Scientists at the U.S. Department of Energy’s (DOE) Brookhaven National Laboratory and the University of North Carolina Chapel Hill (UNC) have demonstrated the selective conversion of carbon dioxide (CO2) into methanol using a cascade reaction strategy. The two-part process is powered by sunlight, occurs at room temperature and at ambient pressure, and employs a recyclable organic reagent that’s similar to a catalyst found in natural photosynthesis.
Tag: artificial photosynthesis
Using photosynthesis for Martian occupation – while making space travel more sustainable
In a study published in Nature Communications, scientists assess a new technique which could convert renewable, green energy from outside the Earth’s atmosphere. They are taking advantage of photosynthesis – the chemical process plants undergo every day to create energy – to help the space industry become more sustainable.

Researchers capture elusive missing step in the final act of photosynthesis
Photosynthesis plays a crucial role in shaping and sustaining life on Earth, yet many aspects of the process remain a mystery. One such mystery is how Photosystem II, a protein complex in plants, algae and cyanobacteria, harvests energy from sunlight and uses it to split water, producing the oxygen we breathe. Now researchers from the Department of Energy’s Lawrence Berkeley National Laboratory and SLAC National Accelerator Laboratory, together with collaborators from Uppsala University and Humboldt University and other institutions have succeeded in cracking a key secret of Photosystem II.
How a Record-Breaking Copper Catalyst Converts CO2 Into Liquid Fuels
Since the 1970s, scientists have known that copper has a special ability to transform carbon dioxide into valuable chemicals and fuels. But for many years, scientists have struggled to understand how this common metal works as an electrocatalyst, a mechanism that uses energy from electrons to chemically transform molecules into different products.
How a soil microbe could rev up artificial photosynthesis
When it comes to fixing carbon, plants have nothing on soil bacteria that can do it 20 times faster. The secret is an enzyme that “juggles” reaction ingredients. Scientists hope to optimize this process for producing fuels, antibiotics and other products from CO2.
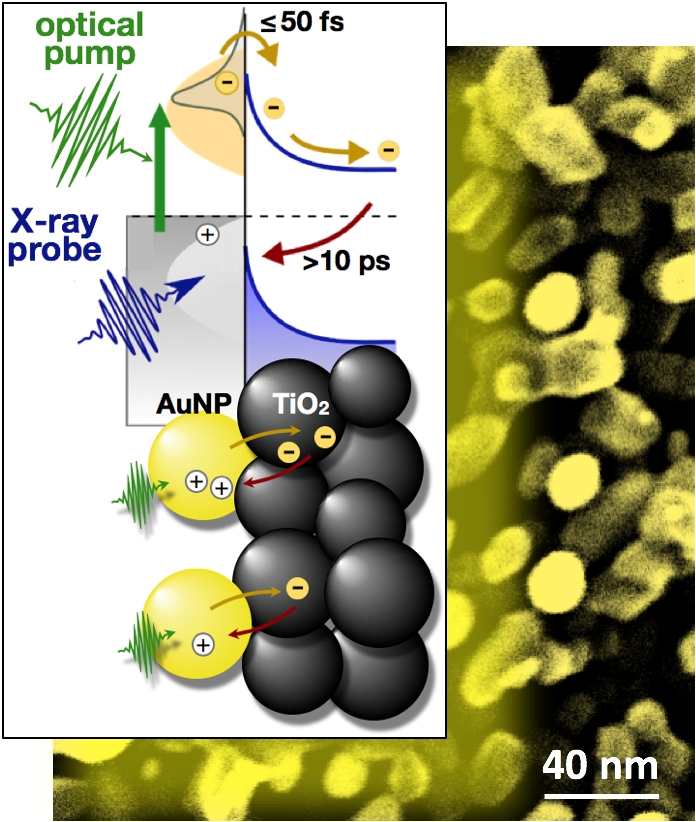
Scientists Capture Candid Snapshots of Electrons Harvesting Light at the Atomic Scale
A team of scientists led by Berkeley Lab has gained important new insight into electrons’ role in the harvesting of light in artificial photosynthesis systems.
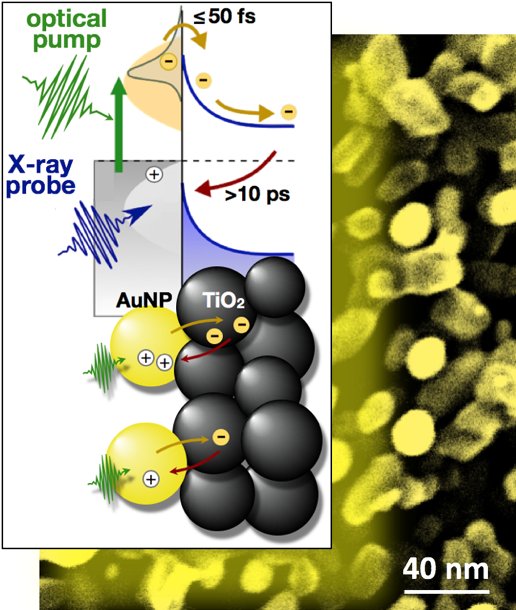
Watching Electrons Harvest Light at the Nanoscale
Mixtures of nanoparticles show promise for solar fuels production. For example, gold donor particles absorb sunlight well, but they need an acceptor material to efficiently make fuel. Now, scientists have found a way to count electrons as they transfer between the two materials during these chemical reactions.
Brookhaven Lab Partners in New $40 M Research Center to Convert Sunlight to Liquid Fuels
UPTON, NY–The U.S. Department of Energy (DOE) has announced $40M in funding over five years for a new research center aimed at developing hybrid photoelectrodes for converting sunlight into liquid fuels. Chemists from DOE’s Brookhaven National Laboratory will be key partners in this effort, dubbed the Center for Hybrid Approaches in Solar Energy to Liquid Fuels (CHASE), which will be led by the University of North Carolina at Chapel Hill (UNC) and includes additional collaborators at Emory University, North Carolina State University, the University of Pennsylvania, and Yale.

Berkeley Lab Part of Multi-Institutional Team Awarded $60M for Solar Fuels Research
The Department of Energy has awarded $60 million to a new solar fuels initiative – called the Liquid Sunlight Alliance (LiSA) – led by Caltech in close partnership with Berkeley Lab. LiSA will build on the foundational work of the Joint Center for Artificial Photosynthesis (JCAP).

The Secret to Renewable Solar Fuels is an Off-and-On Again Relationship
Copper that was once bound with oxygen is better at converting carbon dioxide into renewable fuels than copper that was never bound to oxygen, according to scientists at Berkeley Lab and Caltech.

Nature-Inspired Green Energy Technology Clears Important Development Hurdle
A new design has put the long-sought idea of artificial photosynthesis within reach

How JCAP Is Making Solar Fuels Shine
As we look back at a decade of discovery, we highlight 10 achievements by scientists at Berkeley Lab and the Joint Center for Artificial Photosynthesis that bring us closer to a solar fuels future.
Department of Energy to Provide $100 Million for Solar Fuels Research
The U.S. Department of Energy (DOE) announced a plan to provide up to $100 million over five years for research on artificial photosynthesis for the production of fuels from sunlight.