The reactivity of zirconium on silicon nitride enhances the conversion of propane into propylene, a key commodity chemical needed to make polypropylene. This finding hints at the reactivity researchers might achieve with other nontraditional catalysts.
Tag: Catalysis
Not all calcite crystals are as perfect as they appear
Researchers used complementary imaging techniques to explore the impact of synthesis approaches on the nanoscopic internal structure of apparently perfect calcite particles, which has implications for contaminant sequestration and carbon storage.
Scientists Make and Test Efficient Water-Splitting Catalyst Predicted by Theory
Scientists have developed a new efficient catalyst for the most challenging part of “water splitting,” a series of two simultaneous electrochemical reactions that generate hydrogen gas, a green energy source, from water. The new catalyst was designed based on theoretical predictions and validated in laboratory tests and industrially relevant demonstrations.
Study Reveals Reversible Assembly of Platinum Catalyst
Chemists at the U.S. Department of Energy’s Brookhaven National Laboratory, Stony Brook University (SBU), and their collaborators have uncovered new details of the reversible assembly and disassembly of a platinum catalyst. The new understanding may offer clues to the catalyst’s stability and recyclability.
A Cleaner Way to Produce Ammonia
Ammonia is the starting point for the fertilizers that have secured the world’s food supply for the last century. It’s also a main component of cleaning products, and is even considered as a future carbon-free replacement for fossil fuels in vehicles.
How Scientists Are Accelerating Chemistry Discoveries With Automation
Researchers have developed an automated workflow that could accelerate the discovery of new pharmaceutical drugs and other useful products. The new approach could enable real-time reaction analysis and identify new chemical-reaction products much faster than current laboratory methods.
Recyclable Reagent and Sunlight Convert Carbon Monoxide into Methanol
Scientists at the U.S. Department of Energy’s (DOE) Brookhaven National Laboratory and the University of North Carolina Chapel Hill (UNC) have demonstrated the selective conversion of carbon dioxide (CO2) into methanol using a cascade reaction strategy. The two-part process is powered by sunlight, occurs at room temperature and at ambient pressure, and employs a recyclable organic reagent that’s similar to a catalyst found in natural photosynthesis.
Novel catalyst for green production of fine chemicals and pharmaceuticals
Scientists from the National University of Singapore have developed an innovative catalyst that achieves a significantly lower carbon footprint, paving the way for greener chemical and pharmaceutical manufacturing processes.
Novel ligands for transition-metal catalysis of photoreactions
Transition metals form catalytic complexes that can speed up various chemical processes, especially in the production of pharmaceuticals as well as various pigments, dyes, and laboratory reagents like sulfuric acid.
Researchers “film” novel catalyst at work
A novel catalysis scheme enables chemical reactions that were previously virtually impossible. The method developed at the University of Bonn is also environmentally friendly and does not require rare and precious metals.
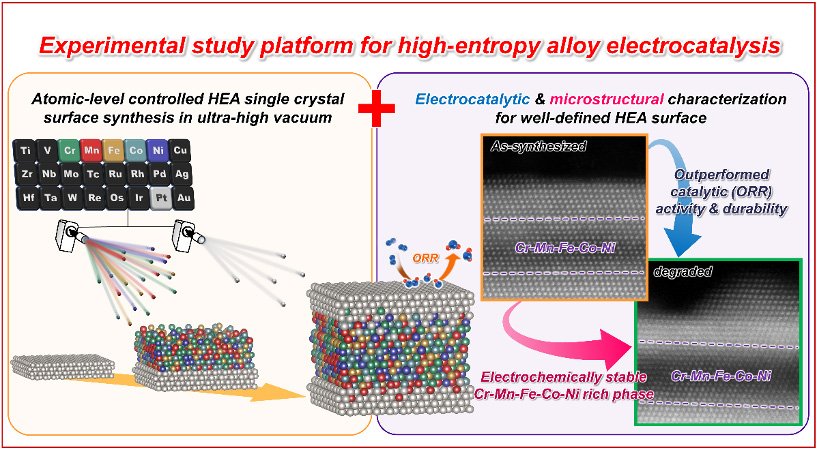
Researchers reveal a powerful platform for studying high-entropy alloy electrocatalysis
Introduced in 2004, high-entropy alloys (HEAs) are alloys composed of multiple principal elements in nearly equiatomic proportions.
A Nickle-Platinum Nanoscale Core with a Platinum Shell Cracks Oxygen Molecules into Useful Ions
Researchers have modified the surface of nickel-platinum nanoparticles to improve their ability to act as catalysts to make reactive oxygen ions. Using a specialized X-ray scattering imaging technique, the researchers examined the modified nanoparticles and discovered a platinum-rich outer layer.
Burning Calories for Energy in the Calorimetry Lab
Research in the Energy Sciences Center explores how heat changes in chemical reactions, paving the way for more efficient fuels and processes.
New catalyst could dramatically cut methane pollution from millions of engines
Today’s catalysts for removing unburnt methane from natural-gas engine emissions are either inefficient at low, start-up temperatures or break down at higher operating temperatures. A new single-atom catalyst solves both these problems and removes 90% of the methane.

A Baking Soda Solution for Clean Hydrogen Storage
PNNL scientists investigate the promising properties of a common, Earth-abundant salt.
Chemists Unravel Reaction Mechanism for Clean Energy Catalyst
Chemists at the University of Kansas and Brookhaven National Laboratory have unraveled the entire reaction mechanism for a key class of water-splitting catalysts. Their work could help pure hydrogen be produced from renewable energy sources such as solar power.
Scientists Use Peroxide to Peer into Metal Oxide Reactions
Researchers at Binghamton University led research partnering with the Center for Functional Nanomaterials (CFN)—a U.S. Department of Energy (DOE) Office of Science User Facility at Brookhaven National Laboratory—to get a better look at how peroxides on the surface of copper oxide promote the oxidation of hydrogen but inhibit the oxidation of carbon monoxide, allowing them to steer oxidation reactions.
Energy-efficient and customisable inorganic membranes for a cleaner future
A team of researchers from the College of Design and Engineering at the National University of Singapore, led by Professor Ho Ghim Wei from the Department of Electrical and Computer Engineering, has developed a revolutionary technique for producing ultrathin inorganic membranes.
What can we do about all the plastic waste?
The Institute for the Cooperative Upcycling of Plastics (iCOUP) is helping to address the plastic waste accumulation problem by developing the science needed to turn used plastic into valuable materials.

Machine learning model speeds up assessing catalysts for decarbonization technology from months to milliseconds
Argonne researchers have developed an artificial intelligence-based model to greatly speed up the process for engineering a low-cost catalyst that converts biomass into fuels and useful chemicals with many possible applications.
Three Argonne researchers inducted into AAAS
John Mitchell, Valerie Taylor and Lisa Utschig were selected by the American Association for the Advancement of Science (AAAS) to be inducted as fellows.
New enzyme could mean better drugs
Just as a choreographer’s notation tells a dancer to strike a particular pose, an enzyme newly discovered by Rice University scientists is able to tell specific molecules precisely how to arrange themselves, down to the angle of single hydrogen bonds.
Chemical researchers discover catalyst to make renewable paints, coatings, and diapers
A team led by University of Minnesota Twin Cities researchers has invented a groundbreaking new catalyst technology that converts renewable materials like trees and corn to the key chemicals, acrylic acid, and acrylates used in paints, coatings, and superabsorbent polymers.
Media Tip: Scientists enhance recyclability of post-consumer plastic
Scientists at the U.S. Department of Energy’s (DOE) Institute for Cooperative Upcycling of Plastics (iCOUP) have developed a new method for recycling high-density polyethylene (HDPE).
Recycled gold from SIM cards could help make drugs more sustainable
Researchers have used gold extracted from electronic waste as catalysts for reactions that could be applied to making medicines.
Argonne researchers awarded $3.8 million to study clean energy
Argonne chemist Karen Mulfort and her research team were awarded $3.8 million across three years to study clean energy.
Looking at oxygen storage dynamics in three-way catalysts
In light of vehicular pollutants contributing to decreasing air quality, governments across the globe are posing stricter emission regulations for automobiles.
Light of transformation: Research explores the inner workings of chemical change
Binghamton University Assistant Professor of Chemistry Jennifer Hirschi recently received a Maximizing Investigators’ Research Award for $1.93 million from the National Institute of General Medical Sciences to study the mechanisms involved in catalytic reactions.

Entrepreneurship program at Argonne National Laboratory opens applications for startups
Chain Reaction Innovations, the entrepreneurship program at Argonne National Laboratory, is accepting applications for its next fellowship cohort.
Emerging Methods for Recycling Plastics Address Barriers, Opportunities
Understanding the fundamentals of these emerging technologies will help design improved systems for chemical recycling and upcycling of waste plastics.
Fueling your curiosity: Argonne answers top questions on hydrogen fuel
As part of National Hydrogen and Fuel Cell Day, Argonne answers common questions surrounding hydrogen as an energy carrier.
Argonne researchers win four 2022 R&D 100 awards
R&D Magazine has recognized four Argonne projects with R&D 100 Awards.
Plastic Upcycling: From Waste to Fuel for Less
Plastic upcycling efficiently converts plastics to valuable commodity chemicals while using less of the precious metal ruthenium. The method could recycle waste plastic pollution into useful products, helping keep it out of landfills.
Enriching Science Education with Thin Films
Tiffany Kaspar’s work has advanced the discovery and understanding of oxide materials, helping develop electronics, quantum computing, and energy production. She strives to communicate her science to the public.
Washington State Academy of Sciences Adds Six PNNL Researchers
The Washington State Academy of Sciences added six people from PNNL to its 2022 class of inductees.
Found: The ‘holy grail of catalysis’ — turning methane into methanol under ambient conditions using light
An international team of researchers, led by scientists at the University of Manchester, has developed a fast and economical method of converting methane, or natural gas, into liquid methanol at ambient temperature and pressure. The method takes place under continuous flow over a photo-catalytic material using visible light to drive the conversion.
To help observe how the process works and how selective it is, the researchers used neutron scattering at the VISION instrument at Oak Ridge National Laboratory’s Spallation Neutron Source.
Converting Methane to Methanol—With and Without Water
Adding water to the catalytic reaction that converts methane into useful methanol makes the process more effective, but it creates challenges for industry due to steam from the water. Now scientists have identified a common industrial catalyst, copper-zinc oxide, that completes the conversion along different pathways depending on whether water is present or not. This could potentially keep methane, a potent greenhouse gas, out of Earth’s atmosphere and instead turn it into useful products.
Chemical Institute of Canada Gives Top Honor to University of Oklahoma Engineering Professor
The 2022 Robert B. Anderson Catalysis Award from the Chemical Institute of Canada’s Catalysis Division was presented to University of Oklahoma engineering professor Daniel Resasco, Ph.D., for his research that deepens the understanding of chemical reactions in the production of sustainable energy.

Machine Learning Framework IDs Targets for Improving Catalysts
Chemists at the U.S. Department of Energy’s Brookhaven National Laboratory have developed a new machine-learning (ML) framework that can zero in on which steps of a multistep chemical conversion should be tweaked to improve productivity. The approach could help guide the design of catalysts — chemical “dealmakers” that speed up reactions.
Seeking Energy Frontiers
Morris Bullock has led PNNL’s pursuit of the efficient conversion of electrical energy and chemical bonds through control of electron and proton transfers.
Converting Methane to Methanol — With and Without Water
UPTON, NY — Chemists have been searching for efficient catalysts to convert methane — a major component of abundant natural gas — into methanol, an easily transported liquid fuel and building block for making other valuable chemicals. Adding water to the reaction can address certain challenges, but it also complicates the process.
Energy Secretary, Washington Governor Dedicate Energy Sciences Center at PNNL
Fundamental research conducted at the $90-million research facility will help the nation meet its clean energy goals.
Catalyst Study Advances Carbon-Dioxide-to-Ethanol Conversion
An international collaboration of scientists has taken a significant step toward the realization of a nearly “green” zero-net-carbon technology that will efficiently convert carbon dioxide, a major greenhouse gas, and hydrogen into ethanol, which is useful as a fuel and has many other chemical applications.
Combining sunlight and wastewater nitrate to make the world’s No. 2 chemical
Engineers at the University of Illinois Chicago have created a solar-powered electrochemical reaction that not only uses wastewater to make ammonia — the second most-produced chemical in the world — but also achieves a solar-to-fuel efficiency that is 10 times better than any other comparable technology.
A new approach creates an exceptional single-atom catalyst for water splitting
Anchoring individual iridium atoms on the surface of a catalyst made them a lot better at splitting water – a reaction that’s been a bottleneck in making sustainable energy production more competitive.
To Reduce Vehicle Pollution, a Single Atom Can Do the Work of Several
A discovery from PNNL and Washington State University could help reduce the amount of expensive material needed to treat vehicle exhaust by making the most of every precious atom.
Scientists show a single catalyst can perform the first step of turning CO2 into fuel in two very different ways
Scientists at Stanford and SLAC made a new catalyst that works with either heat or electricity to accelerate a reaction for turning carbon dioxide into carbon monoxide. It’s an important step toward unifying the understanding of catalytic reactions in these two very different conditions.
Green Diesel for the Road Ahead
A new report led by PNNL identifies the top 13 most promising waste- and biomass-derived diesel blendstocks for reducing greenhouse gas emissions, other pollutants, and overall system costs.
Worldly Experience Is a Catalyst for Change
Bojana Ginovska leads a physical biosciences research team headed for PNNL’s new Energy Sciences Center. She uses the transformative power of molecular catalysis and enzymes to explore scientific principles.
Water Clusters Control Zeolite Reactivity
Computational results clarify the role water plays in the reactivity of a widely used class of porous oxide catalysts.