In comparison with conventional pyrolysis processes, the researchers developed a rapid thermal shock method to achieve a uniform dispersion of metal nanoparticles on carbon supports. This approach effectively prevented nanoparticle aggregation, ensuring the full utilization of active sites.
Tag: Electrocatalysis
X-ray Spectral Microscopy Reveals The Active Edges of a Water-Splitting Material
Splitting water into hydrogen and oxygen is a key process for energy storage. The chemical transitions involved in splitting water require energy, so researchers are designing more efficient new electrodes with energy saving catalytic properties.
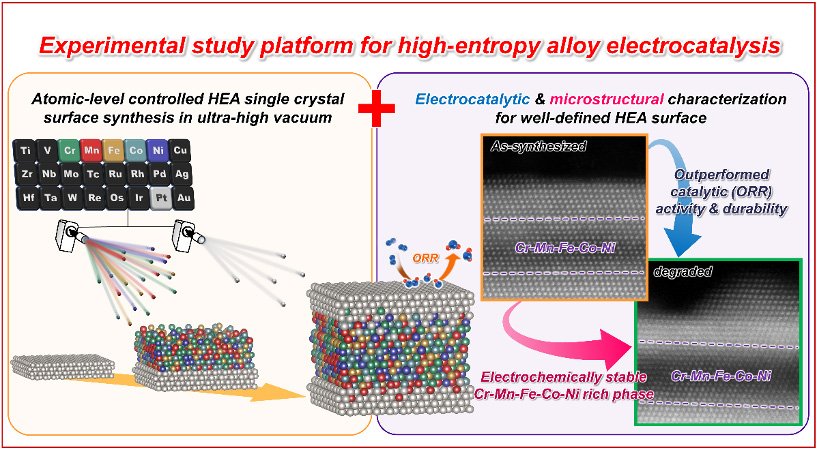
Researchers reveal a powerful platform for studying high-entropy alloy electrocatalysis
Introduced in 2004, high-entropy alloys (HEAs) are alloys composed of multiple principal elements in nearly equiatomic proportions.
How a Record-Breaking Copper Catalyst Converts CO2 Into Liquid Fuels
Since the 1970s, scientists have known that copper has a special ability to transform carbon dioxide into valuable chemicals and fuels. But for many years, scientists have struggled to understand how this common metal works as an electrocatalyst, a mechanism that uses energy from electrons to chemically transform molecules into different products.
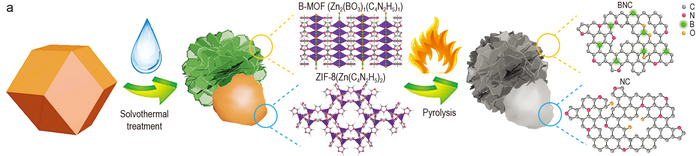
A Janus carbon electrocatalyst can balance the intrinsic activity and electronic conductivity
Carbon-based electrocatalysts are considered as promising alternatives to the state-of-the-art precious metal catalysts.
Expanded Partnership Helps Secure Texas Tech’s Place in Energy Industry’s Future
American Resources Corporation will operate its sponsored research program in electrolytic cells through the Innovation Hub at Research Park and in collaboration with Gerardine Botte.
First nanoscale look at a reaction that limits the efficiency of generating clean hydrogen fuel
Transitioning to a hydrogen economy will require massive production of cheap, clean hydrogen gas for fuel and chemical feedstocks. New tools allow scientists to zoom in on a catalytic reaction that’s been a bottleneck in efforts to generate hydrogen from water more efficiently.