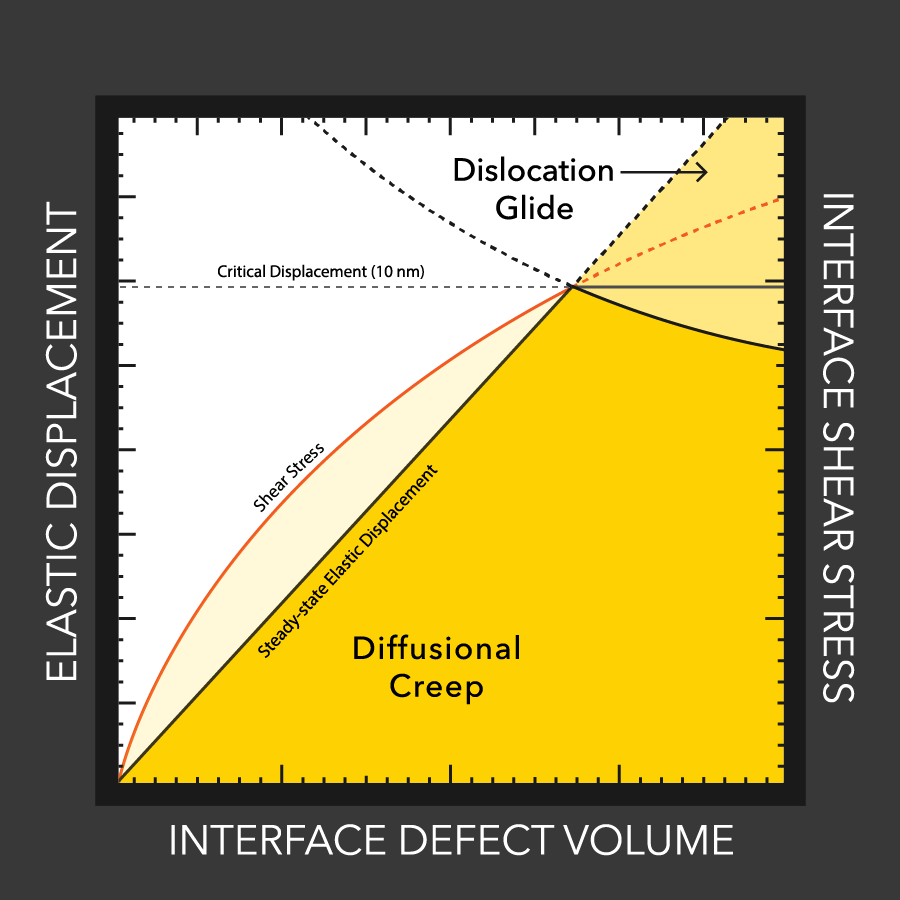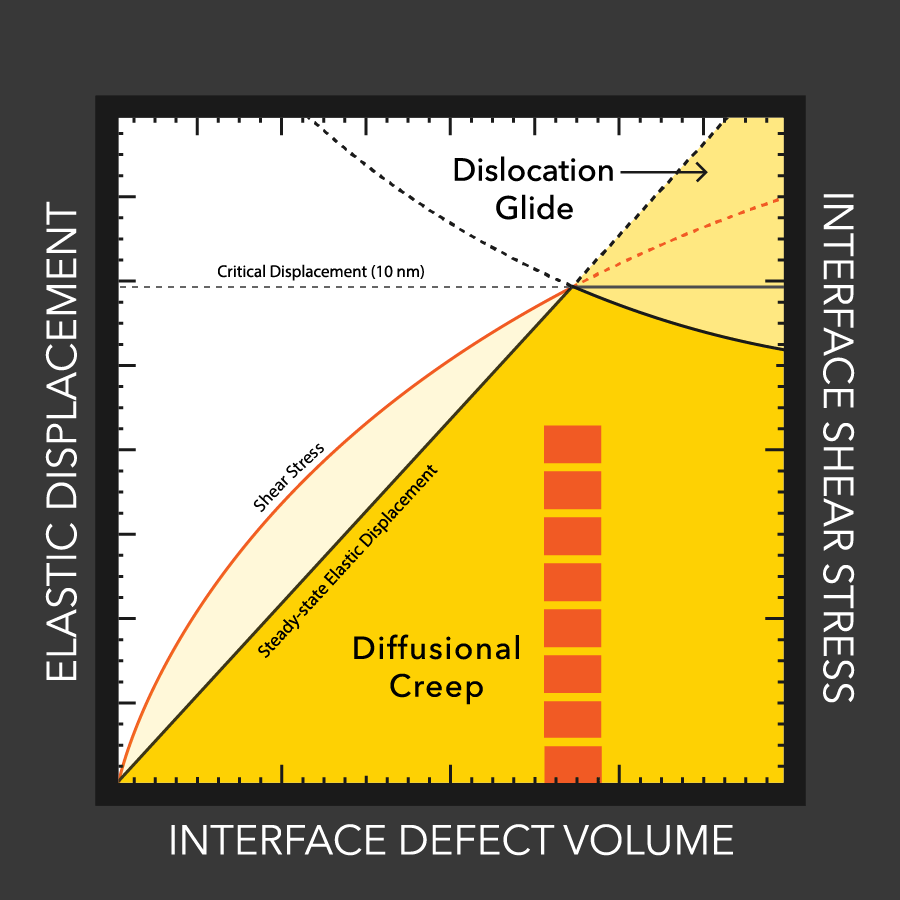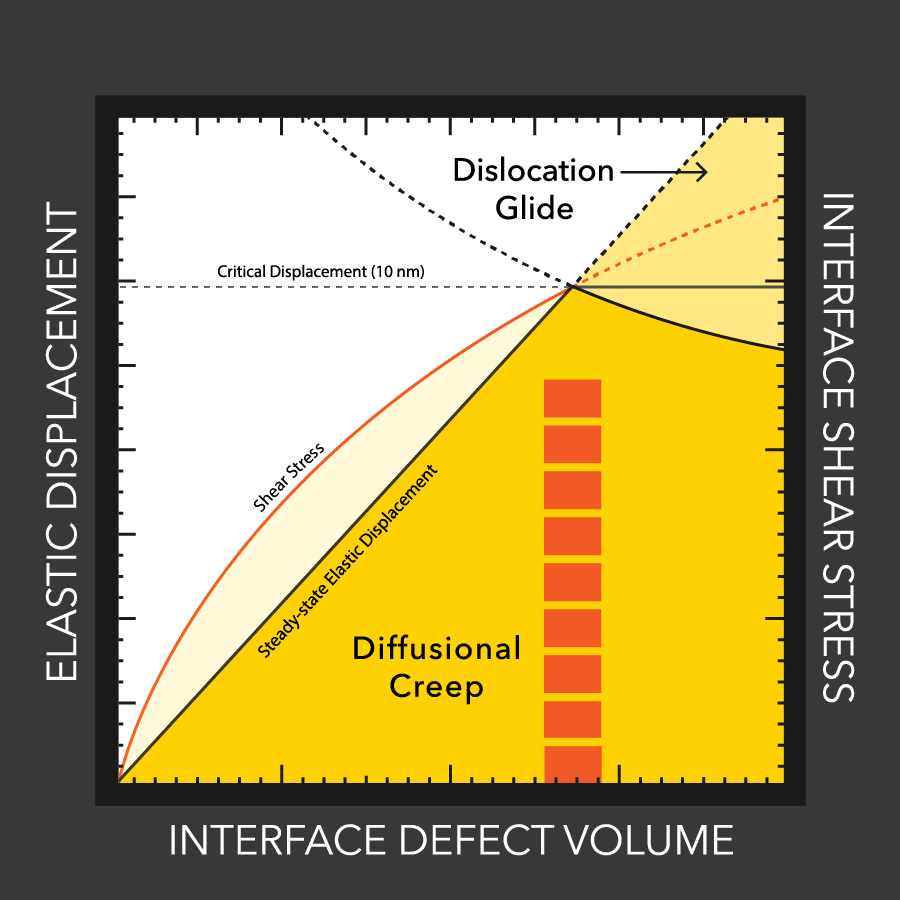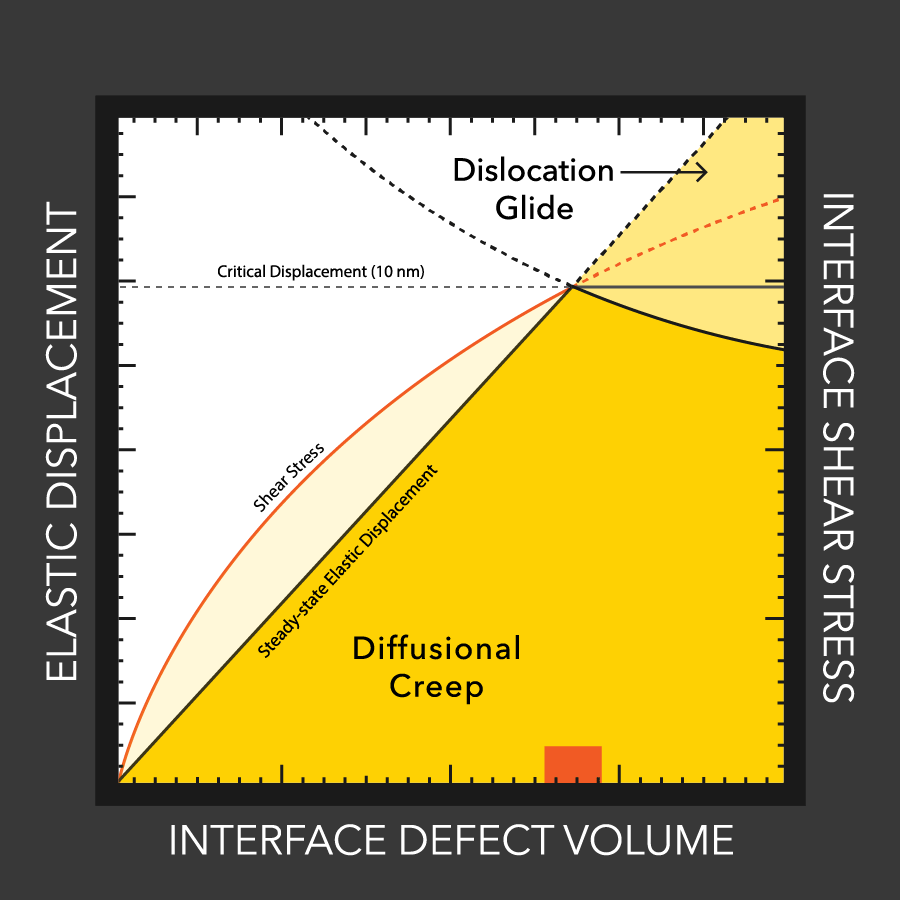
A team of scientists led by chemists at the U.S. Department of Energy’s Brookhaven National Laboratory and Pacific Northwest National Laboratory has unraveled the complex chemical mechanisms of a battery component that is crucial for boosting energy density: the interphase.