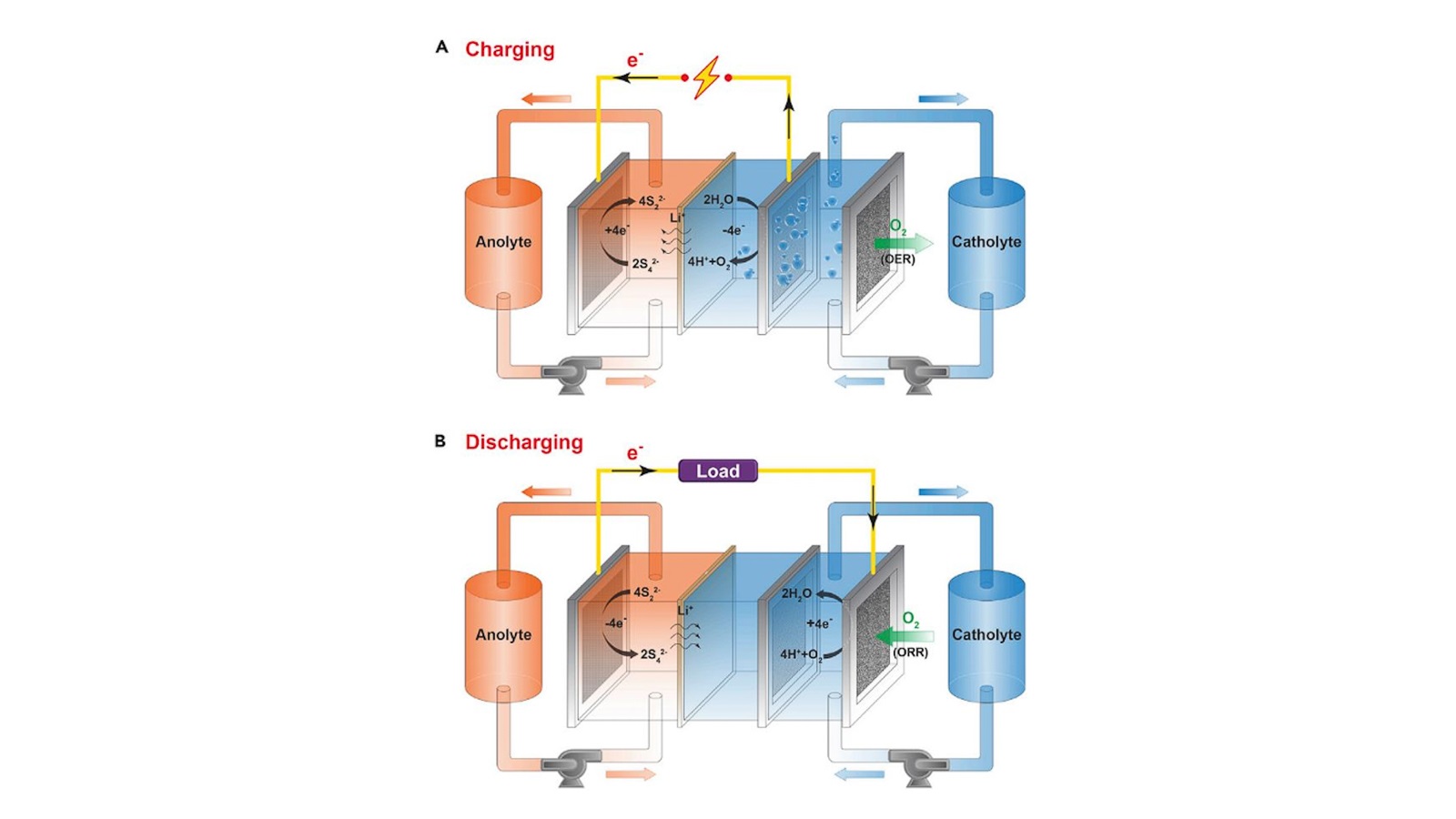
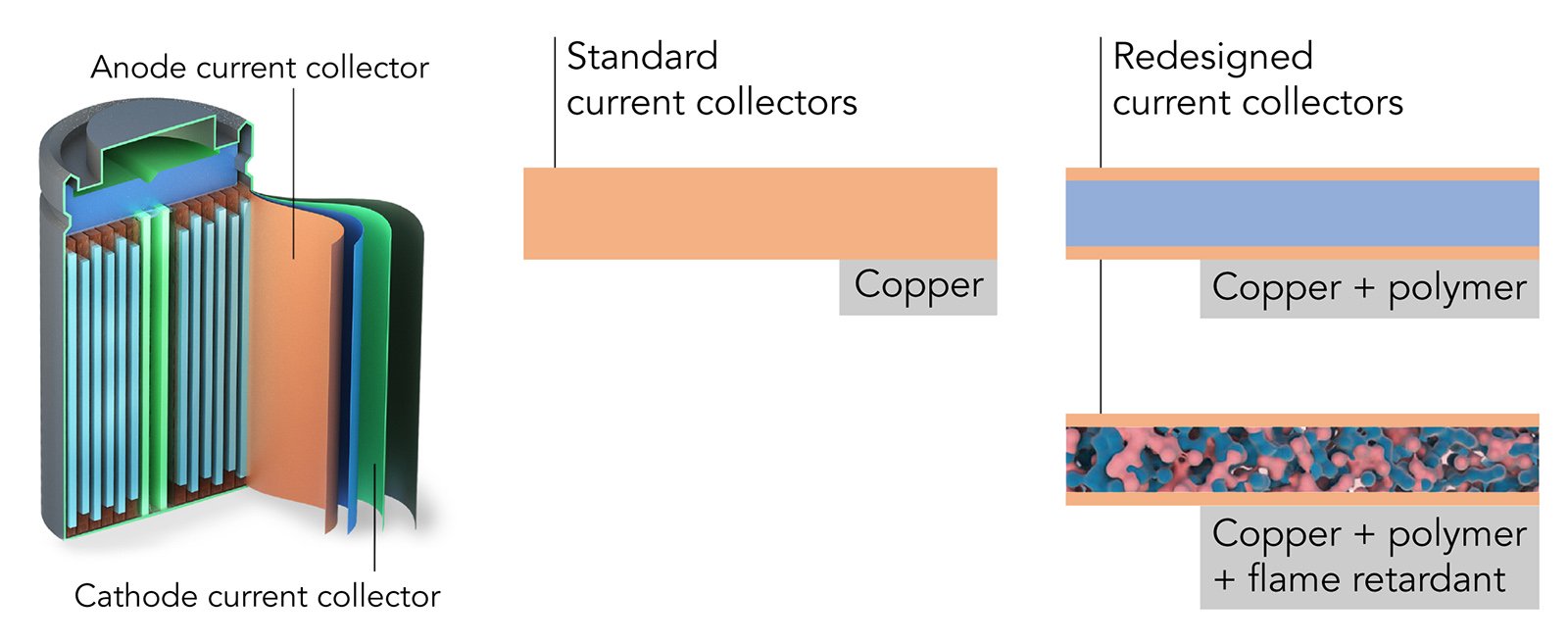
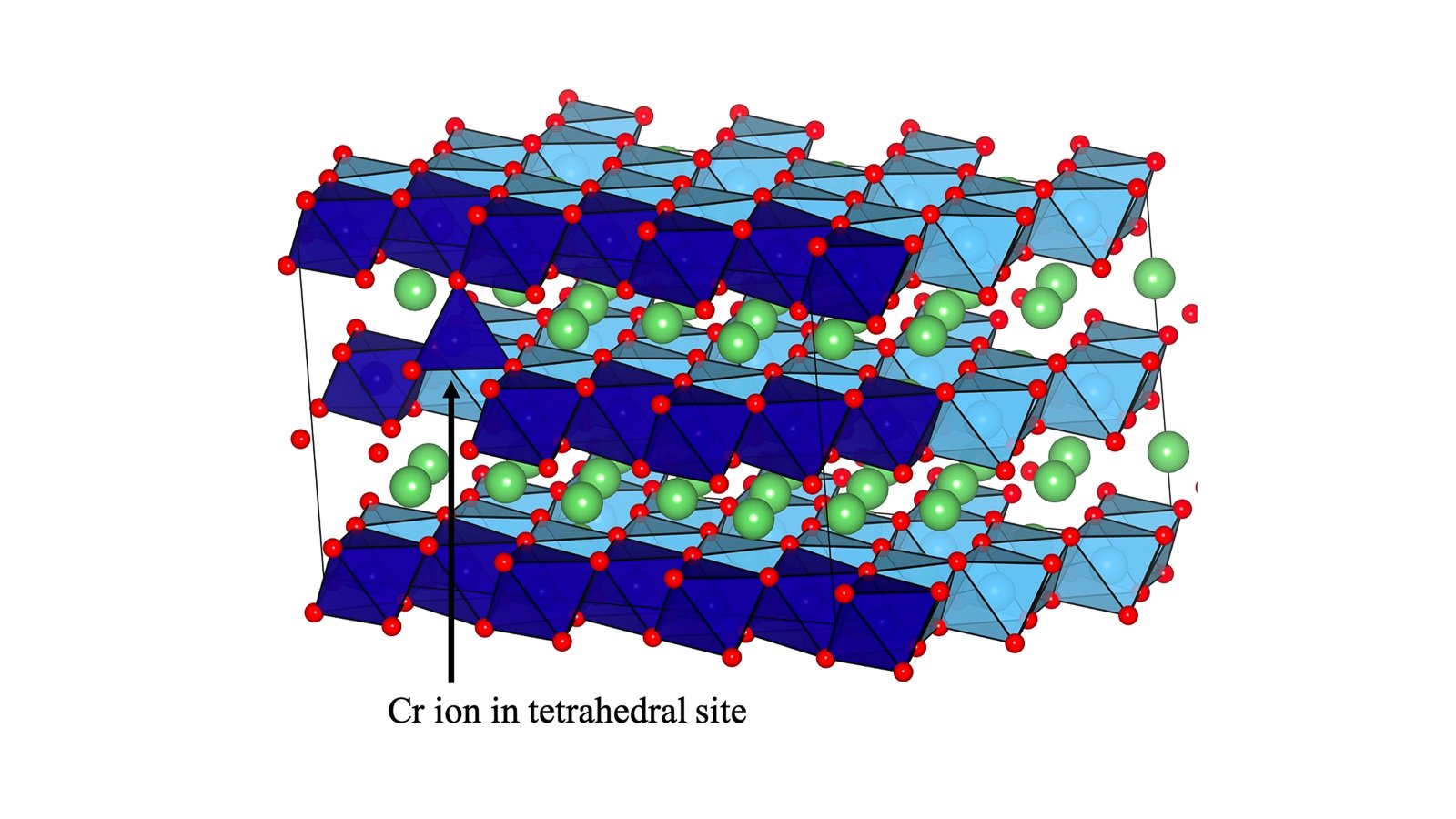
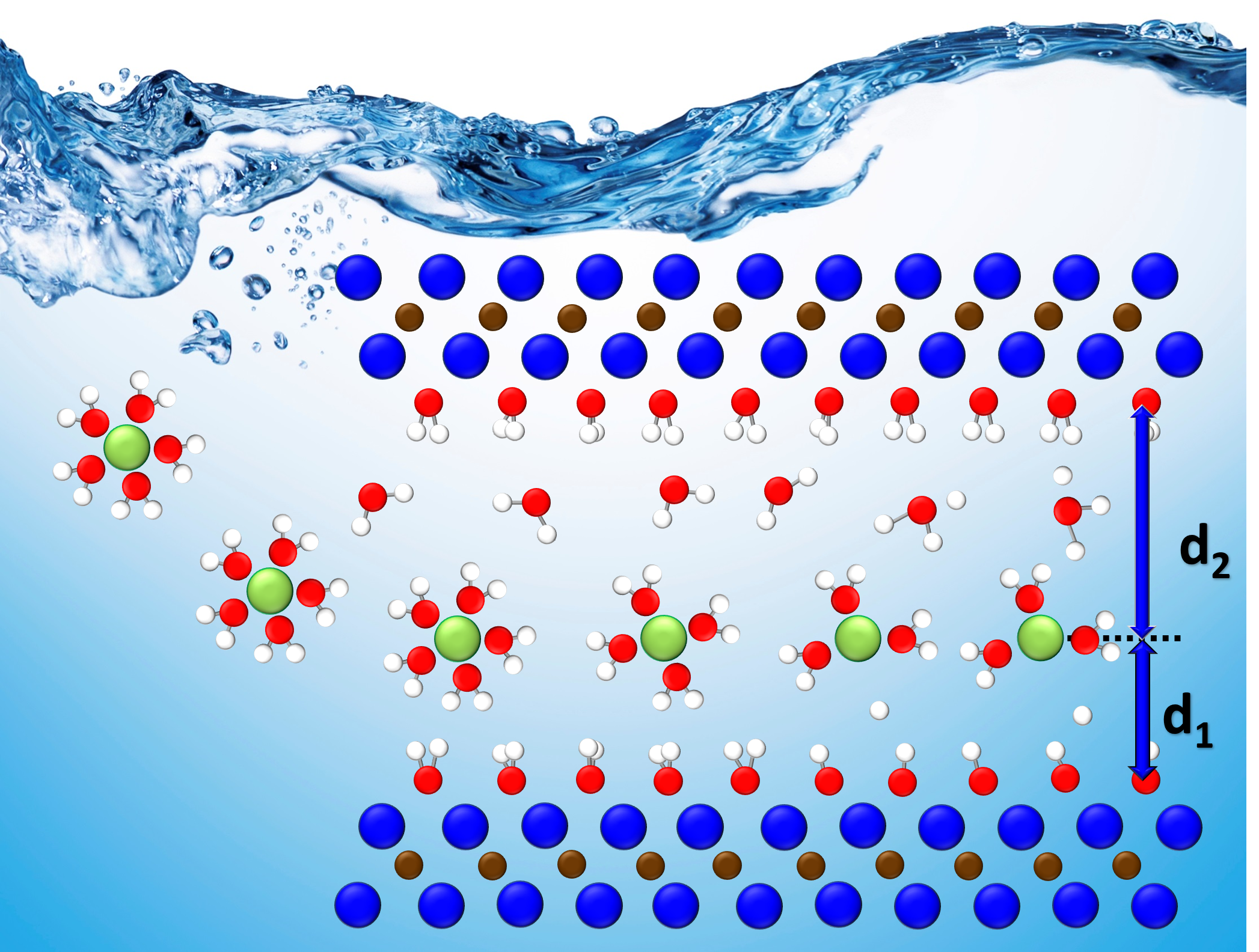
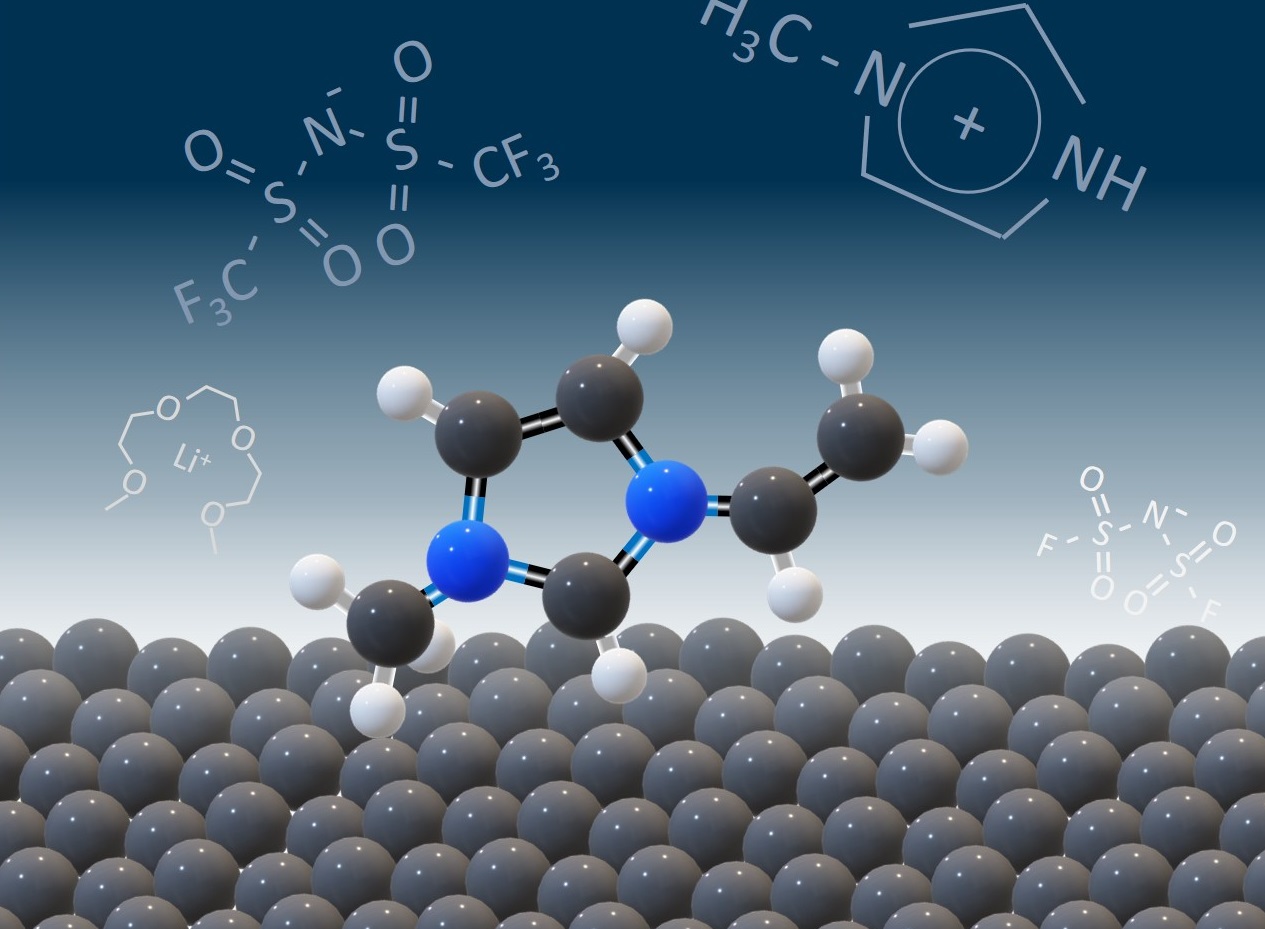
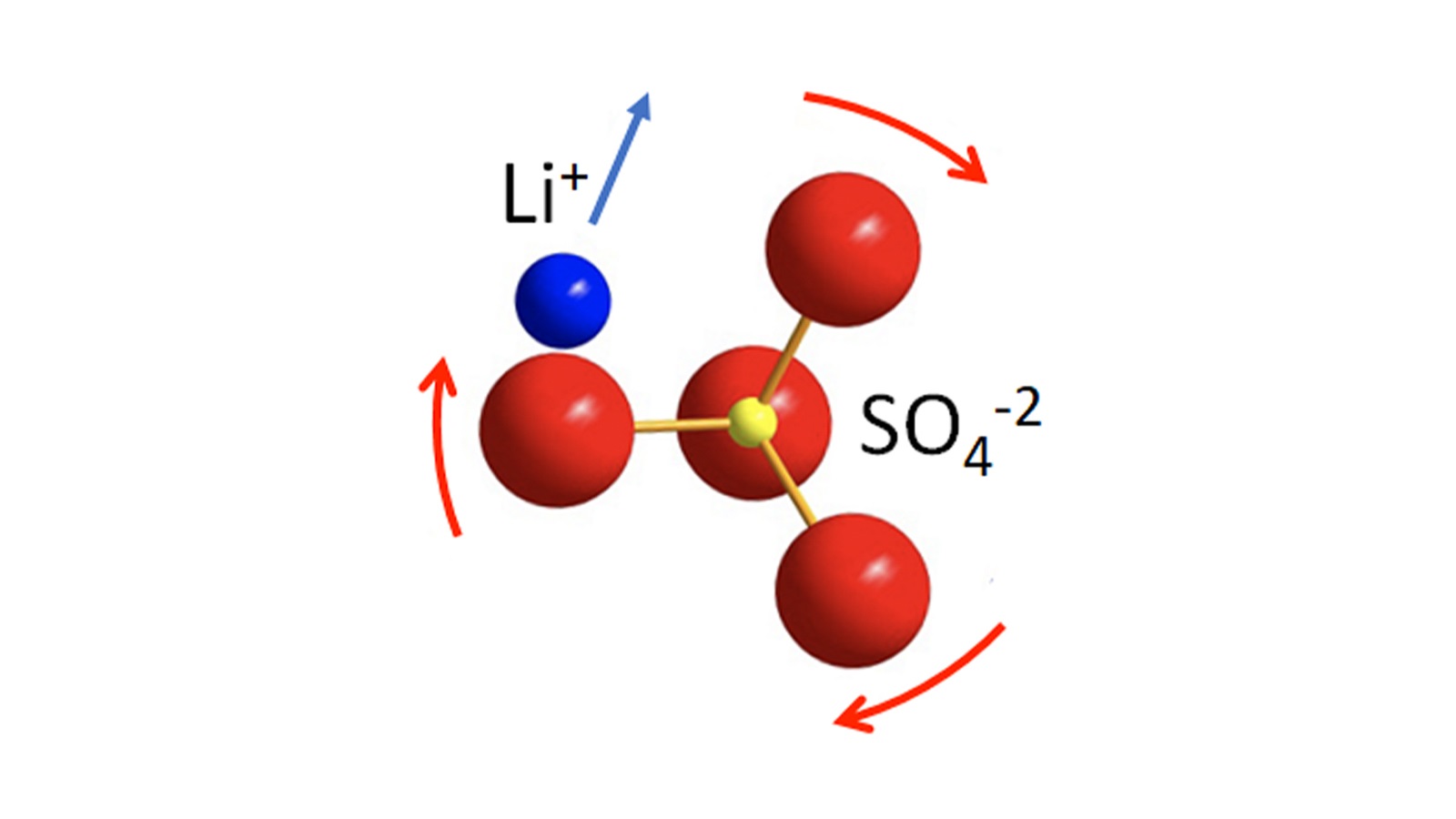
Using a polymer to make a strong yet springy thin film, scientists led by the Department of Energy’s Oak Ridge National Laboratory are speeding the arrival of next-generation solid-state batteries. This effort advances the development of electric vehicle power enabled by flexible, durable sheets of solid-state electrolytes.