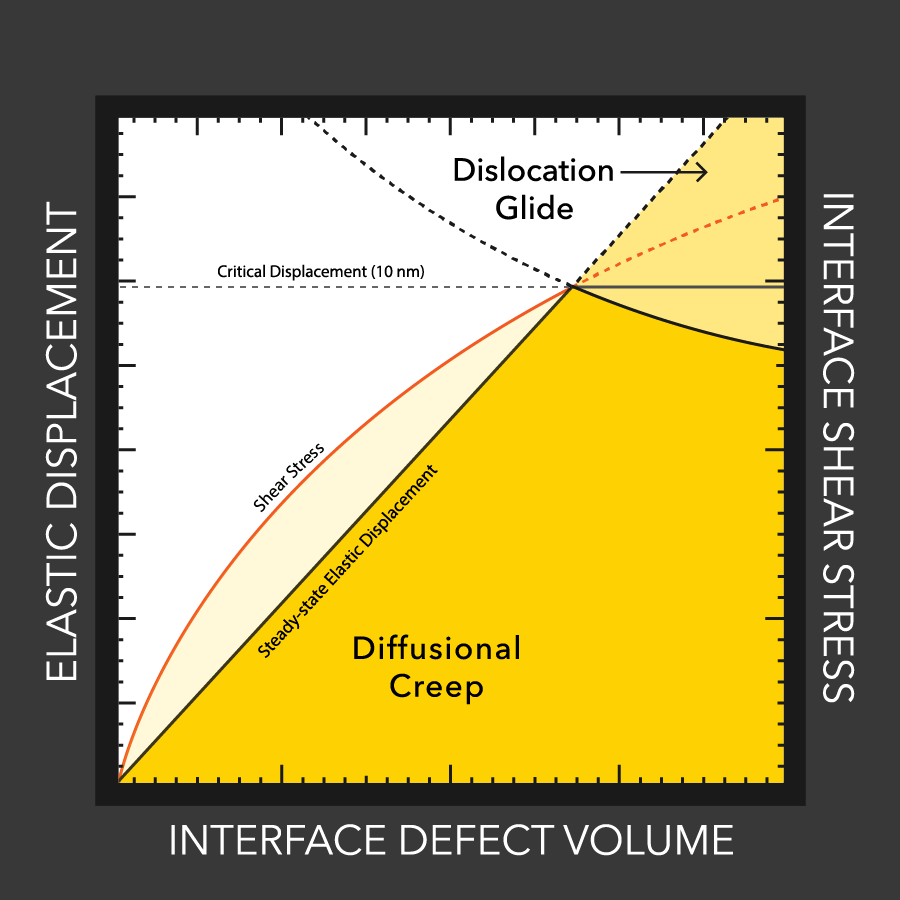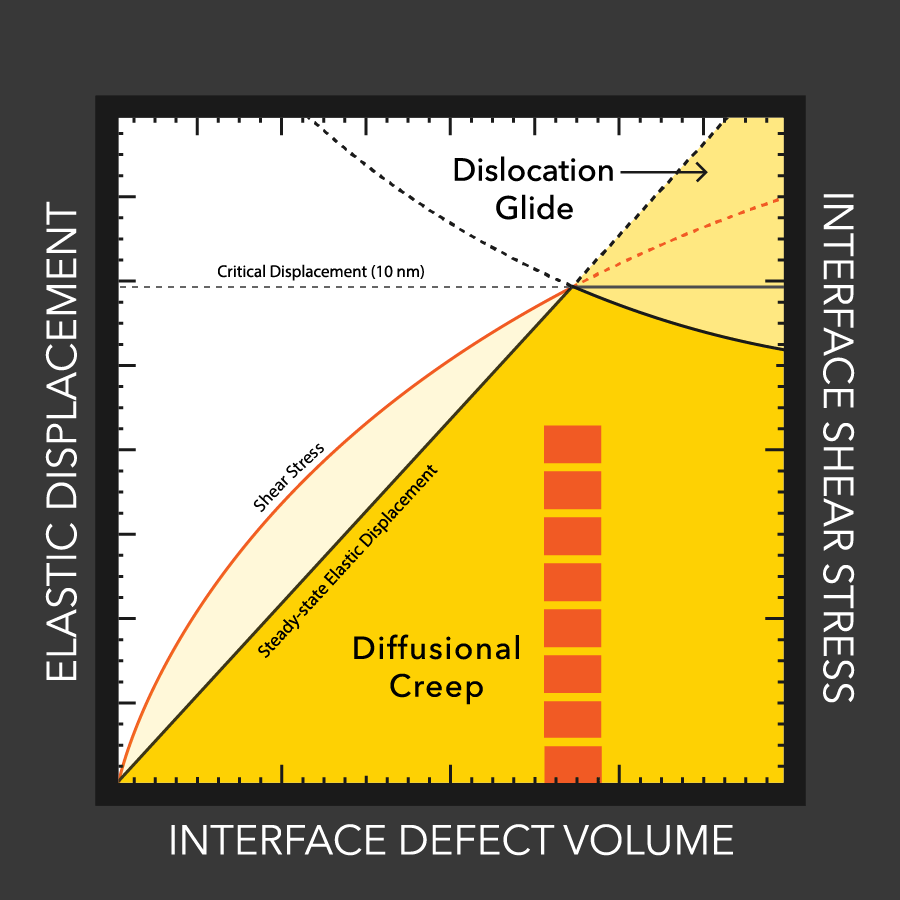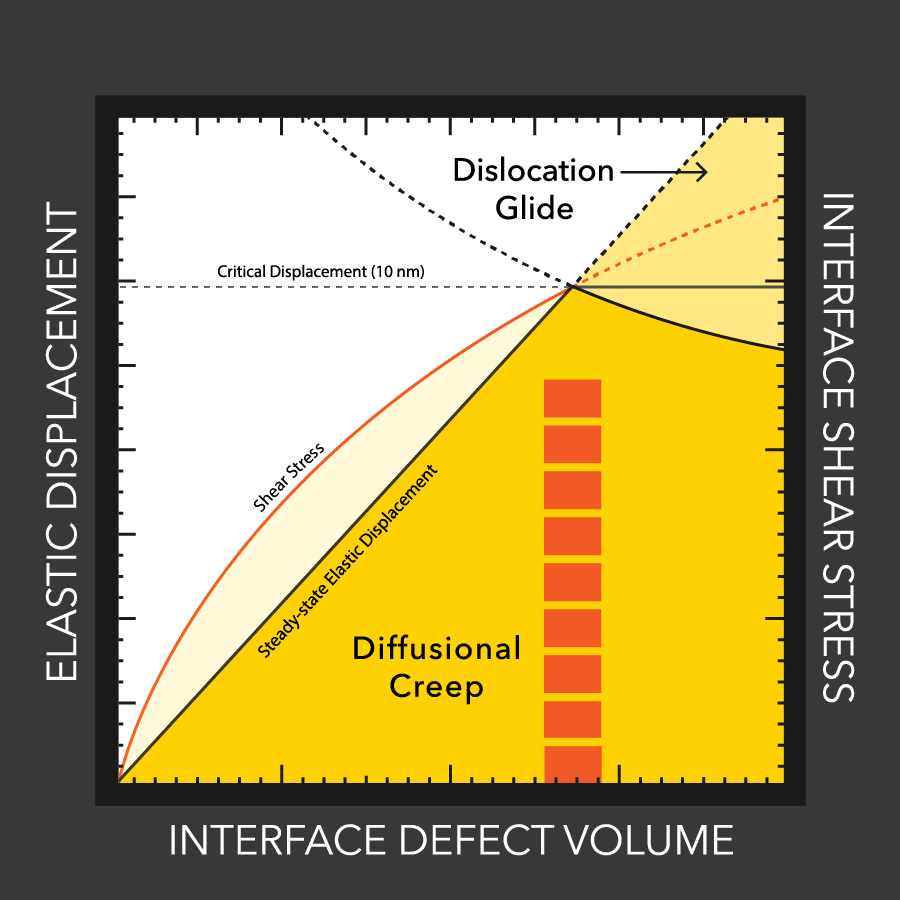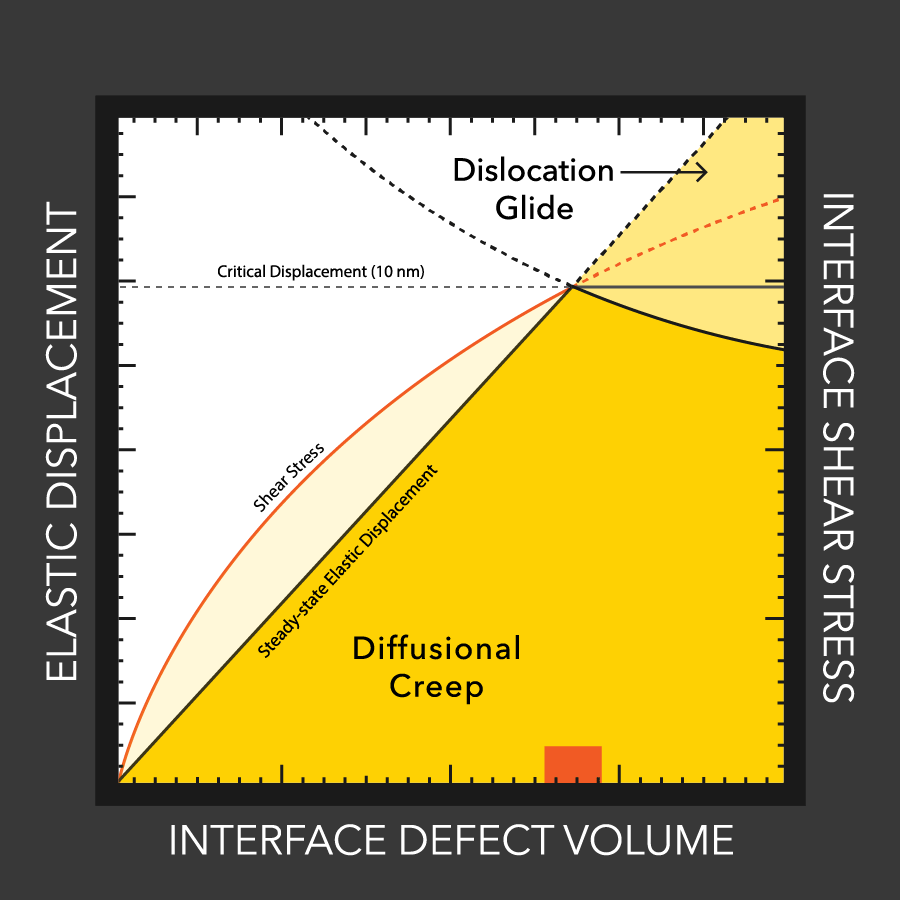
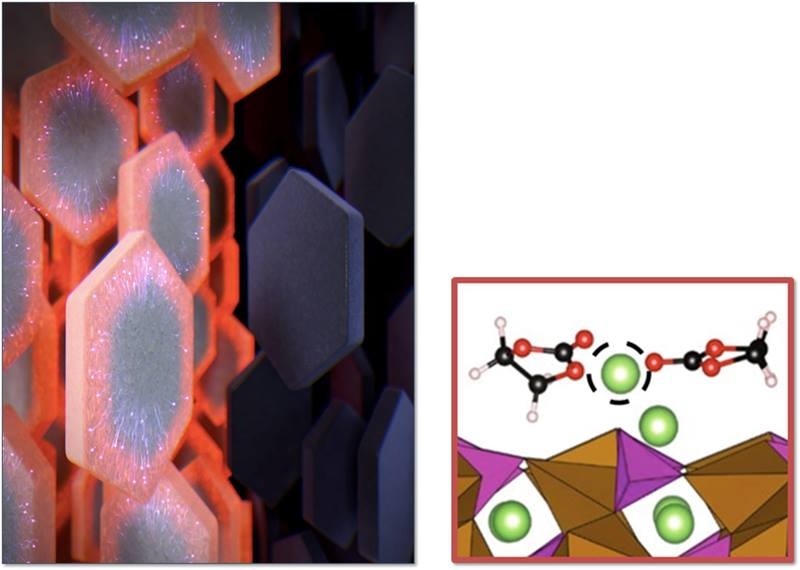
Using electronic devices for too long can cause them to overheat. Now, researchers reporting in ACS’ Nano Letters have developed a hydrogel that can both cool down electronics, such as cell phone batteries, and convert their waste heat into electricity.