Utility-scale batteries can revolutionize how we harness renewable power. Coupled with wind and solar, these batteries could increase the reliability of green energy by storing excess energy during times of high generation and low demand. Then, utilities can tap the stored energy when demand increases.
Tag: Batteries
New open-access battery lab aims to boost U.S. manufacturing and workforce development for electric vehicles and beyond
Expansion of Washington Clean Energy Testbeds will enable fabrication of pouch cells to accelerate the next generation of faster-charging, higher-energy-density, and earth-friendly batteries.
A Quick and Easy Way to Produce Anode Materials for Sodium-Ion Batteries Using Microwaves
KERI succeeds in producing hard carbon anode material in 30 seconds through microwave induction heating. A solution for commercialization of a next-generation battery with reduced fire risk: results published in an internationally renowned journal.

Argonne’s ReCell Center hosts Industry Collaboration Meeting, highlighting battery recycling partnerships
Argonne’s ReCell Center hosted a two-day Industry Collaboration Meeting. It was attended by major energy storage and battery recycling stakeholders in the Chicagoland community.
AI-Guided Experiments Speed Scientific Discovery
Finding a needle in a haystack is the quintessentially impossible task. But what if new tools could make it straightforwardly achievable? Imagine if, instead of searching through everything by hand, you could portion out small piles of hay and use magnets.
Manganese Cathodes Could Boost Lithium-ion Batteries
Manganese is earth-abundant and cheap. A new process could help make it a contender to replace nickel and cobalt in batteries.
Korean Researchers Overcome Critical Challenges in Developing Fire-Risk-Free Aqueous Zinc Batteries
The operation performance of the battery and the stability of large-area deposition have been verified, bringing the technology closer to commercialization. Also, the research was published as the cover article in the August issue of the world-renowned journal in the field of energy and materials, Advanced Energy Materials.
Esther Takeuchi Honored in Special Festschrift Issue
Esther Sans Takeuchi, a materials scientist and chemical engineer at the U.S. Department of Energy’s (DOE) Brookhaven National Laboratory, was honored by the Journal of Physical Chemistry C in a special Festschrift issue earlier this year.
Innovation Crossroads welcomes seven entrepreneurs for Cohort 2024
Seven entrepreneurs comprise the next cohort of Innovation Crossroads, a Department of Energy Lab-Embedded Entrepreneurship Program node based at Oak Ridge National Laboratory. The program provides energy-related startup founders from across the nation with access to ORNL’s unique scientific resources and capabilities.
Batería de iones de litio totalmente elástica para dispositivos electrónicos flexibles
Los investigadores de ACS Energy Letters informan sobre una batería de iones de litio con componentes totalmente extensibles, incluida una capa de electrolito que puede expandirse un 5000 %, y que conserva su capacidad de almacenamiento de carga después de casi 70 ciclos de carga y descarga.
Completely stretchy lithium-ion battery for flexible electronics
Researchers in ACS Energy Letters report a lithium-ion battery with entirely stretchable components, including an electrolyte layer that can expand by 5000%, and it retains its charge storage capacity after nearly 70 charge/discharge cycles. Potential applications include flexible electronics.
Biological Science Helps Fuel the Future of Electric Air Travel
In a new study, a team of researchers led by Berkeley Lab used a bioscience technique to study the intricate interactions within the anode, cathode, and electrolyte of electric aircraft batteries. One of the most significant findings was the discovery that certain salts mixed into the battery electrolyte formed a protective coating on cathode particles, making them far more resistant to corrosion, thereby enhancing battery life.
ARPA-E IGNIITE award launches new initiatives of ORNL researchers Yang and Westover
Guang Yang and Andrew Westover of Oak Ridge National Laboratory have been selected to join the first cohort of the Department of Energy’s Advanced Research Projects Agency-Energy, or ARPA-E, Inspiring Generations of New Innovators to Impact Technologies in Energy 2024, or IGNIITE 2024, program.
Synergy of green energy technologies through critical materials circularity
Abstract Synergies between technology flows is essential to balance the consumption of their related critical materials and promote a sustainable green economy transition. Using dynamics modelling, a comprehensive analysis of silicon flows applied in green energy technologies such as photovoltaic…
Better battery manufacturing: Robotic lab vets new reaction design strategy
New chemistries for batteries, semiconductors and more could be easier to manufacture, thanks to a new approach to making chemically complex materials that researchers at the University of Michigan and Samsung’s Advanced Materials Lab have demonstrated.
Powering Up to Solve Challenges in Energy Storage
In 2006, battery research was practically non-existent at PNNL. Today, the lab is lauded for its battery research. How did PNNL go from a new player to a leader in state-of-the-art storage for EVs and the grid?

More than flying cars: eVTOL battery analysis reveals unique operating demands
Researchers at the Department of Energy’s Oak Ridge National Laboratory are taking cleaner transportation to the skies by creating and evaluating new batteries for airborne electric vehicles that take off and land vertically.

Healable Cathode Could Unlock Potential of Solid-state Lithium-sulfur Batteries
UC San Diego engineers developed a cathode material for lithium-sulfur (Li-S) batteries that is healable and highly conductive, overcoming longstanding challenges of traditional sulfur cathodes. The advance holds promise for bringing more energy dense and low-cost Li-S batteries closer to market.
KIST-LLNL raises expectations for commercialization of high-energy-density all-solid-state batteries
The Korea Institute of Science and Technology (KIST) announced that a KIST-LLNL joint research team led by Dr. Seungho Yu of the Energy Storage Research Center, Dr. Sang Soo Han of the Computational Science Research Center, and Dr. Brandon Wood of Lawrence Livermore National Laboratory (LLNL) has developed a fluorine substituted high-voltage stable chloride-based solid-state electrolyte through computational science.
Photo battery achieves competitive voltage
Networked intelligent devices and sensors can improve the energy efficiency of consumer products and buildings by monitoring their consumption in real time. Miniature devices like these being developed under the concept of the Internet of Things require energy sources that are as compact as possible in order to function autonomously.
New designs for solid-state electrolytes may soon revolutionize the battery industry
Researchers led by Professor KANG Kisuk of the Center for Nanoparticle Research within the Institute for Basic Science (IBS), have announced a major breakthrough in the field of next-generation solid-state batteries. It is believed that their new findings will enable the creation of batteries based on a novel chloride-based solid electrolyte that exhibits exceptional ionic conductivity.
Scientists illuminate the mechanics of solid-state batteries
A team led by researchers at the Department of Energy’s Oak Ridge National Laboratory developed a framework for designing solid-state batteries, or SSBs, with mechanics in mind. Their paper, published in Science, reviewed how these factors change SSBs during their cycling.
Thicker, denser, better: New electrodes may hold key to advanced batteries
To improve battery performance and production, Penn State researchers and collaborators have developed a new fabrication approach that could make for more efficient batteries that maintain energy and power levels.
Next-generation Flow Battery Design Sets Records
A new flow battery design achieves long life and capacity for grid energy storage from renewable fuels.
UC Irvine scientists create long-lasting, cobalt-free, lithium-ion batteries
Irvine, Calif., June 14, 2023 – In a discovery that could reduce or even eliminate the use of cobalt – which is often mined using child labor – in the batteries that power electric cars and other products, scientists at the University of California, Irvine have developed a long-lasting alternative made with nickel. “Nickel doesn’t have child labor issues,” said Huolin Xin, the UCI professor of physics & astronomy whose team devised the method, which could usher in a new, less controversial generation of lithium-ion batteries.
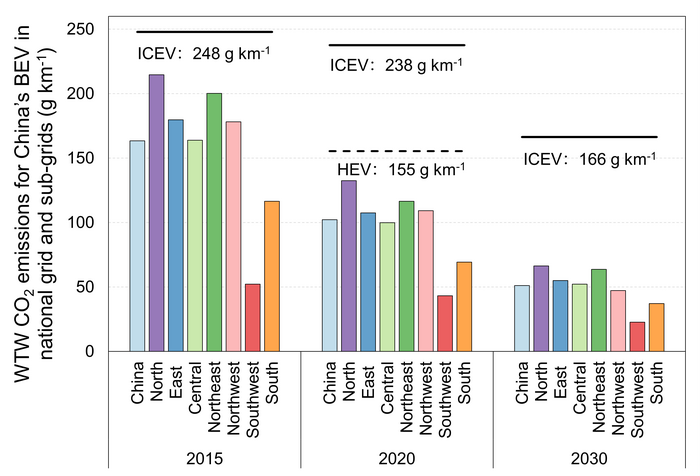
Emissions reductions of Chinese EVs
Chinese electric vehicles (EVs) drive larger emissions reductions over time, due to increased operating efficiency and a greener electricity mix, according to a study.
Andrew Ullman, Wigner Fellow, gets a charge out of batteries
As a Distinguished Staff Fellow in the Chemical Sciences Division focused on energy storage and conversion, Andrew Ullman of Oak Ridge National Laboratory is using chemistry to devise a better battery.
Empa Entrepreneur Fellowships awarded
Two young Empa scientists each receive an Empa Entrepreneur Fellowship to develop innovative products based on their research. Abdessalem Aribia is developing environmentally friendly and safe batteries, while Subas Scheibler is working on nanoparticles for cancer therapy.

Answering Big Questions with Thin Oxide Films
Thin oxide films play an important role in electronics and energy storage. Researchers in PNNL’s film growth laboratory create, explore, and improve new thin oxide films.

UAH researcher seeks to explain why lithium-ion batteries abruptly fail; earns $598K NSF CAREER Award
Research focused on why and how lithium-ion batteries may suddenly fail energetically, causing smoke, fire or even an explosion, a phenomenon called thermal runaway, has earned a researcher at The University of Alabama in Huntsville (UAH) a National Science Foundation (NSF) CAREER Award totaling $598,181.
New Sodium, Aluminum Battery Aims to Integrate Renewables for Grid Resiliency
A new sodium battery technology shows promise for helping integrate renewable energy into the electric grid. The battery uses Earth-abundant raw materials such as aluminum and sodium.

Brookhaven Lab Battery Scientist, Hydrogeologist, and DOE Site Office Manager Among Secretary of Energy’s 2022 Honorees
U.S. Secretary of Energy Jennifer Granholm honored 44 teams with the Secretary of Energy Achievement Award and five individuals for their work. Among the recipients are Distinguished Professor Esther Takeuchi, a battery researcher with a joint appointment at the Department of Energy’s (DOE) Brookhaven National Laboratory and Stony Brook University; Douglas Paquette, a hydrogeologist in Brookhaven Lab’s Environmental Protection Division; and Robert Gordon, manager of the DOE-Brookhaven Site Office that oversees operations at Brookhaven Lab.
UCI researchers decipher atomic-scale imperfections in lithium-ion batteries
As lithium-ion batteries have become a ubiquitous part of our lives through their use in consumer electronics, automobiles and electricity storage facilities, researchers have been working to improve their power, efficiency and longevity. As detailed in a paper published today in Nature Materials, scientists at the University of California, Irvine and Brookhaven National Laboratory conducted a detailed examination of high-nickel-content layered cathodes, considered to be components of promise in next-generation batteries.
Researchers Create Smaller, Cheaper Flow Batteries for Clean Energy
Flow batteries offer a solution. Electrolytes flow through electrochemical cells from storage tanks in this rechargeable battery. The existing flow battery technologies cost more than $200/kilowatt hour and are too expensive for practical application, but Liu’s lab in the School of Chemical and Biomolecular Engineering (ChBE) developed a more compact flow battery cell configuration that reduces the size of the cell by 75%, and correspondingly reduces the size and cost of the entire flow battery. The work could revolutionize how everything from major commercial buildings to residential homes are powered.
Turning abandoned mines into batteries
A novel technique called Underground Gravity Energy Storage turns decommissioned mines into long-term energy storage solutions, thereby supporting the sustainable energy transition.
WashU Expert: 2023 will be the year of the battery
Major advances in battery technologies will bring us a big step closer this year to large-scale renewable energy goals, international energy independence and a big reduction in greenhouse gases, according to an expert from Washington University in St. Louis. “One of the major challenges to a fully renewable-energy future of wind and solar power is energy storage,” said Michael Wysession, a professor of earth and planetary sciences in Arts & Sciences at Washington University in St.
Idaho technology changing outlook of advanced materials development
On their way to market, technologies often reach what is called the “valley of death,” the point where a researcher or institution has developed a promising idea, has received funding through grants, and then runs out of cash to move the idea beyond the laboratory.
Capsule-sized ingestible biobatteries could allow new view of digestive system
A new biobattery being developed at Binghamton University, State University of New York could power ingestible cameras in the small intestine.

X-rays Reveal Elusive Chemistry for Better EV Batteries
A team of scientists led by chemists at the U.S. Department of Energy’s Brookhaven National Laboratory and Pacific Northwest National Laboratory has unraveled the complex chemical mechanisms of a battery component that is crucial for boosting energy density: the interphase.
Nanoengineers Develop a Predictive Database for Materials
Nanoengineers at the University of California San Diego’s Jacobs School of Engineering have developed an AI algorithm that predicts the structure and dynamic properties of any material—whether existing or new—almost instantaneously. Known as M3GNet, the algorithm was used to develop matterverse.ai, a database of more than 31 million yet-to-be-synthesized materials with properties predicted by machine learning algorithms. Matterverse.ai facilitates the discovery of new technological materials with exceptional properties.
Liquid-to-solid battery electrolyte technology licensed exclusively to Safire
The Department of Energy’s Oak Ridge National Laboratory has exclusively licensed battery electrolyte technology to Safire Technology Group. The collection of five patented technologies are designed for a drop-in additive for lithium-ion batteries that prevents explosions and fire from impact.
Designing Next-Generation Metals, One Atom at a Time
Direct visualization of metal atoms during shear deformation has broad applications from battery design to vehicle lightweighting.

UCI and national lab researchers develop a cobalt-free cathode for lithium-ion batteries
Irvine, Calif., Sept. 21, 2022 – Researchers at the University of California, Irvine and four national laboratories have devised a way to make lithium-ion battery cathodes without using cobalt, a mineral plagued by price volatility and geopolitical complications. In a paper published today in Nature, the scientists describe how they overcame thermal and chemical-mechanical instabilities of cathodes composed substantially of nickel – a common substitute for cobalt – by mixing in several other metallic elements.
Binghamton University-led battery initiative wins $113 million to bolster domestic battery manufacturing and supply chain, reinvigorate region
Binghamton University’s New Energy New York project has been awarded more than $113 million to establish a hub for battery technology innovation in upstate New York. The U.S. Economic Development Administration announced Friday that the region would receive $63.7 million; the State of New York will support the project with an additional $50 million.
Media Briefing Schedule for ACS Fall 2022
Media Briefing Schedule for ACS Fall 2022
Super-fast electric car charging, with a tailor-made touch
Speeding up electrical vehicle charging can damage the battery. Now, scientists report that they’ve designed superfast charging methods tailored to power different electric vehicle batteries in 10 minutes or less without harm. The researchers will present their results today at ACS Fall 2022.
Longer Lasting Sodium-Ion Batteries on the Horizon
A new longer-lasting sodium-ion battery design is much more durable and reliable in lab tests. After 300 charging cycles, it retained 90 percent of its charging capacity.
These energy-packed batteries work well in extreme cold and heat
Researchers developed lithium-ion batteries that perform well at freezing cold and scorching hot temperatures, while packing a lot of energy. This could help electric cars travel farther on a single charge in the cold and reduce the need for cooling systems for the cars’ batteries in hot climates.
New biobatteries use bacterial interactions to generate power for weeks
Researchers at Binghamton University, State University of New York have developed a “plug-and-play” biobattery that lasts for weeks at a time and can be stacked to improve output voltage and current.
Lithium-ion batteries that last longer in extreme cold
To improve lithium-ion batteries’ performance in extreme cold, researchers reporting in ACS Central Science replaced the traditional graphite anode with a bumpy carbon-based material, which maintains its rechargeable storage capacity down to -31 F.