Abstract Recently, there has been a vast increase in interest in renewable energy technologies. In the present era of sustainable energy evolution, battery thermal energy storage has emerged as one of the most popular areas. A clean energy alternative to…
Tag: Battery
Confidence-aware reinforcement learning for energy management of electrified vehicles
Abstract The reliability of data-driven techniques, such as deep reinforcement learning (DRL) frequently diminishes in scenarios beyond their training environments. Despite DRL-based energy management strategies (EMS) having gained great popularity in optimizing the energy economy of electrified vehicles (EVs), their performance degradation in untrained…
Sustainability challenges throughout the electric vehicle battery value chain
Abstract The global commitment to decarbonizing the transport sector has resulted in an unabated growth in the markets for electric vehicles and their batteries. Consequently, the demand for battery raw materials is continuously growing. As an illustration, to meet the net-zero…
Robust energy storage system for stable in wind and solar
Abstract Energy storage systems (ESS) have become a conspicuous research hotspot since they store power and supply it during peak hours. Existing storage systems must be replaced by advanced energy storage with improved performance, energy management, and a control interface due to…
Successful Workshop for Leading the ‘Carnot Battery’, the Future of Energy Storage
On May 17th (Friday), the Korea Institute of Energy Research (KIER) held a workshop at its main headquarters in Daejeon with experts from industry, academia, research, and government to discuss the technology, policy status, and development direction of the Carnot Battery in relation to energy storage and heat pumps.
Reduce the risk of electric vehicle fires by analyzing nanostructures!
Dr. Hyeon-woo Son and his research team from the Department of Aluminum in the Advanced Metals Division at KIMS have successfully developed an aluminum alloy for electric vehicles that dramatically improves thermal stability.

Batteries: Modeling Tomorrow’s Materials Today
Which factors determine how quickly a battery can be charged? This and other questions are studied by researchers of Karlsruhe Institute of Technology (KIT) with the help of computer-based simulations.
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells
New research from The Grainger College of Engineering suggests that observing how heat flows in conjunction with electricity can give important insights into battery chemistry.
On the Road to Better Solid-State Batteries
A team from Berkeley Lab and Florida State University has designed a new blueprint for solid-state batteries that are less dependent on specific chemical elements. Their work could advance efficient, affordable solid-state batteries for electric cars.
Idaho technology changing outlook of advanced materials development
On their way to market, technologies often reach what is called the “valley of death,” the point where a researcher or institution has developed a promising idea, has received funding through grants, and then runs out of cash to move the idea beyond the laboratory.
Taking Freight Trucks Electric Would Have Big Economic and Environmental Benefits for India
Diesel-fueled freight trucks play an outsized role in producing India’s total greenhouse gas and air pollution emissions. While the country has promoted policies to transition to electric vehicles for public transportation buses and cars, batteries that can power such large trucks have been too heavy and expensive to make their electrification possible. A new study shows that advances in battery technology and dramatically decreased battery costs in recent years have changed that. With the right policies and incentives, battery electric trucks would be more affordable to operate than diesel, and India could become a world leader in producing electric vehicles.
Small business tests promising new battery at Idaho National Laboratory
Researchers are working to find an alternative to lithium-ion batteries that, despite their use in everything from smartphones to electric vehicles, have some disadvantages, especially their relatively low energy density compared to gasoline.
Energy, Maritime Leaders to Convene for DOE InnovationXLab Summit
Top scientists and officials from government, academia, Alaskan Native communities, and industry are heading to Alaska to focus on driving energy technologies for a more sustainable Arctic region.

Center advancing beyond lithium battery technologies generates 30-plus patents for licensing
Researchers at the Joint Center for Energy Storage Research have invented a wide and diverse range of technologies in the “beyond lithium-ion” space, including 30-plus patents now available for licensing.
Argonne taps internationally recognized researcher Shirley Meng as chief scientist for energy storage strategy
A pioneer in material science, Meng’s new role comes with a joint appointment as a professor at the Pritzker School of Molecular Engineering at The University of Chicago.
Argonne and the National Electrical Manufacturers Association cooperate to develop battery recycling standards
The U.S. Department of Energy’s (DOE) Argonne National Laboratory has signed a memorandum of understanding with the National Electrical Manufacturers Association (NEMA)
Tulane scientist leads study on faster charging energy storage
A Tulane University researcher has led a team in discoveries that could result in significantly faster charging electric vehicles and portable devices such as cell phones and laptops.
Tracking Pileups on Battery Charging Route to Drive Performance
An understanding of this mechanism could help scientists increase the total amount of energy stored by next-generation lithium-ion batteries.
MEDIA ADVISORY: AIP Publishing Hosts Expert Sessions on Energy Storage and Conversion at Virtual Conference
Twelve distinguished speakers will be covering critical topics impacting energy storage and conversion at the upcoming AIP Publishing Horizons Virtual Conference on Aug. 4-6. The three-day event is organized by the journal Applied Physics Reviews and brings together leaders in the field of energy science to present their latest research in six sessions
The biodegradable battery
The number of data-transmitting microdevices, for instance in packaging and transport logistics, will increase sharply in the coming years. All these devices need energy, but the amount of batteries would have a major impact on the environment. Empa researchers have developed a biodegradable mini-capacitor that can solve the problem. It consists of carbon, cellulose, glycerin and table salt. And it works reliably.
PNNL Invention Reduces Risk of Battery Explosions
A deceptively simple sensor system developed at PNNL can prevent dangerous battery fires.
Battery parts can be recycled without crushing or melting
Researchers at Aalto University have discovered that electrodes in lithium batteries containing cobalt can be reused as is after being newly saturated with lithium. In comparison to traditional recycling, which typically extracts metals from crushed batteries by melting or dissolving them, the new process saves valuable raw materials, and likely also energy.
Mapping Performance Variations to See How Lithium-Metal Batteries Fail
Scientists have identified the primary cause of failure in a state-of-the-art lithium-metal battery, of interest for long-range electric vehicles: electrolyte depletion.

April Snapshots
Science Snapshots from Berkeley Lab: X-rays accelerate battery R&D; infrared microscopy goes off grid; substrates support 2D tech
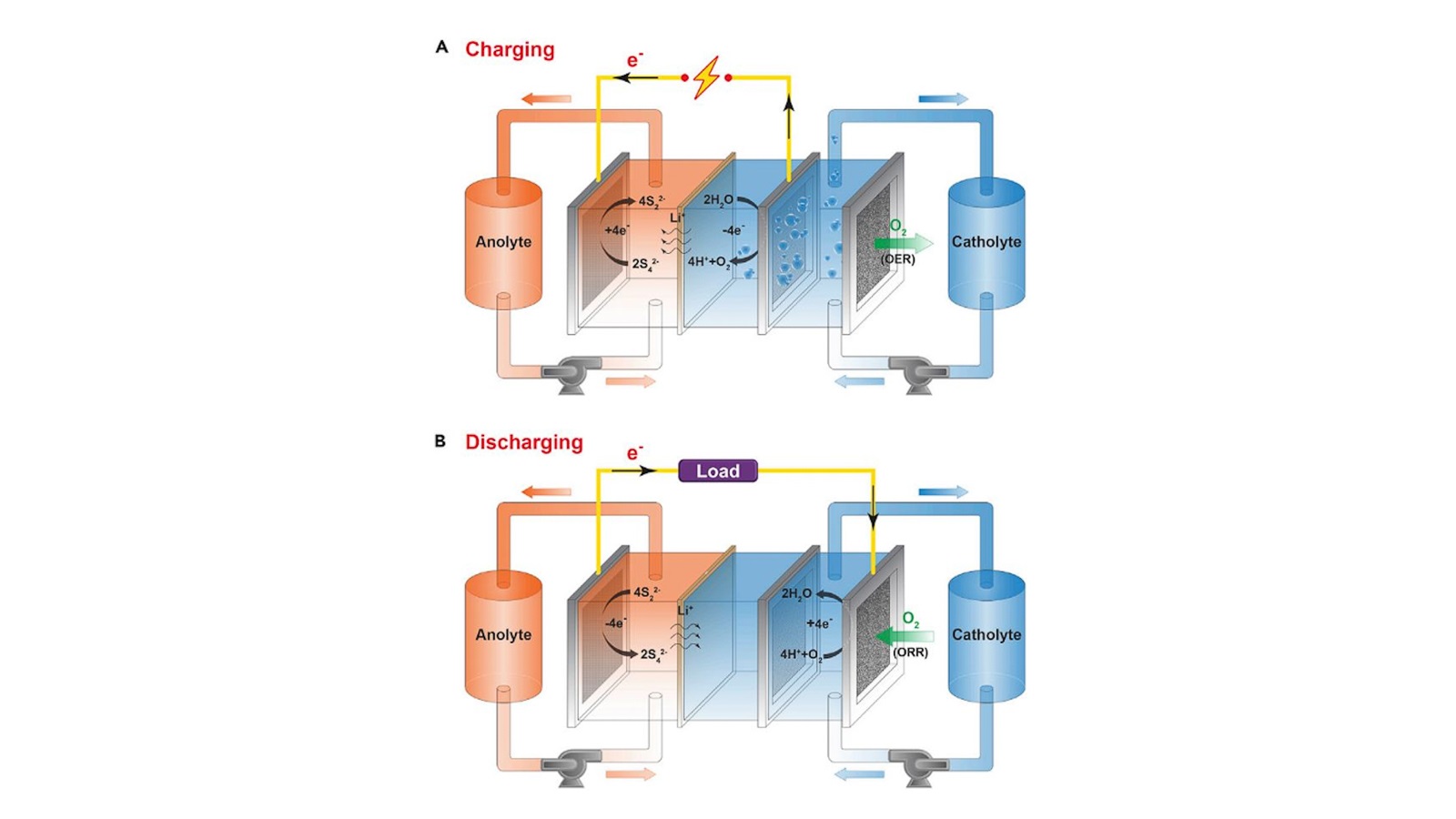
Reshaping the future of the electric grid through low-cost, long-duration discharge batteries
Research begun at the Department of Energy’s Joint Center for Energy Storage Research and continued at spinoff company Form Energy may launch a new era of renewable energy.

Worth their salt: New battery anodes use salt for energy, stability
Researchers at the U.S. Department of Energy’s Argonne National Laboratory and the University of California San Diego have discovered that a material that looks geometrically similar to rock salt could be an interesting candidate for lithium battery anodes that would be used in fast charging applications.

Inside the battery in 3D: Powerful X-rays watch solid state batteries charging and discharging
Using high-speed X-ray tomography, researchers captured images of solid-state batteries in operation and gained new insights that may improve their efficiency.

ORNL receives three 2021 FLC Awards for technology transfer
Three technologies developed by researchers at Oak Ridge National Laboratory have won National Technology Transfer Awards from the Federal Laboratory Consortium. The annual FLC Awards recognize significant accomplishments in transferring federal laboratory technologies to the marketplace.

X-Ray Tomography Lets Researchers Watch Solid-State Batteries Charge, Discharge
Using X-ray tomography, a research team has observed the internal evolution of the materials inside solid-state lithium batteries as they were charged and discharged. Detailed three-dimensional information from the research could help improve the reliability and performance of the batteries, which use solid materials to replace the flammable liquid electrolytes in existing lithium-ion batteries.
An anode-free zinc battery that could someday store renewable energy
Researchers reporting in ACS’ Nano Letters have made a prototype of an anode-free, zinc-based battery that uses low-cost, naturally abundant materials.

The continuing quest to find a better battery
Taking a look back at the paths taken by the Joint Center for Energy Storage Research.

Creating Higher Energy Density Lithium-Ion Batteries for Renewable Energy Applications
Lithium-ion batteries that function as high-performance power sources for renewable applications, such as electric vehicles and consumer electronics, require electrodes that deliver high energy density without compromising cell lifetimes. In the Journal of Vacuum Science and Technology A, researchers investigate the origins of degradation in high energy density LIB cathode materials and develop strategies for mitigating those degradation mechanisms and improving LIB performance.
Lawrence Livermore Lab to Co-host Bay Area Battery Summit
Lawrence Livermore National Laboratory (LLNL), New Energy Nexus and CalCharge are co-hosting the sixth annual Bay Area Battery Summit (BABS), to be held virtually on Nov. 17-18.

Improving High-Energy Lithium-Ion Batteries with Carbon Filler
Lithium-ion batteries are the major rechargeable power source for many portable devices as well as electric vehicles, but their use is limited, because they do not provide high power output while simultaneously allowing reversible energy storage. Research reported in Applied Physics Reviews aims to offer a solution by showing how the inclusion of conductive fillers improves battery performance.
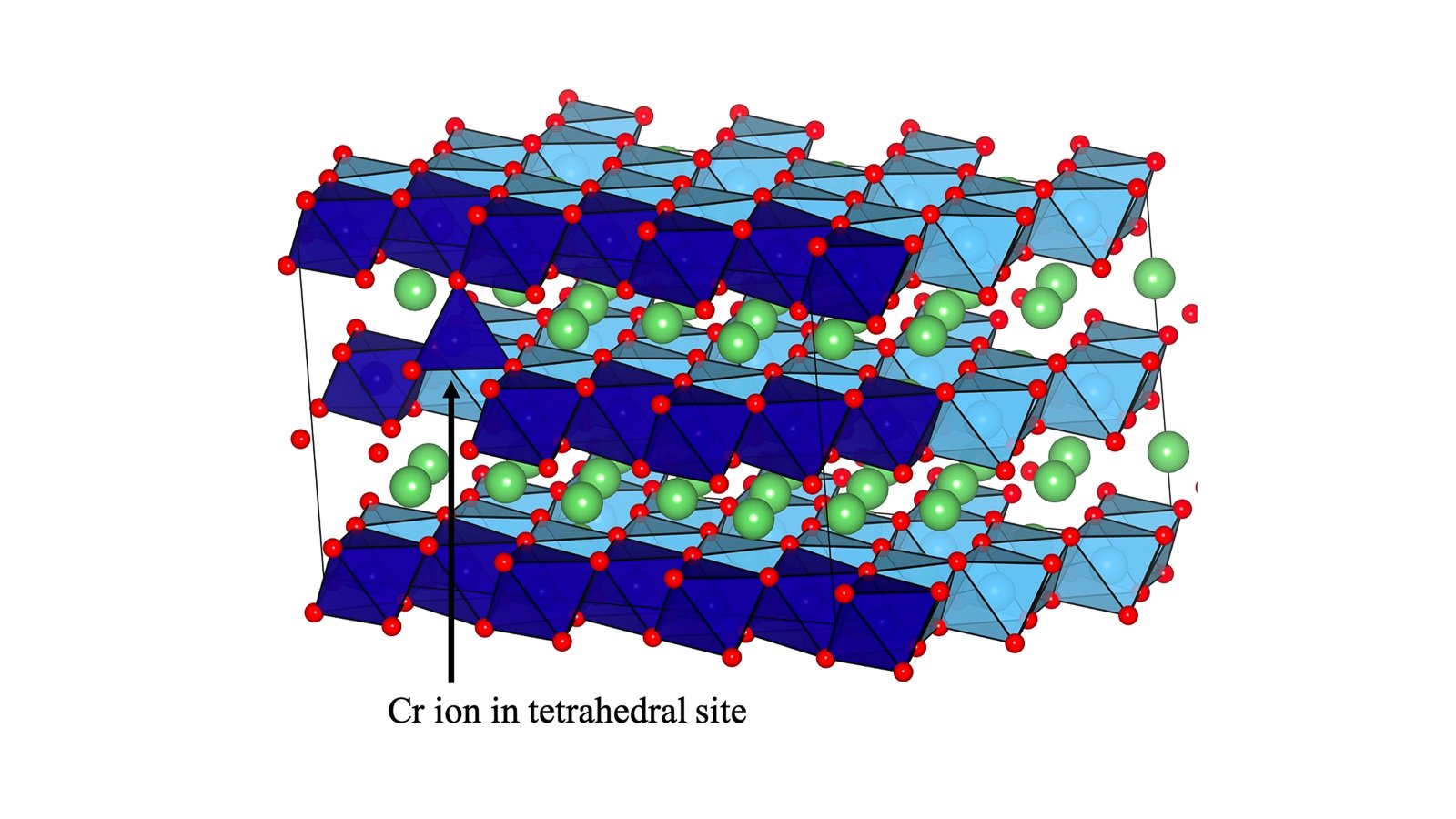
Argonne researchers target lithium-rich materials as key to more sustainable, cost-effective, next-generation batteries
Researchers are developing new ways to advance lithium-rich batteries and using new materials for practical use, according to researchers with the U.S. Department of Energy’s Argonne National Laboratory.

Battery Breakthrough Gives Boost to Electric Flight and Long-Range Electric Cars
Researchers at Berkeley Lab, in collaboration with Carnegie Mellon University, have developed a new battery material that could enable long-range electric vehicles that can drive for hundreds of miles on a single charge, and electric planes called eVTOLs for fast, environmentally friendly commutes.

Spontaneous Formation of Nanoscale Hollow Structures Could Boost Battery Storage
An unexpected property of nanometer-scale antimony crystals — the spontaneous formation of hollow structures — could help give the next generation of lithium ion batteries higher energy density without reducing battery lifetime. The reversibly hollowing structures could allow lithium ion batteries to hold more energy and therefore provide more power between charges.
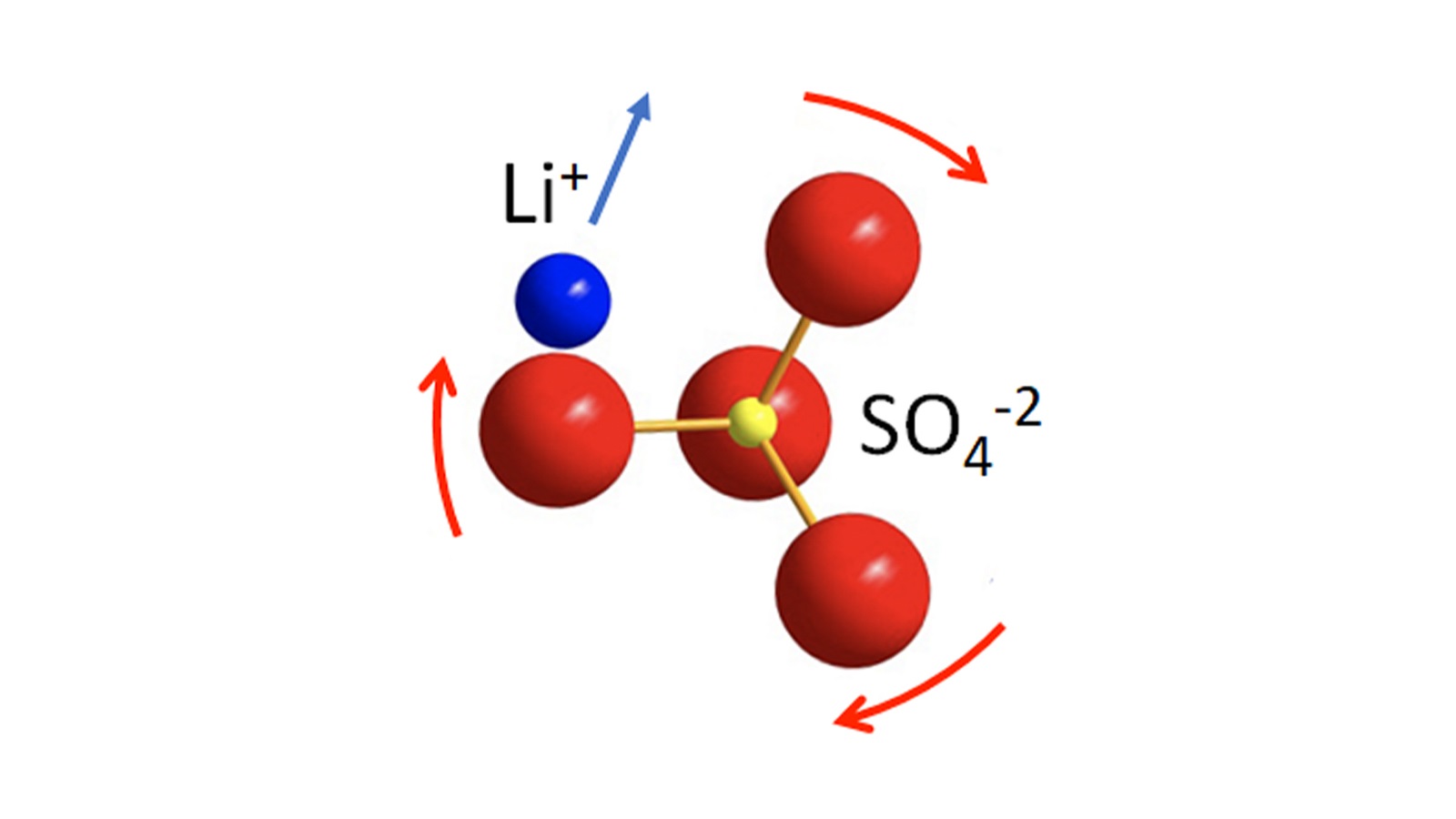
JCESR lays foundation for safer, longer-lasting batteries
Researchers sped-up the motion of lithium ions in solid-state batteries using the paddlewheel effect.

Esther Takeuchi on Batteries
In the First-Person Science series, scientists describe how they made significant discoveries over years of research. Esther Takeuchi is a professor at Stony Brook University and the director of the Center for Mesoscale Transport Properties, a Department of Energy Office of Science Energy Frontier Research Center.

Story Tips: Tracking populations, UPS’ special delivery and a long-awaited benchmark
Story tips from the Department of Energy’s Oak Ridge National Laboratory
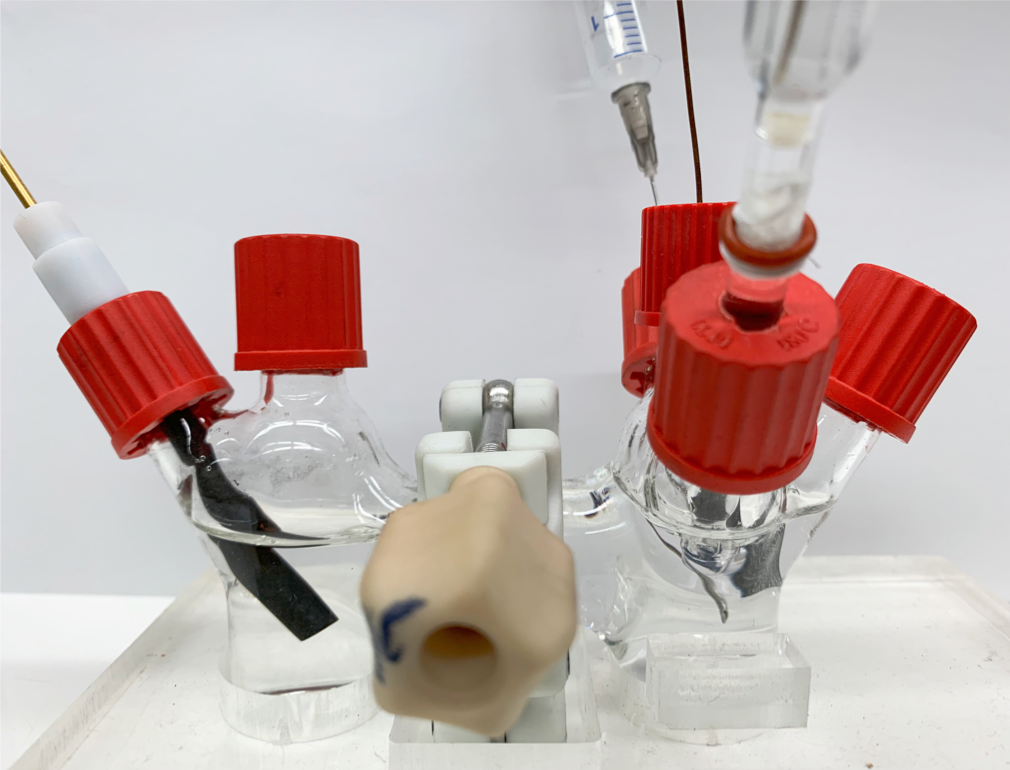
Green method could enable hospitals to produce hydrogen peroxide in house
A team of researchers has developed a portable, more environmentally friendly method to produce hydrogen peroxide. It could enable hospitals to make their own supply of the disinfectant on demand and at lower cost.
Chasing Lithium Ions on the Move in a Fast-Charging Battery
Atomic distortions emerging in the electrode during operation provide a “fast lane” for the transport of lithium ions.
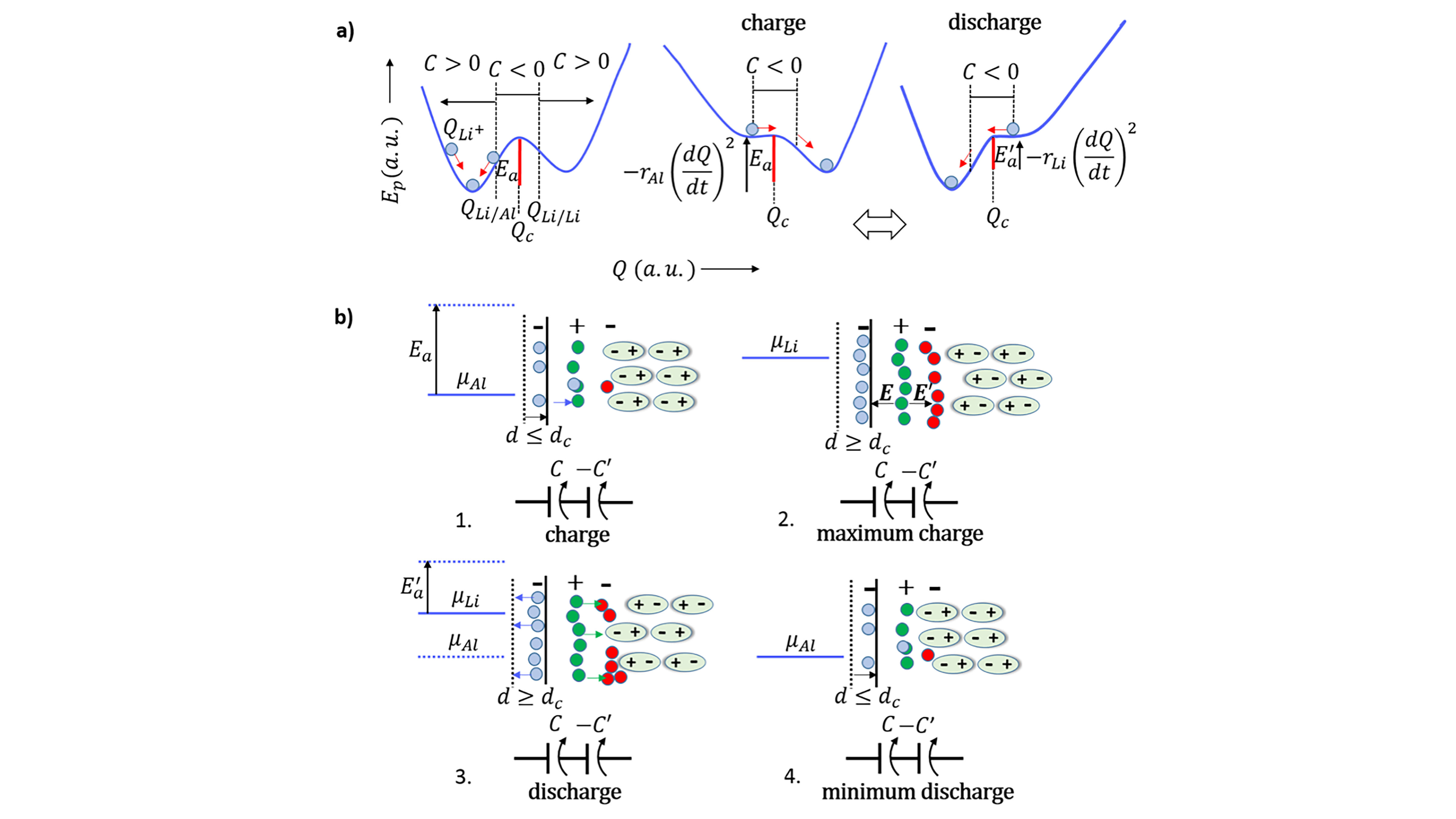
Simple Self-Charging Battery Offers Power Solutions for Devices
A new type of battery combines negative capacitance and negative resistance within the same cell, allowing the cell to self-charge without losing energy, which has important implications for long-term storage and improved output power for batteries. In Applied Physics Reviews, researchers at the University of Porto and the University of Texas at Austin report making their very simple battery with two different metals, as electrodes and a lithium or sodium glass electrolyte between them.
Battery collaboration meeting discusses new pathways to recycle lithium-ion batteries
At a conference held by the ReCell Center, an advanced battery recycling collaboration based at Argonne, representatives from industry, government, and academia discussed innovative approaches for lithium-ion battery recycling.
Building a better battery with machine learning
In two new papers, researchers at the U.S. Department of Energy’s (DOE) Argonne National Laboratory have turned to the power of machine learning and artificial intelligence to dramatically accelerate battery discovery.