Switching to ammonia as a marine fuel, with the goal of decarbonisation, can instead create entirely new problems. This is shown in a study from Chalmers University of Technology in Sweden, where researchers carried out life cycle analyses for batteries and for three electrofuels including ammonia.
Tag: Hydrogen Fuel
Scientists Probe the Source of Stochastic Occurrence of Super-Knock in Engines Running on Hydrogen/Methane Fuels
Blends of hydrogen and methane are a promising alternative vehicle fuel that could help reduce carbon dioxide emissions. To make these fuels viable, researchers need to understand how they burn, especially in small, turbocharged internal combustion engines. In this study, researchers examined the impact of non-thermal chemical kinetics on “super-knock,” a combustion mechanism that can cause severe engine damage.
Transforming ‘sewer gas’ into clean hydrogen fuel
Scientists have found a new chemical process to turn a stinky, toxic gas into a clean-burning fuel.
Science Snapshots
Science Snapshots from Berkeley Lab on forest soils, vaping cannabis risks, reusable PPE, hydrogen fuel generation
First nanoscale look at a reaction that limits the efficiency of generating clean hydrogen fuel
Transitioning to a hydrogen economy will require massive production of cheap, clean hydrogen gas for fuel and chemical feedstocks. New tools allow scientists to zoom in on a catalytic reaction that’s been a bottleneck in efforts to generate hydrogen from water more efficiently.
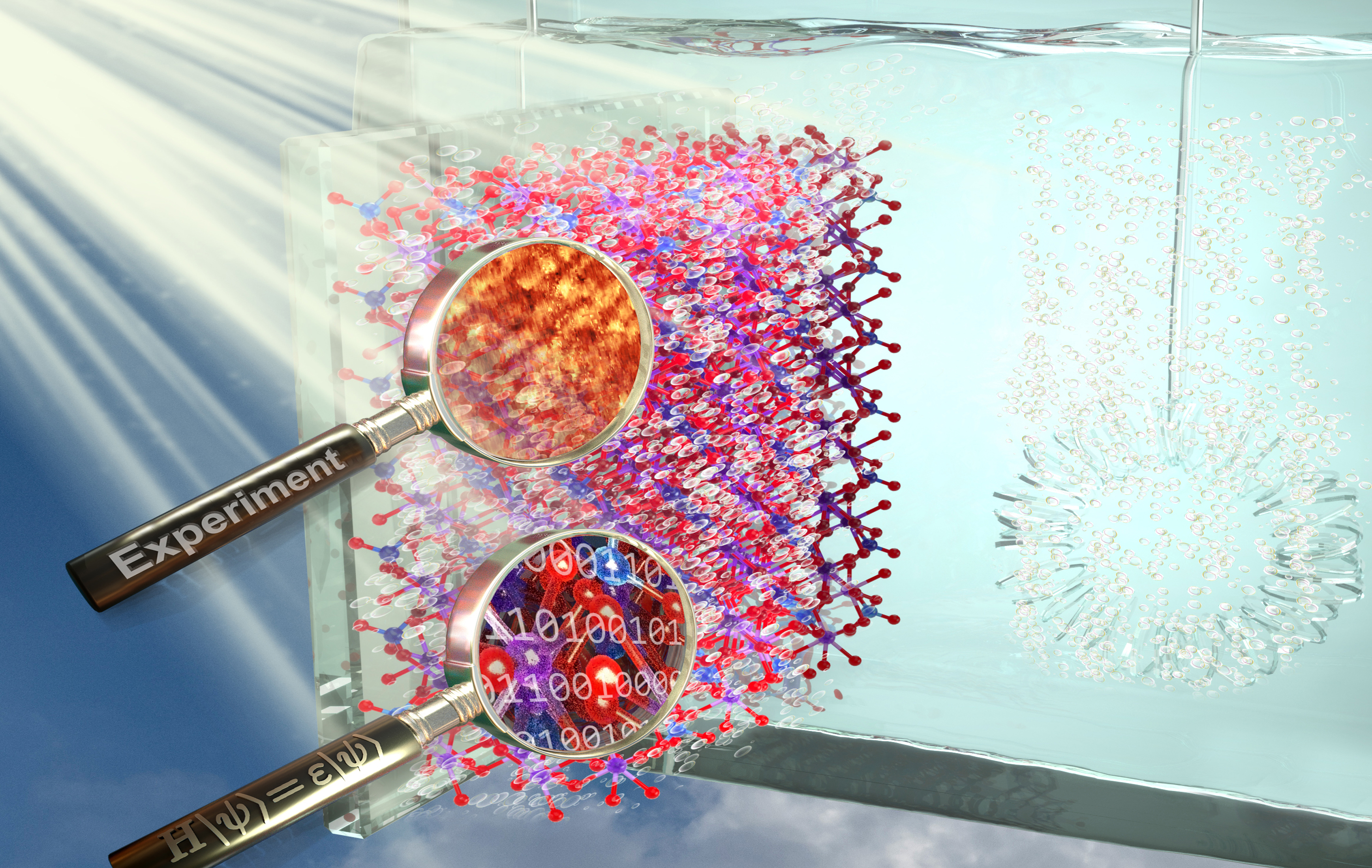
Tuning Electrode Surfaces to Optimize Solar Fuel Production
Changing the topmost layer of atoms on electrode surfaces can impact the activity of splitting water into oxygen and hydrogen—a clean fuel.
Study Reveals Platinum’s Role in Clean Fuel Conversion
Scientists at the U.S. Department of Energy’s Brookhaven National Laboratory, Stony Brook University (SBU), and other collaborating institutions have uncovered dynamic, atomic-level details of how an important platinum-based catalyst works in the water gas shift reaction. The experiments provide definitive evidence that only certain platinum atoms play an important role in the chemical conversion, and could therefore guide the design of catalysts that use less of this precious metal.
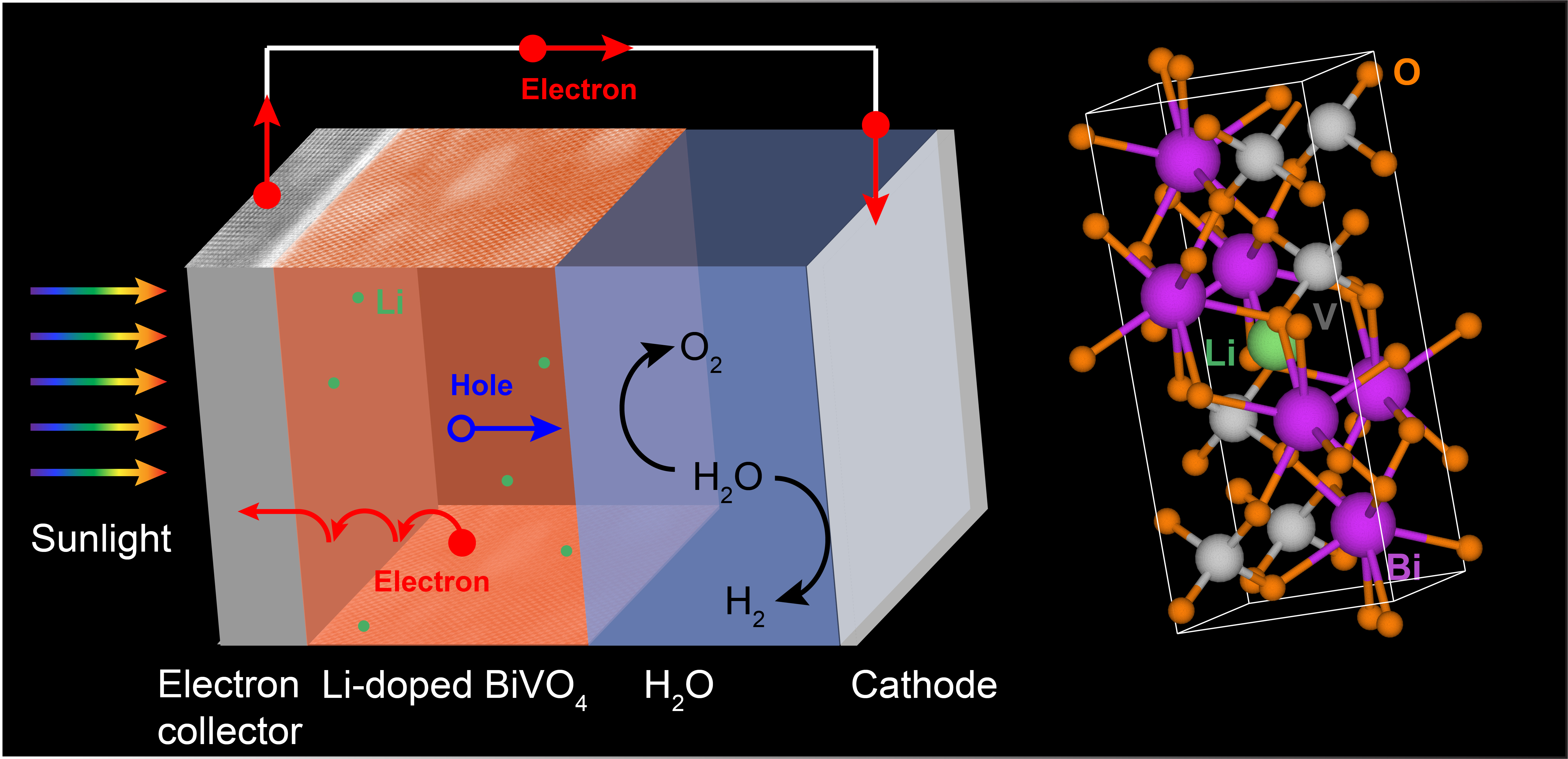
Driving Water Splitting to Create Chemical Fuels
Scientists improved the performance of bismuth vanadate, an electrode material for converting solar energy to hydrogen—an energy-dense and clean-burning fuel.

Berkeley Lab Part of Multi-Institutional Team Awarded $60M for Solar Fuels Research
The Department of Energy has awarded $60 million to a new solar fuels initiative – called the Liquid Sunlight Alliance (LiSA) – led by Caltech in close partnership with Berkeley Lab. LiSA will build on the foundational work of the Joint Center for Artificial Photosynthesis (JCAP).