Abstract The rising demand for Li, paramount for energy storage, necessitates expanded supply. As the supply is concentrated in a few countries, this poses supply chain risks for Li-ion battery makers. To diversify suppliers, alternative Li ore deposits such as…
Tag: Energy Storage
Biological Science Helps Fuel the Future of Electric Air Travel
In a new study, a team of researchers led by Berkeley Lab used a bioscience technique to study the intricate interactions within the anode, cathode, and electrolyte of electric aircraft batteries. One of the most significant findings was the discovery that certain salts mixed into the battery electrolyte formed a protective coating on cathode particles, making them far more resistant to corrosion, thereby enhancing battery life.
ARPA-E IGNIITE award launches new initiatives of ORNL researchers Yang and Westover
Guang Yang and Andrew Westover of Oak Ridge National Laboratory have been selected to join the first cohort of the Department of Energy’s Advanced Research Projects Agency-Energy, or ARPA-E, Inspiring Generations of New Innovators to Impact Technologies in Energy 2024, or IGNIITE 2024, program.
National Inventor of the Year Honor Goes to PNNL Energy Storage Expert
Ji-Guang (Jason) Zhang has been awarded the inaugural Department of Energy Lab Inventor of the Year Award, recognizing his contributions to research and his efforts to bring the benefit of new technologies to society as quickly as possible.
Exergoeconomic assessment of a high-efficiency compressed air energy storage system
Abstract Energy storage systems have a critical part in enabling greater use of intermittent energy resources. For a sustainable energy supply mix, compressed air energy storage systems offer several advantages through the integration of practical and flexible types of equipment…
Successful Workshop for Leading the ‘Carnot Battery’, the Future of Energy Storage
On May 17th (Friday), the Korea Institute of Energy Research (KIER) held a workshop at its main headquarters in Daejeon with experts from industry, academia, research, and government to discuss the technology, policy status, and development direction of the Carnot Battery in relation to energy storage and heat pumps.
Join Us at the International Conference on Sustainable Energy and Green Technology 2024 (SEGT 2024)
The Faculty of Engineering, Chulalongkorn University cordially invites you to attend the International Conference on Sustainable Energy and Green Technology 2024 (SEGT 2024). This year’s theme, ” Sustaining the Future with Green Energy and Clean Environmental Technology,” highlights our commitment to fostering sustainable solutions. The conference will be held in Bangkok, Thailand, from December 15-18, 2024.
KITECH Develops Deformable Energy Storage Device via Laser Technology
The joint research team, led by Dr. Chanwoo Yang and Researcher Seong Ju Park from Korea Institute of Industrial Technology(KITECH), along with Prof. Jin Kon Kim and Dr. Keon-Woo Kim from POSTECH, has successfully developed a compact energy storage device with excellent elasticity.
Groundbreaking Microcapacitors Could Power Chips of the Future
Berkeley Lab scientists have achieved record-high energy and power densities in microcapacitors made with engineered thin films, using materials and fabrication techniques already widespread in chip manufacturing.
Novel material supercharges innovation in electrostatic energy storage
Artificial heterostructures made of freestanding 2D and 3D membranes developed by WashU’s Sang-Hoon Bae have an energy density up to 19 times higher than commercially available capacitors.
Powering Up to Solve Challenges in Energy Storage
In 2006, battery research was practically non-existent at PNNL. Today, the lab is lauded for its battery research. How did PNNL go from a new player to a leader in state-of-the-art storage for EVs and the grid?

Microdroplets, macro results: Beckman researchers pursue Energy Earthshots
Researchers at the Beckman Institute will conduct electrochemical reactions in microdroplets to produce clean hydrogen, sequester carbon dioxide, and store renewable energies like wind and solar inexpensively and sustainably. Their project, called DROPLETS, received $4.5 million from the U.S. Department of Energy’s Office of Science through its Energy Earthshots Initiative.

New energy-storing material could also be used to build electronic gadgets
Gadgets and vehicles powered by the very materials they’re built from may soon be possible, thanks to a new structural supercapacitor developed by UC San Diego engineers. The device doubles as structural support and energy storage, potentially adding more energy capacity without adding weight.
ORNL to lead new center to create sustainable chemical industry processes
The Department of Energy’s Oak Ridge National Laboratory has been selected to lead an Energy Earthshot Research Center, or EERC, focused on developing chemical processes that use sustainable methods instead of burning fossil fuels to radically reduce industrial greenhouse gas emissions to stem climate change and limit the crisis of a rapidly warming planet.
Lawrence Livermore National Laboratory, California State University, Bakersfield and Livermore Lab Foundation sign MOU to advance clean energy
Lawrence Livermore National Laboratory (LLNL), California State University, Bakersfield (CSUB) and the Livermore Lab Foundation (LLF) have signed an agreement to collaborate on advanced and clean-energy technologies, research opportunities and community partnerships that have the potential to shape the future of energy in the state and bring high-quality jobs to the region.
Improving the properties of sweeteners for enhanced thermal energy storage
As we seek more efficient utilization of waste thermal energy, use of “phase change materials (PCMs)” is a good option. PCMs have a large latent heat capacity and the ability to store-and-release heat as they change from one state of matter to another.
Seven entrepreneurs join Innovation Crossroads seventh cohort
Seven entrepreneurs will embark on a two-year fellowship as the seventh cohort of Innovation Crossroads kicks off this month at the Department of Energy’s Oak Ridge National Laboratory. Representing a range of transformative energy technologies, Cohort 7 is a diverse class of innovators with promising new companies.
Harnessing the power of water: Argonne and NREL study shows the potential of pumped storage hydropower in Alaska
Scientists study the role of pumped storage hydropower in Alaska’s clean energy future.

Researchers Achieve Historic Milestone in Energy Capacity of Supercapacitors
In a new landmark chemistry study, researchers describe how they have achieved the highest level of energy storage — also known as capacitance — in a supercapacitor ever recorded.
Machine learning takes materials modeling into new era
Researchers have now pioneered a machine learning-based simulation method that supersedes traditional electronic structure simulation techniques. Their Materials Learning Algorithms (MALA) software stack enables access to previously unattainable length scales.
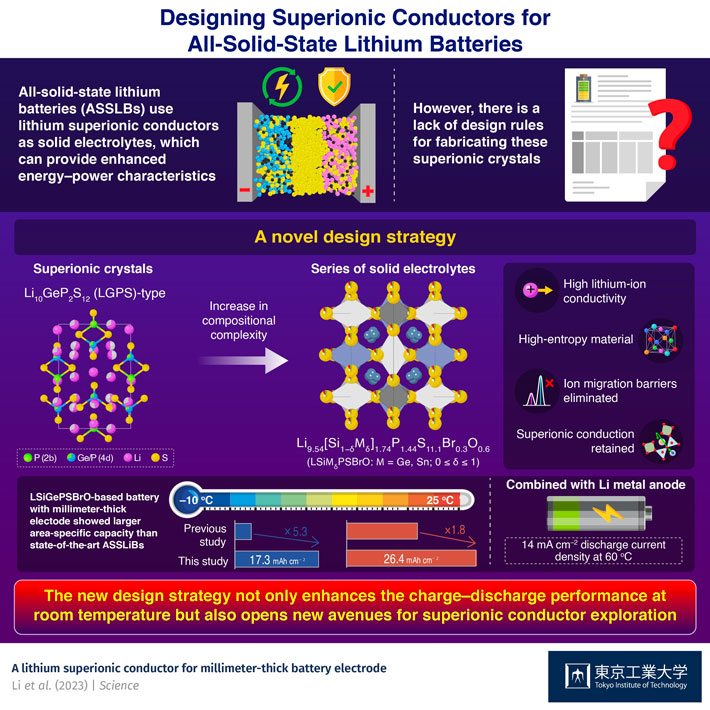
New design rule for high-entropy superionic solid-state conductors
Solid electrolytes with high lithium-ion conductivity can be designed for millimeter-thick battery electrodes by increasing the complexity of their composite superionic crystals, report researchers from Tokyo Tech.
Recycling of valuable metals from spent lithium ion batteries using spinning reactors
In a world that is slowly distancing itself from carbon-based energy, there has been a meteoric rise in the use of lithium-ion batteries as a next-generation energy storage solution.
Andrew Ullman, Wigner Fellow, gets a charge out of batteries
As a Distinguished Staff Fellow in the Chemical Sciences Division focused on energy storage and conversion, Andrew Ullman of Oak Ridge National Laboratory is using chemistry to devise a better battery.
Underground Water Could be the Solution to Green Heating and Cooling
About 12% of the total global energy demand comes from heating and cooling homes and businesses. A new study suggests that using underground water to maintain comfortable temperatures could reduce consumption of natural gas and electricity in this sector by 40% in the U.S. The approach, called aquifer thermal energy storage (ATES), could also help prevent blackouts caused by high power demand during extreme weather events.

3D battery imaging reveals the secret real-time life of lithium metal cells
Innovative battery researchers have cracked the code to creating real-time 3D images of the promising but temperamental lithium metal battery as it cycles.
On the Road to Better Solid-State Batteries
A team from Berkeley Lab and Florida State University has designed a new blueprint for solid-state batteries that are less dependent on specific chemical elements. Their work could advance efficient, affordable solid-state batteries for electric cars.
New compound that withstands extreme heat and electricity could lead to next-generation energy storage devices
Society’s growing demand for high-voltage electrical technologies—including pulsed power systems, cars and electrified aircraft, and renewable energy applications—requires a new generation of capacitors that store and deliver large amounts of energy under intense thermal and electrical conditions. Researchers at Lawrence Berkeley National Laboratory and Scripps Research have now developed a new polymer-based device that efficiently handles record amounts of energy while withstanding extreme temperatures and electric fields.
Department of Energy Announces $125 Million for Research to Enable Next-Generation Batteries and Energy Storage
Today, the U.S. Department of Energy (DOE) announced $125 million for basic research on rechargeable batteries to provide foundational knowledge needed to transform and decarbonize our energy system through the development and adoption of cost-effective and clean energy sources. The national, economic, and environmental security challenges will not be met solely by incremental improvements to existing clean energy technologies but instead will require transformational technologies founded on new fundamental knowledge and capabilities developed through basic scientific research.
Turning abandoned mines into batteries
A novel technique called Underground Gravity Energy Storage turns decommissioned mines into long-term energy storage solutions, thereby supporting the sustainable energy transition.
Israel Ministry of Energy selects Bar-Ilan University and the Technion to establish $36.8 million national research institute in energy storage
Bar-Ilan University and the Technion – Israel Institute of Technology have won a call published by the Israel Ministry of Energy for the establishment of a national research institute in the field of energy storage.
Taking Freight Trucks Electric Would Have Big Economic and Environmental Benefits for India
Diesel-fueled freight trucks play an outsized role in producing India’s total greenhouse gas and air pollution emissions. While the country has promoted policies to transition to electric vehicles for public transportation buses and cars, batteries that can power such large trucks have been too heavy and expensive to make their electrification possible. A new study shows that advances in battery technology and dramatically decreased battery costs in recent years have changed that. With the right policies and incentives, battery electric trucks would be more affordable to operate than diesel, and India could become a world leader in producing electric vehicles.
Designing Next-Generation Metals, One Atom at a Time
Direct visualization of metal atoms during shear deformation has broad applications from battery design to vehicle lightweighting.
Binghamton University-led battery initiative wins $113 million to bolster domestic battery manufacturing and supply chain, reinvigorate region
Binghamton University’s New Energy New York project has been awarded more than $113 million to establish a hub for battery technology innovation in upstate New York. The U.S. Economic Development Administration announced Friday that the region would receive $63.7 million; the State of New York will support the project with an additional $50 million.
DOE Announces $540 Million for Technologies to Transform Energy Production and Cut Emissions
The U.S. Department of Energy (DOE) today announced more than $540 million in awards for university- and National Laboratory-led research into clean energy technologies and low-carbon manufacturing. Most greenhouse-gas emissions come from the production and use of energy, so building strong scientific foundations for reducing emissions across the energy lifecycle is crucial to meeting President Biden’s goal of creating a net-zero emissions economy by 2050.
Washington State Academy of Sciences Adds Six PNNL Researchers
The Washington State Academy of Sciences added six people from PNNL to its 2022 class of inductees.
Department of Energy Awards 18 Million Node-Hours of Computing Time to Support Cutting-Edge Research
Today, the U.S. Department of Energy (DOE) announced that 18 million node-hours have been awarded to 45 scientific projects under the Advanced Scientific Computing Research (ASCR) Leadership Computing Challenge (ALCC) program. The projects, with applications ranging from advanced energy systems to climate change to cancer research, will use DOE supercomputers to uncover unique insights about scientific problems that would otherwise be impossible to solve using experimental approaches.
Story tips: Tailor-made molecules, better battery electrolytes, beyond Moore’s Law and improving climate model accuracy
ORNL Story tips: Tailor-made molecules, better battery electrolytes, beyond Moore’s Law and improving climate model accuracy
Energy, Maritime Leaders to Convene for DOE InnovationXLab Summit
Top scientists and officials from government, academia, Alaskan Native communities, and industry are heading to Alaska to focus on driving energy technologies for a more sustainable Arctic region.
Senator Cantwell, Science Leaders Help Break Ground on $75 Million Grid Storage Launchpad
New facility will accelerate energy storage innovation, increase clean energy adoption and grid resilience.
Fitbits and Other Devices Measure Energy Expended Well, But Less Reliable for Tracking Energy Storage and Intake in Research
IAFNS-supported study finds that commercial devices do well in estimating energy outlays compared to gold-standard measures but less well on storage and intake.
New scalable method resolves materials joining in solid-state batteries
Scientists at the Department of Energy’s Oak Ridge National Laboratory have developed a scalable, low-cost method to improve the joining of materials in solid-state batteries, resolving one of the big challenges in the commercial development of safe, long-lived energy storage systems.
Energy Secretary, Washington Governor Dedicate Energy Sciences Center at PNNL
Fundamental research conducted at the $90-million research facility will help the nation meet its clean energy goals.
Stretching the capacity of flexible energy storage (video)
Researchers in ACS’ Nano Letters report a flexible supercapacitor with electrodes made of wrinkled titanium carbide — a type of MXene nanomaterial — that maintained its ability to store and release electronic charges after repetitive stretching.
High-energy shape memory polymer could someday help robots flex their muscles
Researchers reporting in ACS Central Science have developed a shape memory polymer that stores almost six times more energy than previous versions.

Making Methane from CO2: Carbon Capture Grows More Affordable
PNNL researchers can make methane from captured CO2 and renewably sourced hydrogen, offering a path toward cheaper synthetic natural gas.
Bacteria may hold key for energy storage, biofuels
Cornell University bioengineer Buz Barstow is trying to solve a big problem: How to build a low-cost, environmentally friendly and large-scale system for storing and retrieving energy from renewable sources such as wind and solar.
LED Material Shines Under Strain
A team led by researchers at Lawrence Berkeley National Laboratory (Berkeley Lab) and UC Berkeley has demonstrated an approach for achieving LEDs with near 100% light-emission efficiency at all brightness levels.
Hydrogen Technologies Take Leading Role Toward Net Zero
Achieving Net Zero energy, where the total amount of energy used is equal to the amount of renewable energy created, is closer than ever before, and hydrogen technologies will play an important role in achieving that goal, but needs and gaps need to be addressed before a true hydrogen-powered future can take form. There are many opportunities in the global public and private sectors for research, development, and deployment collaboration.
Waste to Energy: Biofuel from Kelp Harvesting and Fish
Using existing fish processing plants, kelp and fish waste can be converted to a diesel-like fuel to power generators or fishing boats in remote, coastal Alaska.
MEDIA ADVISORY: AIP Publishing Hosts Expert Sessions on Energy Storage and Conversion at Virtual Conference
Twelve distinguished speakers will be covering critical topics impacting energy storage and conversion at the upcoming AIP Publishing Horizons Virtual Conference on Aug. 4-6. The three-day event is organized by the journal Applied Physics Reviews and brings together leaders in the field of energy science to present their latest research in six sessions