Scientists from Trinity College Dublin have unearthed a secret that may explain why some people are able to resist viral infections, having screened the immune systems of women exposed to hepatitis C (HCV) through contaminated anti-D transfusions given over 40 years ago in Ireland.
Tag: Hepatitis C
Mount Sinai’s Yvette Calderon, MD, MS, Elected to National Academy of Medicine for Contributions to Emergency Medicine
Yvette Calderon, MD, MS, Chair of Emergency Medicine at Mount Sinai Beth Israel and Professor of Emergency Medicine at the Icahn School of Medicine at Mount Sinai, has been elected to the National Academy of Medicine (NAM). Election to the NAM is considered one of the highest honors in health and medicine, recognizing individuals who have demonstrated outstanding professional achievement and commitment to service. With her election, Mount Sinai has 26 faculty members in the NAM.
New Low-cost Device Rapidly, Accurately Detects Hepatitis C Infection
The entire virus detection process is executed inside a uniquely designed, portable, inexpensive, disposable, and self-driven microfluidic chip. The fully automated sample-in–answer-out molecular diagnostic set-up rapidly detects Hepatitis C virus in about 45 minutes and uses relatively inexpensive and reusable equipment costing about $50 for sample processing and disease detection. The disposable microfluidic chip also offers shorter times for a reliable diagnosis and costs about $2.
Access to hep C treatment reducing morbidity and mortality among people who inject drugs
A longitudinal cohort study of persons with a history of injection drug use has found that more people who inject drugs (PWID) are receiving Hepatitis C virus (HCV) treatment, which is associated with significant reductions in liver disease and mortality.
COVID-19 research campaign moves from basic science to antiviral drug design
ORNL researchers have developed and tested novel small-molecule antivirals in an effort to design new drugs to treat COVID-19. The so called hybrid inhibitor molecules are made from repurposed drugs used to treat hepatitis C and the original coronavirus outbreak in the early 2000s. The experimental research results show the molecules are similarly as effective as some of the leading drugs on the market today.
Hepatitis C Infections Among Pregnant People Increased Substantially Between 2009 and 2019
The leading cause of HCV in the U.S. is injection drug use as a result of opioid use disorder (OUD), which has seen a rise in most populations, including pregnant people, in recent years. HCV rates have also risen. Between 2009 and 2019, the overall rate per 1,000 live births of HCV in pregnant people increased from 1.8 to 5.1.
August Issue of Issue of The American Journal of Gastroenterology Includes Diet-Associated NAFLD Risk and Increased Risk of Mortality from COVID-19 Among PPI Users
The August issue of The American Journal of Gastroenterology includes clinical discussions of diet-associated NAFLD risk and increased risk of mortality from COVID-19 among PPI users. In addition, this issue features clinical research and reviews on IBS, gender barriers for CRC screening, hepatitis C, eosinophilic esophagitis, and more.
Hepatitis C Drugs Combined with Remdesivir Show Strong Effectiveness Against COVID-19
A combination of remdesivir, a drug currently approved in the United States for treating COVID-19 patients, and repurposed drugs for hepatitis C virus (HCV) was 10 times more effective at inhibiting SARS-CoV-2, the virus that causes COVID-19.
Hepatitis C Drugs Multiply Effect of COVID-19 Antiviral Remdesivir
When combined with drugs currently used to treat hepatitis C, the antiviral remdesivir is 10 times more effective in treating cells infected with SARS-CoV-2, the virus that causes COVID-19.
ACTG TO PRESENT 24 NEW STUDIES AT CROI 2021
The AIDS Clinical Trials Group (ACTG), the largest global HIV research network, will present four oral and 20 scientific spotlight sessions at the Conference on Retroviruses and Opportunistic Infections (CROI 2021) held virtually, March 6-10.
ACTG Presents Data Showing Minimal Monitoring Approach to Hepatitis C Treatment is Safe and Successful at AASLD’s 2020 Liver Meeting
Study finds that in a diverse, global patient population, a minimal monitoring (MINMON) approach to hepatitis C treatment was safe and achieved comparable sustained virologic response (SVR) to current standard of care.
More Women Diagnosed with HCV During Pregnancy, but Many Infants Still Not Tested Despite Recommendations from Leading Health Organizations
Data from a new study presented this week at The Liver Meeting Digital Experience® – held by the American Association for the Study of Liver Diseases (AASLD)– found that among pregnant women with hepatitis C virus (HCV), more than 25 percent were initially diagnosed during pregnancy screenings, which supports prenatal care as an important opportunity to screen for HCV in women. However, the study also found that less than one third of infants receive appropriate HCV testing, a significant care gap.
Screening for Hepatitis C: Not Just for Baby Boomers
While screening guidelines for hepatitis C previously targeted baby boomers, experts say more universal testing guidelines should be followed for early detection and better outcomes.

Trial Finds Drug Safe and Effective in Treating Hep-C During Pregnancy
Critics of the universal hepatitis C screening argue that it’s wasteful to test pregnant women for a disease that can’t be immediately treated, but results of a small phase I clinical trial suggest otherwise: pregnancy could be an excellent time to diagnose and cure hepatitis C infection.
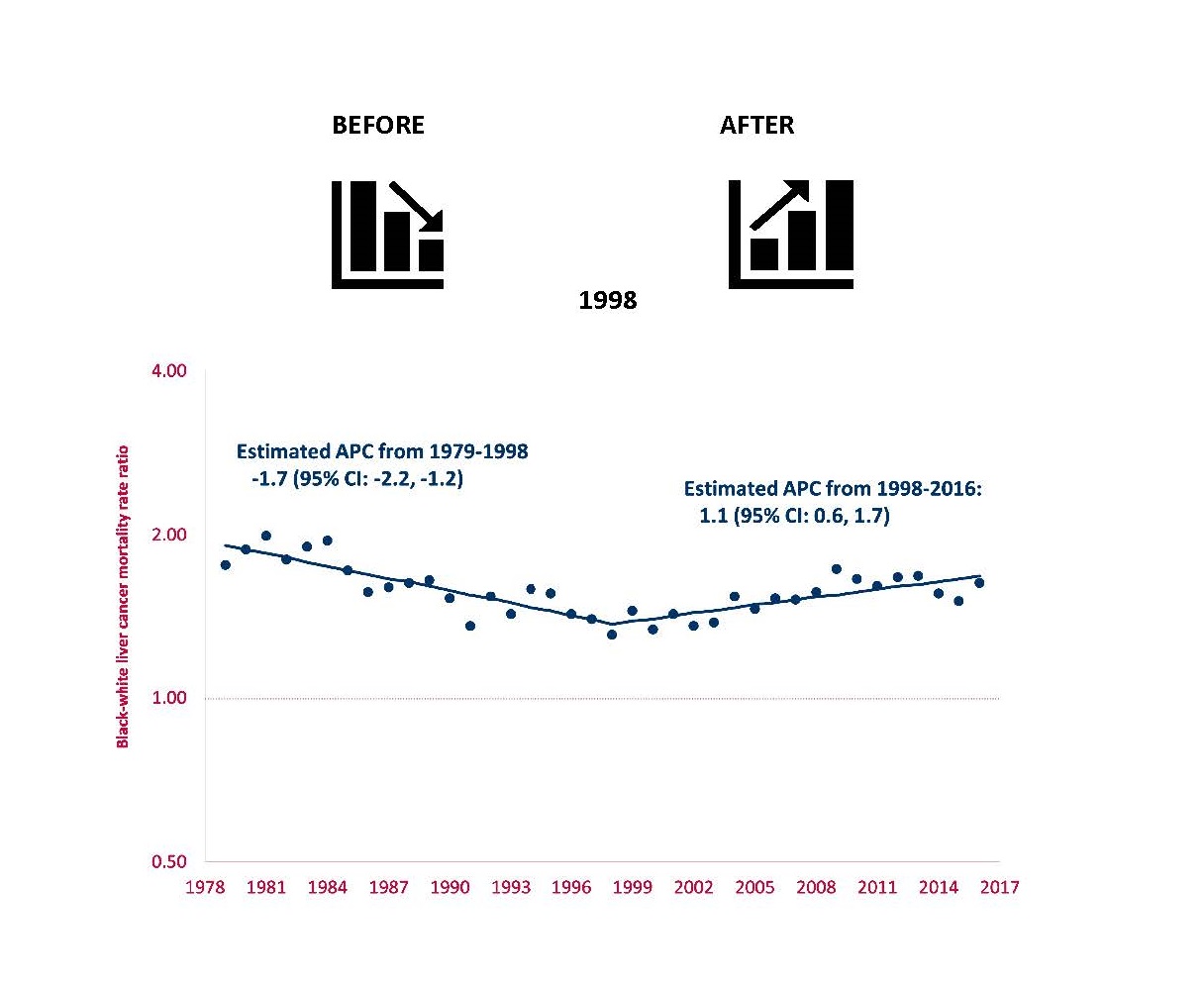
Racial Inequalities in Liver Cancer Deaths Soared After Launch of Hepatitis C Drugs
A study explored racial inequalities in death from liver cancer before and after the introduction of lifesaving drugs for hepatitis C. Results showed that from 1979 to 1998, racial inequalities in mortality from liver cancer in the U.S. were declining. But, from 1998 to 2016, of the 16,770 deaths from liver cancer among blacks, the excess relative to whites increased from 27.8 percent to 45.4 percent. Concurrently, racial inequalities in death decreased for major risk factors for liver cancer, such as alcohol and diabetes.
NOT JUST FOR BABY BOOMERS: Physicians Urge Hepatitis C Screening for Adults of All Ages
A risk factor for liver disease, cirrhosis and liver cancer, hepatitis C virus (HCV) infection is linked to the deaths of more U.S. patients than the next 60 infectious disease combined – including HIV and tuberculosis. Worryingly, its incidence is…
House Drug Pricing Bill Serves Patients, Public Health
H.R. 3, the Elijah E. Cummings Lower Drug Costs Now Act passed by the House of Representatives today introduces critically needed and significant steps to reduce costs and improve access to life-saving therapies for conditions including HIV and hepatitis C. Importantly, the legislation also brings essential resources to combat antibiotic resistance, find and develop new infection fighting drugs and bring them to market. The balanced approach of this legislation will serve patients and public health.
HCV Guidance Updates Recommendations for Identification and Management of Chronic Hep C
HCVguidelines.org — a website developed by the American Association for the Study of Liver Diseases and the Infectious Diseases Society of America to provide up-to-date guidance on the management of hepatitis C — was recently revised to reflect important developments in the identification and management of chronic hepatitis C (HCV)
Injection drug use: not the same across Canada
A new study by researchers at the University of Montreal shows close to 172,000 Canadians injected drugs in 2016, up from 130,000 just five years earlier, but support varies.

All-Oral Direct-Acting Antiviral Treatments Improve Survival in Patients with HCV-Related Hepatocellular Carcinoma
Data from a new study presented this week at The Liver Meeting® – held by the American Association for the Study of Liver Diseases – found that hepatitis C virus-related hepatocellular carcinoma (HCC) patients who achieved sustained virologic response (SVR) – denoting an undetectable level of HCV virus – with any oral direct-acting antiviral (DAA) had over 60-70 percent improvement in five-year survival.