Abstract The rising demand for Li, paramount for energy storage, necessitates expanded supply. As the supply is concentrated in a few countries, this poses supply chain risks for Li-ion battery makers. To diversify suppliers, alternative Li ore deposits such as…
Tag: Lithium
Making batteries takes lots of lithium. Almost half of it could come from Pennsylvania wastewater.
A new analysis using compliance data from the Pennsylvania Department of Environmental Protection suggests that if it could be extracted with complete efficiency, lithium from the wastewater of Marcellus shale gas wells could supply up to 40% of the country’s demand. The research is by University of Pittsburgh and National Energy Technology Laboratory scientists.
Scientists are shaking up lithium extraction with a different kind of chemistry
Scientists at the Critical Materials Innovation (CMI) Hub, led by the U.S. Department of Energy’s Ames National Laboratory, have developed a new process, mechanochemical extraction of lithium at low temperatures, or MELLT, to increase and diversify the supply of lithium in the United States.
Adding just enough fuel to the fire
PPPL researchers have determined the maximum density of uncharged particles at the edge of a plasma before certain instabilities become unpredictable. This is the first time such a level has been established for Lithium Tokamak Experiment-Beta. Knowing this level is a big step in their mission to prove lithium is the ideal choice for an inner-wall coating in a tokamak because it guides them toward the best practices for fueling their plasmas.
Cathode active materials for lithium-ion batteries could be produced at low temperatures
Layered lithium cobalt oxide, a key component of lithium-ion batteries, has been synthesized at temperatures as low as 300°C and durations as short as 30 minutes.
Sustainable Lithium for Many Decades
On the way towards climate neutrality, Europe will need large amounts of lithium for battery storage systems. So far, however, its share in the worldwide lithium extraction volume has been one percent only.
Lithium imaging method could shine new light on bipolar disorder, treatment
Researchers reporting in ACS Central Science developed a method for imaging lithium in living cells, allowing them to discover that neurons from bipolar disorder patients accumulate higher levels of lithium than healthy controls.
Novel method of imaging silicon anode degradation may lead to better batteries
A novel method of characterizing the structural and chemical evolution of silicon and a thin layer that governs battery stability may enable better, cheaper batteries.
Magnetic Nanoparticles Pull Valuable Elements from Water Sources
Earth-friendly process may help make critical materials sourcing economically feasible in the U.S.

Egemen Kolemen wins 2020 Excellence in Fusion Engineering Award
Egemen Kolemen, Princeton University assistant professor and PPPL physicist, wins prestigious Fusion Power Associates award.

Geothermal Brines Could Propel California’s Green Economy
Deep beneath the surface of the Salton Sea, a shallow lake in California’s Imperial County, sits an immense reserve of critical metals that, if unlocked, could power the state’s green economy for years to come. These naturally occurring metals are dissolved in geothermal brine, a byproduct of geothermal energy production. Now the race is on to develop technology to efficiently extract one of the most valuable metals from the brine produced by the geothermal plants near the Salton Sea: lithium.

First results of an upgraded experiment highlight the value of lithium for the creation of fusion energy
Initial results of the Lithium Tokamak Experiment-Beta (LTX-β) at PPPL show that the enhancements significantly improve performance of the plasma that will fuel future fusion reactors.

Lithium Ion Battery Waste Used in Biodiesel Production from Discarded Vegetable Oil
Brazilian researchers demonstrated a new chemical approach for producing biodiesel from domestic cooking oil waste by using hydroxide lithium mixed with either sodium hydroxides or potassium hydroxides as catalysts. Their work, published in the Journal of Renewable and Sustainable Energy, could enable future studies related to the use of lithium from waste lithium ion batteries. The work marks one of the first times lithium has been used for such purposes.

The Electrochemical Society and Toyota Motor Engineering & Manufacturing North America, Inc. Announce 2020-2021 Fellowship Winners for Projects in Green Energy Technology
Prof. Dr. Shoji Hall, Prof. Dr. Piran Ravichandran Kidambi, and Dr. Haegyeom Kim have been awarded the 2020-2021 ECS Toyota Young Investigator Fellowships. Through this, ECS and Toyota aim to promote innovative and unconventional technologies borne from electrochemical research. The fellowship encourages young professors and scholars to pursue innovative electrochemical research in green energy technology.
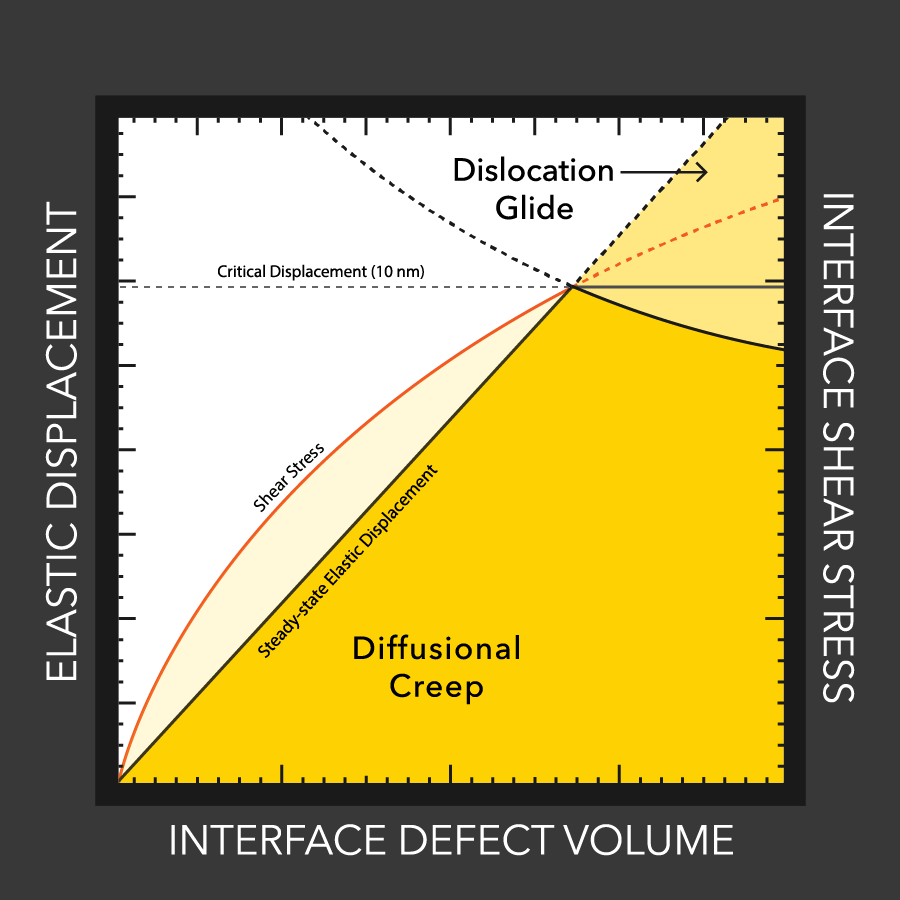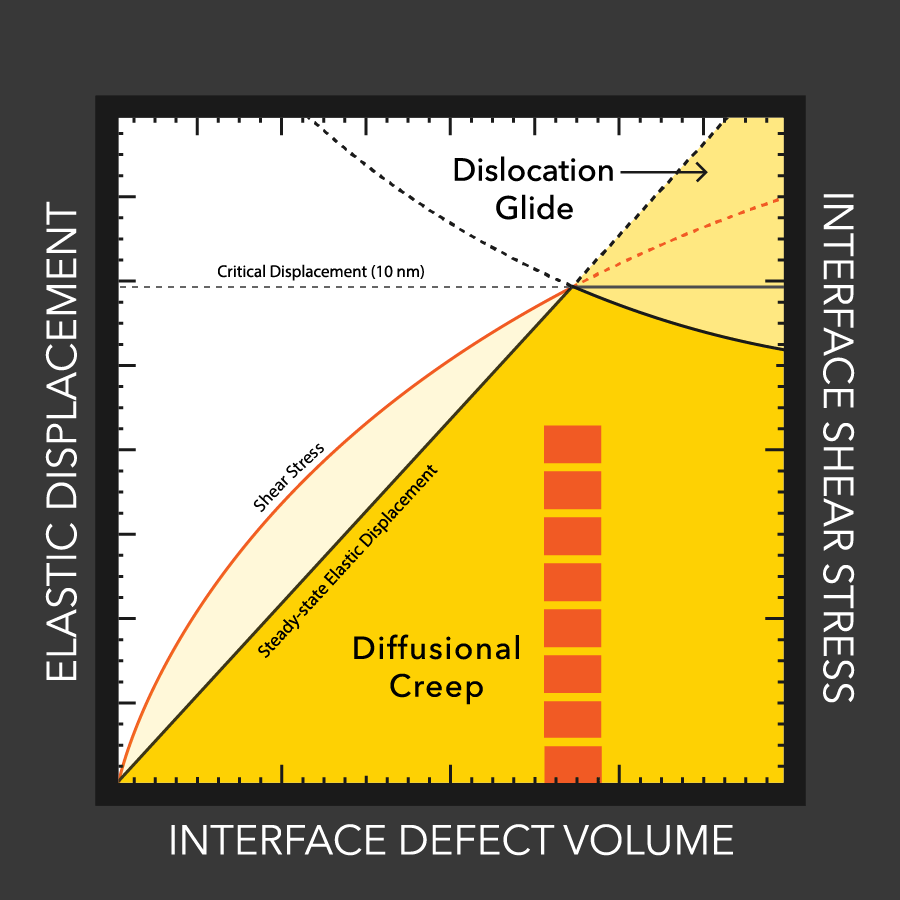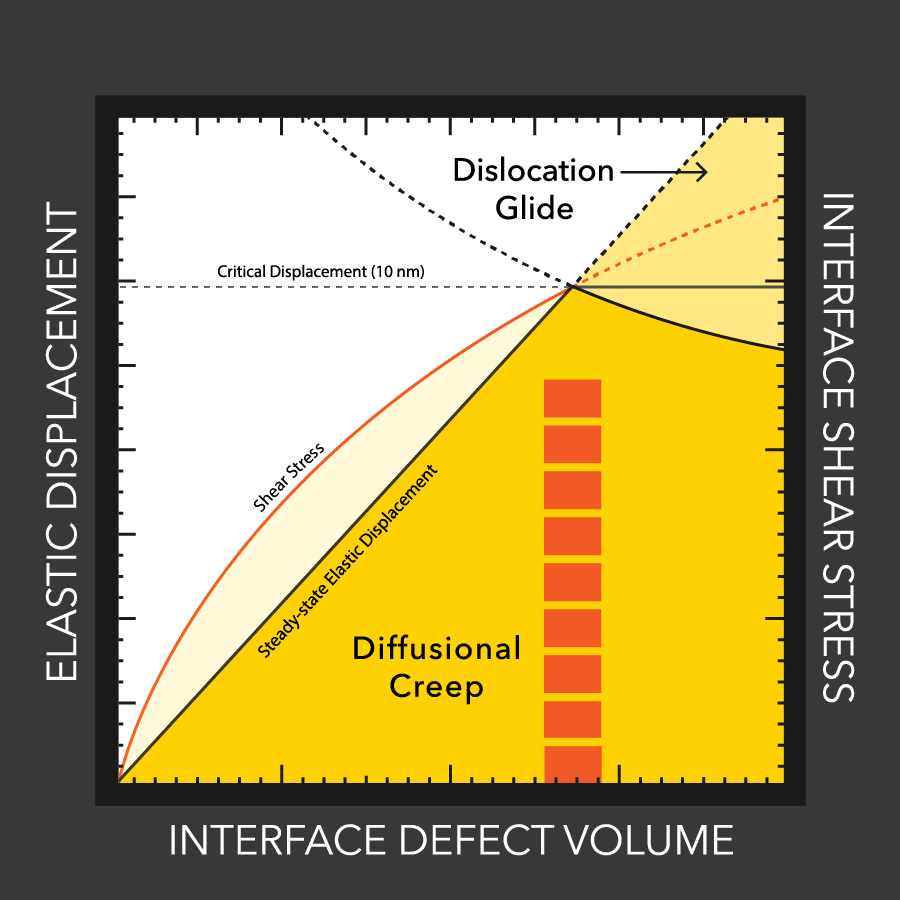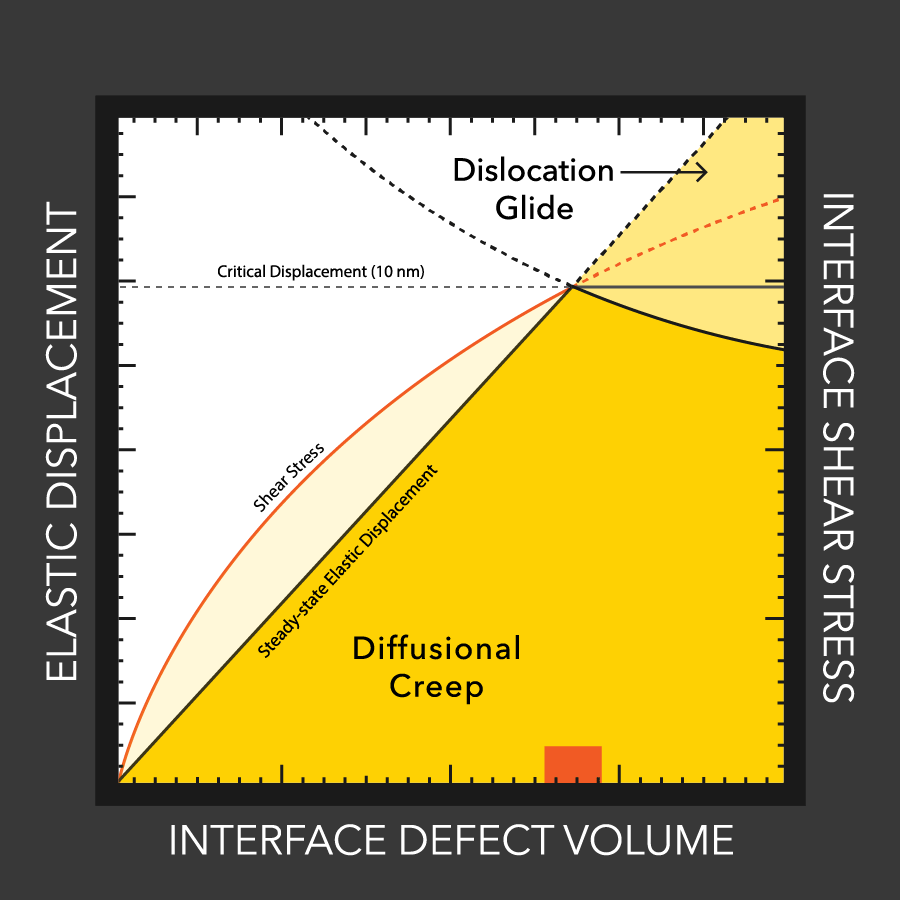
MTU engineers examine lithium battery defects
Lithium dendrites cause poor performance and even explosions in batteries with flammable liquid electrolytes. How these dendrites grow, even with a solid electrolytes, is still a mystery, but materials engineers at MTU and Oak Ridge study the conditions that enable dendrites and how to stop them.

Using High Energy Density Material in Electrode Design Enhances Lithium Sulfur Batteries
To develop higher capacity batteries, researchers have looked to lithium sulfur batteries because of sulfur’s high theoretical capacity and energy density. But there are still several problems to solve before they can be put into practical applications. The biggest is the shuttling effect that occurs during cycling. To solve this problem and improve lithium sulfur battery performance, the researchers created a sandwich-structured electrode using a novel material that traps polysulfides and increases the reaction kinetics.
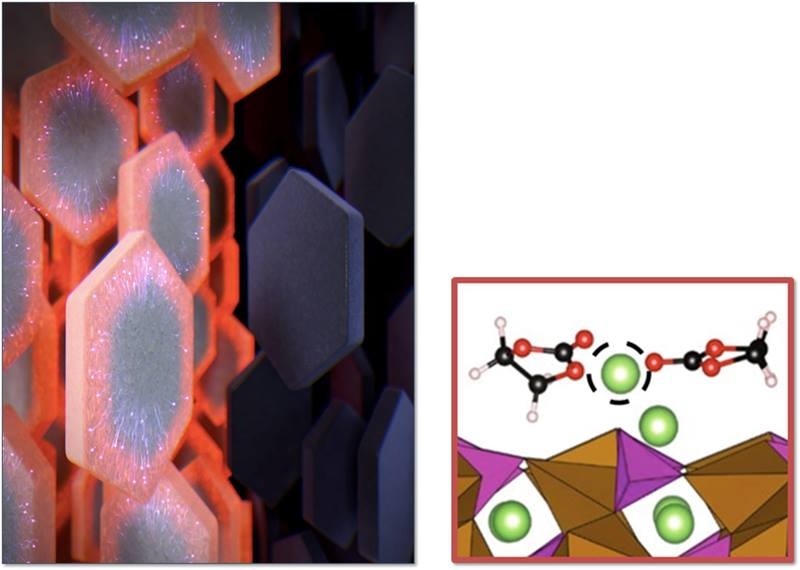
End-run Spreads Lithium Throughout Battery Electrodes
Scientists used chemically sensitive X-ray microscopy to map lithium transport during battery operation.