Learn more about five research centers at Argonne National Laboratory that help researchers bring climate science from the lab to the world.
Tag: Lithium Ion Batteries
Recycling of valuable metals from spent lithium ion batteries using spinning reactors
In a world that is slowly distancing itself from carbon-based energy, there has been a meteoric rise in the use of lithium-ion batteries as a next-generation energy storage solution.
On the Road to Better Solid-State Batteries
A team from Berkeley Lab and Florida State University has designed a new blueprint for solid-state batteries that are less dependent on specific chemical elements. Their work could advance efficient, affordable solid-state batteries for electric cars.
Flameproofing lithium-ion batteries with salt
A polymer-based electrolyte makes for batteries that keep working – and don’t catch fire – when heated to over 140 degrees F.
Nanoengineers Develop a Predictive Database for Materials
Nanoengineers at the University of California San Diego’s Jacobs School of Engineering have developed an AI algorithm that predicts the structure and dynamic properties of any material—whether existing or new—almost instantaneously. Known as M3GNet, the algorithm was used to develop matterverse.ai, a database of more than 31 million yet-to-be-synthesized materials with properties predicted by machine learning algorithms. Matterverse.ai facilitates the discovery of new technological materials with exceptional properties.
ORNL to lead new center on polymer electrolytes for energy storage
The Department of Energy’s Oak Ridge National Laboratory has been selected to lead an Energy Frontier Research Center, or EFRC, focused on polymer electrolytes for next-generation energy storage devices such as fuel cells and solid-state electric vehicle batteries.
Super-fast electric car charging, with a tailor-made touch
Speeding up electrical vehicle charging can damage the battery. Now, scientists report that they’ve designed superfast charging methods tailored to power different electric vehicle batteries in 10 minutes or less without harm. The researchers will present their results today at ACS Fall 2022.
Rensselaer Researchers Propose an Affordable and Sustainable Alternative to Lithium-Ion Batteries
Concerns regarding scarcity, high prices, and safety regarding the long-term use of lithium-ion batteries has prompted a team of researchers from Rensselaer Polytechnic Institute to propose a greener, more efficient, and less expensive energy storage alternative.
What drives rechargeable battery decay? Depends on how many times you’ve charged it
How quickly a battery electrode decays depends on properties of individual particles in the battery – at first. Later on, the network of particles matters more.
How extreme cold can crack lithium-ion battery materials, degrading performance
Storing the rechargeable batteries at sub-freezing temperatures can crack the battery cathode and separate it from other parts of the battery, a new study shows.
How to Make Lithium-ion Batteries Invincible
Berkeley Lab scientists have made significant progress in developing battery cathodes using a new class of materials that provide batteries with the same if not higher energy density than conventional lithium-ion batteries but can be made of inexpensive and abundant metals. Known as DRX, which stands for disordered rocksalts with excess lithium, this novel family of materials was invented less than 10 years ago and allows cathodes to be made without nickel or cobalt.

Creating Higher Energy Density Lithium-Ion Batteries for Renewable Energy Applications
Lithium-ion batteries that function as high-performance power sources for renewable applications, such as electric vehicles and consumer electronics, require electrodes that deliver high energy density without compromising cell lifetimes. In the Journal of Vacuum Science and Technology A, researchers investigate the origins of degradation in high energy density LIB cathode materials and develop strategies for mitigating those degradation mechanisms and improving LIB performance.

The Road to Cheaper Electric Vehicles
A next-generation battery that will power our electric devices longer and, potentially, make them more affordable and accessible. That’s the focus of Koffi Pierre Yao’s research which has earned a $1 million grant from the U.S. Department of Energy’s Advanced Vehicle Technologies Research program.

How dangerous are burning electric cars?
What happens if an electric car burns in a road tunnel or an underground car park? In the Hagerbach test tunnel in Switzerland, Empa researchers and tunnel safety expert Lars Derek Mellert set fire to battery cells of electric cars, analyzed the distribution of soot and smoke gases and the chemical residues in the extinguishing water.

Fluorine recycling for lithium-ion batteries
Lithium-ion batteries contain salts rich in fluorine, which decompose in humid air to toxic, highly corrosive hydrogen fluoride. The hazardous nature of this substance makes recycling more difficult and more expensive. A research project entitled “Fluoribat” is now being launched at Empa to solve this problem. This could help to make the life cycle of a rechargeable battery less expensive and at the same time safer.

West Virginia researchers use neutrons to study materials for power plant improvements
Researchers from West Virginia University are using neutron scattering at Oak Ridge National Laboratory to study novel materials called high entropy oxides, or HEOs. Their goal is to collect insights into how the atoms in the HEOs bind together and whether the materials can be used to develop useful applications to improve power plant operations.

CFN User Spotlight: Nik Singh Seeks Better Battery Materials
Since 2011, Nikhilendra (Nik) Singh has been a senior scientist in the Materials Research Department at the Toyota Research Institute of North America. His quest to find alternatives to lithium-ion batteries has brought him to Brookhaven Lab’s Center for Functional Nanomaterials (CFN).
Fireproof, lightweight solid electrolyte for safer lithium-ion batteries
But some of the most-studied SSEs are themselves flammable, leaving the original safety concern unaddressed. Researchers now report in ACS’ Nano Letters that they have developed an SSE that won’t burn up.

Seeing inside a battery while it’s working
A new paper-thin radio-frequency detector, developed by researchers at Sandia National Laboratories and designed to work inside a lithium-ion battery, provides information about the battery’s health while charging and discharging.
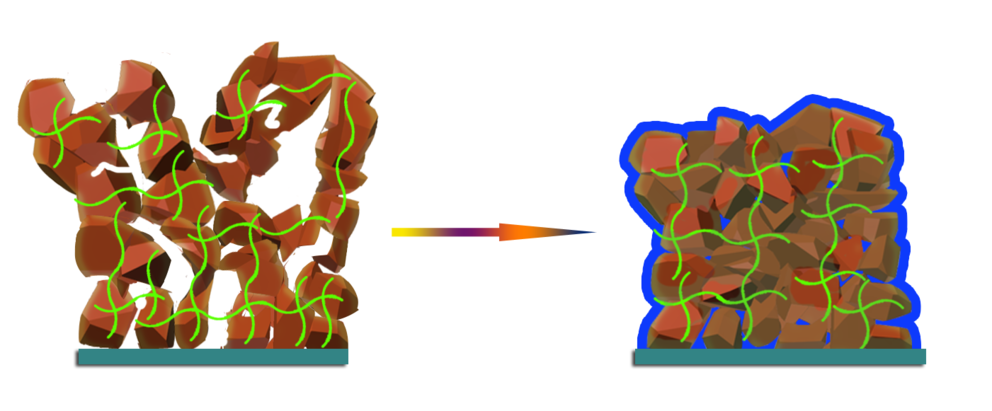
Novel polymer to increase energy density, stability of lithium ion batteries
Penn State researchers have developed a novel method that could enable the widespread use of silicon-based anodes, which allow electricity to enter a device, in rechargeable lithium ion batteries.

Charging Up the Development of Lithium-Ion Batteries
On October 9, the Nobel committee recognized work in developing lithium-ion batteries. These batteries have enabled a huge number of advances, including mobile phones and plug-in electric vehicles. The DOE Office of Science is proud to have supported research by Drs. Whittingham and Goodenough and to have funded research by many scientists who have built upon their innovations.

Binghamton University professor wins Nobel Prize in Chemistry
The 2019 Nobel Prize in Chemistry has been awarded to M. Stanley Whittingham, distinguished professor of chemistry and materials science at Binghamton University, State University of New York.
American Chemical Society’s president comments on award of 2019 Nobel Prize in Chemistry
On behalf of the American Chemical Society (ACS), President Bonnie Charpentier, Ph.D., congratulates today’s winners of the Nobel Prize in Chemistry.

UW-Milwaukee battery startup awarded a second federal commercialization grant
A pair of academic researchers have federal funding for their startup, which will make battery parts from a unique, patented material called graphene monoxide. Their material dramatically boosts the energy storage capacity of li-ion batteries.