New research from The Grainger College of Engineering suggests that observing how heat flows in conjunction with electricity can give important insights into battery chemistry.
Tag: Lithium Ion
Battery parts can be recycled without crushing or melting
Researchers at Aalto University have discovered that electrodes in lithium batteries containing cobalt can be reused as is after being newly saturated with lithium. In comparison to traditional recycling, which typically extracts metals from crushed batteries by melting or dissolving them, the new process saves valuable raw materials, and likely also energy.
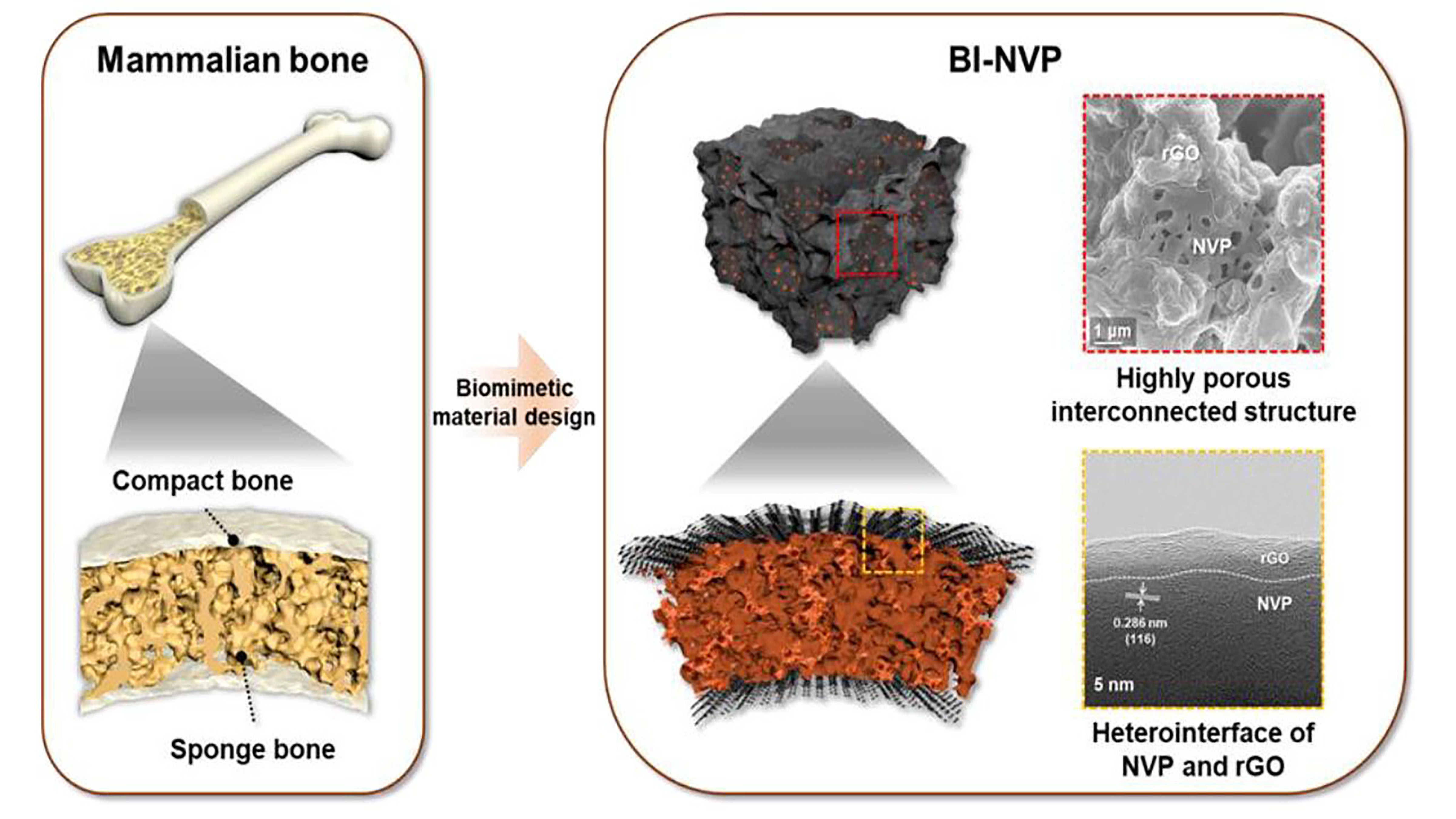
Batteries Mimic Mammal Bones for Stability
Sodium-ion batteries offer several advantages over lithium-ion batteries; however, it is difficult to develop sodium cathodes, materials through which electrons can enter a battery. Many candidate materials are unstable or cannot withstand high voltages. To find a solution, researchers turned to nature. They created a porous system of NVP structures, surrounded by a dense shell of reduced graphene oxide. They describe the mammal bone-inspired sodium cathode in the journal Applied Physics Reviews.

Creating Higher Energy Density Lithium-Ion Batteries for Renewable Energy Applications
Lithium-ion batteries that function as high-performance power sources for renewable applications, such as electric vehicles and consumer electronics, require electrodes that deliver high energy density without compromising cell lifetimes. In the Journal of Vacuum Science and Technology A, researchers investigate the origins of degradation in high energy density LIB cathode materials and develop strategies for mitigating those degradation mechanisms and improving LIB performance.