hand sanitizers to consumers in public settings…that are often sold in bulk and used to fill existing dispensers, which can create a serious risk to public health and safety.”
AACI Applauds Approval of COVID-19 Vaccine, Supports Widespread Vaccination Efforts
The Association of American Cancer Institutes (AACI) commends the U.S. Food and Drug Administration (FDA) for its swift action to grant Emergency Use Authorization (EUA) to Pfizer’s COVID-19 vaccine.
UCLA, UCSF gain FDA approval for prostate cancer imaging technique
The University of California’s two nationally ranked medical centers, UCSF and UCLA, and their nuclear medicine teams have obtained approval from the U.S. Food and Drug Administration to offer a new imaging technique for prostate cancer that locates cancer lesions in the pelvic area and other parts of the body to which the tumors have migrated.

Mass Eye and Ear Experts Urge Food and Drug Administration to Allow Over-The-Counter Hearing Aid Devices
Hearing experts advocate in The New England Journal of Medicine for the FDA to implement the Over-the-Counter Hearing Aid Act of 2017. The FDA missed the deadline to release the much-anticipated regulations on August 18, 2020
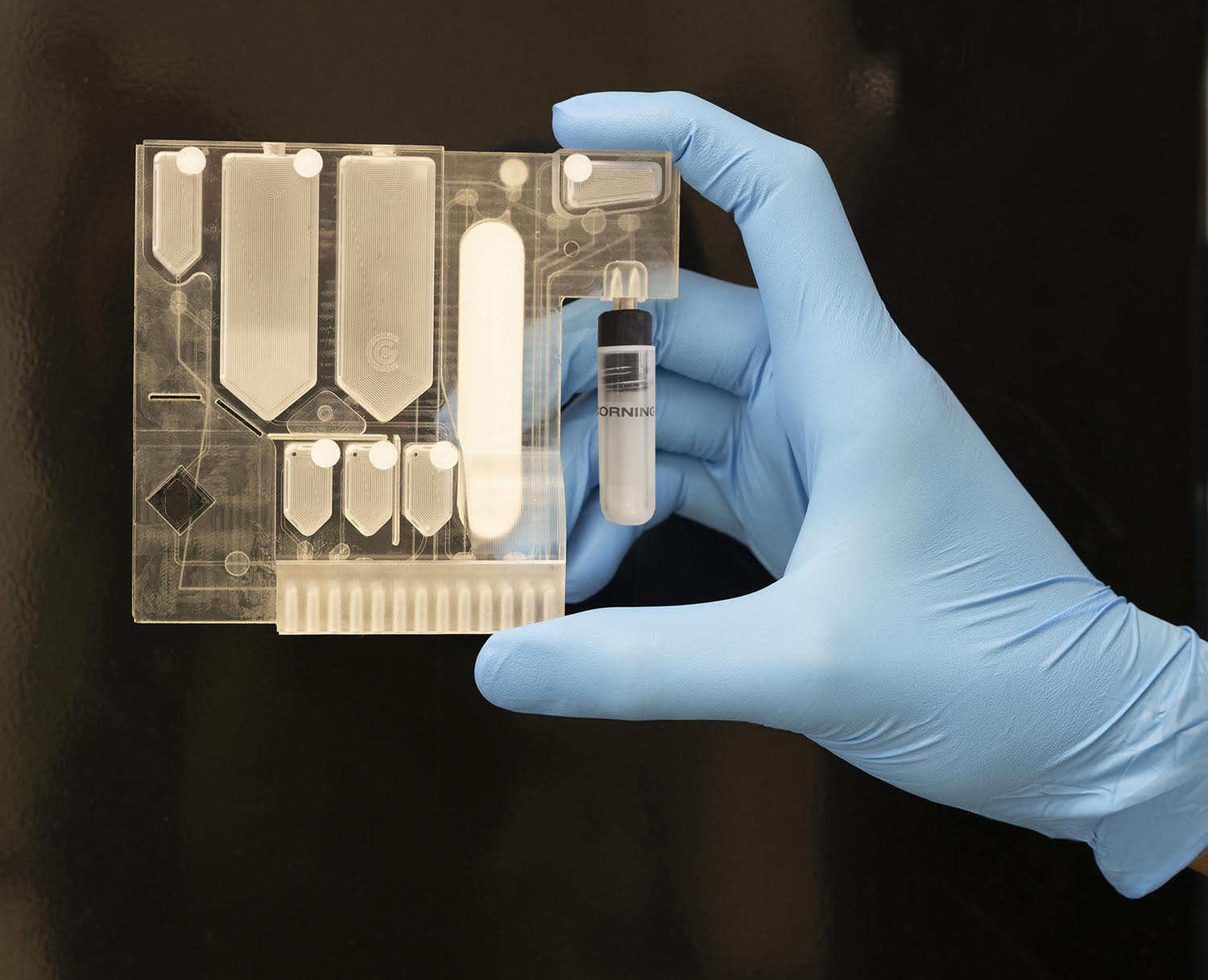
Rapid disease pathogen identification a step closer following successful UAH startup demonstration
Soon it could only take an hour to find out what pathogen is making you ill, following the successful demonstration of the world’s first multi-pathogen identification using non-amplified RNA detection by GeneCapture, a company cofounded by researchers at The University of Alabama in Huntsville (UAH).
Rutgers Experts Urge Ban of Menthol Cigarettes Nationwide
Rutgers experts discuss why actions at the state and federal level need to be taken to ban menthol-flavored tobacco products

Government health, safety regulations backfire with conservatives, study shows
A new study from the University of Notre Dame shows government-imposed restrictions can backfire, depending on political ideology.
NCCN Policy Summit Explores 21st Century Cures Act and the Impact of Health Information Technology
Online summit from NCCN on 21st Century Cures Act includes bipartisan sponsors, regulatory authorities; examines the impact on and from COVID-19 on cancer care and technology; presents policy recommendations for using data to advance patient access to high-quality cancer care
Food Safety and Coronavirus
Covid Conversations on Risk featuring Jade Mitchell, Ph.D., and Felicia Wu, Ph.D. both from Michigan State University addresses food safety and risk. A recording of the webinar can be found on the SRA website at https://sra.org/covid-19-resources
New Legislation Would Jeopardize Patient Access to Medical Tests Across the Board by Restricting Policy that Removed Barriers to Coronavirus Testing
On March 5, U.S. House and Senate lawmakers introduced the VALID Act, which would give the Food and Drug Administration (FDA) new, expansive powers to regulate laboratory developed tests—tests that are already regulated by the Centers for Medicare and Medicaid Services (CMS) and are subject to stringent personnel, quality control, and proficiency testing requirements. This bill promotes duplicative, costly federal regulations for clinical laboratories that will result in decreased patient access to essential medical tests. AACC urges Congress not to act on this bill until its impact on healthcare can be thoroughly evaluated.
AACC Statement on New FDA Guidance That Allows Certified Labs to Perform Coronavirus Testing
AACC thanks the FDA for being responsive to the concerns of the clinical laboratory community and amending the coronavirus guidance to allow CMS-certified labs to develop and implement new tests for coronavirus prior to FDA approval.
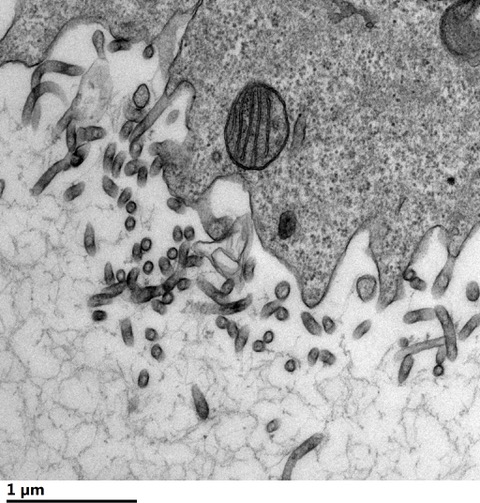
Researchers Show How Ebola Virus Hijacks Host Lipids
Robert Stahelin studies some of the world’s deadliest viruses. Filoviruses, including Ebola virus and Marburg virus, cause viral hemorrhagic fever with high fatality rates. Stahelin, professor at Purdue University, examines how these viruses take advantage of human host cells.

ISPOR-FDA Summit 2020 Will Focus on the Use of Patient-Preference Information in Medical Devices
ISPOR—the professional society for health economics and outcomes research, announced that it will be holding a joint ISPOR-FDA Summit, “Using Patient-Preference Information in Medical Device Regulatory Decisions: Benefit-Risk and Beyond,” on March 31, 2020 in Silver Spring, MD, USA.
ASTRO applauds Senate confirmation of Dr. Stephen Hahn as U.S. Food and Drug Administration Commissioner
The American Society for Radiation Oncology (ASTRO) applauded the U.S. Senate for voting today to confirm radiation oncologist Stephen Hahn, MD, FASTRO, as the next Commissioner of the Food and Drug Administration (FDA).
FDA awards $1.7 million to IU and other universities for quality management research
The U.S. Food and Drug Administration has awarded $1.7 million to five universities, including Indiana University, for a wide-ranging research project designed to help the agency better understand how to employ its quality management resources when inspecting drug companies’ manufacturing operations, thus benefiting consumers.

The E-cigarette Backlash
CFR In Brief by Claire Felter. An outbreak of a lung illness linked to vaping is raising the pressure on countries to rein in the booming e-cigarette industry.