Abstract The ammonia industry plays a crucial role in various aspects of human progress and the global economy, particularly in fields such as fertilizers and the production of emerging clean fuels. The Haber-Bosch process, a commercially used method for ammonia…
Tag: Ammonia
A Cleaner Way to Produce Ammonia
Ammonia is the starting point for the fertilizers that have secured the world’s food supply for the last century. It’s also a main component of cleaning products, and is even considered as a future carbon-free replacement for fossil fuels in vehicles.
Ammonia reduction should be prioritized in Europe’s fight against air pollution
A new study identified the reduction of ammonia emissions as a cost-effective measure to reduce concentrations of fine particulate matter concentrations in the atmosphere.
Cutting ammonia emissions is a cost-effective way to prevent air pollution deaths
Tackling pollution from the emission of nitrogen compounds, particularly ammonia, could reduce many of the 23.3 million years of life that were lost prematurely across the world in 2013 due to nitrogen-related air pollution.
Combining sunlight and wastewater nitrate to make the world’s No. 2 chemical
Engineers at the University of Illinois Chicago have created a solar-powered electrochemical reaction that not only uses wastewater to make ammonia — the second most-produced chemical in the world — but also achieves a solar-to-fuel efficiency that is 10 times better than any other comparable technology.
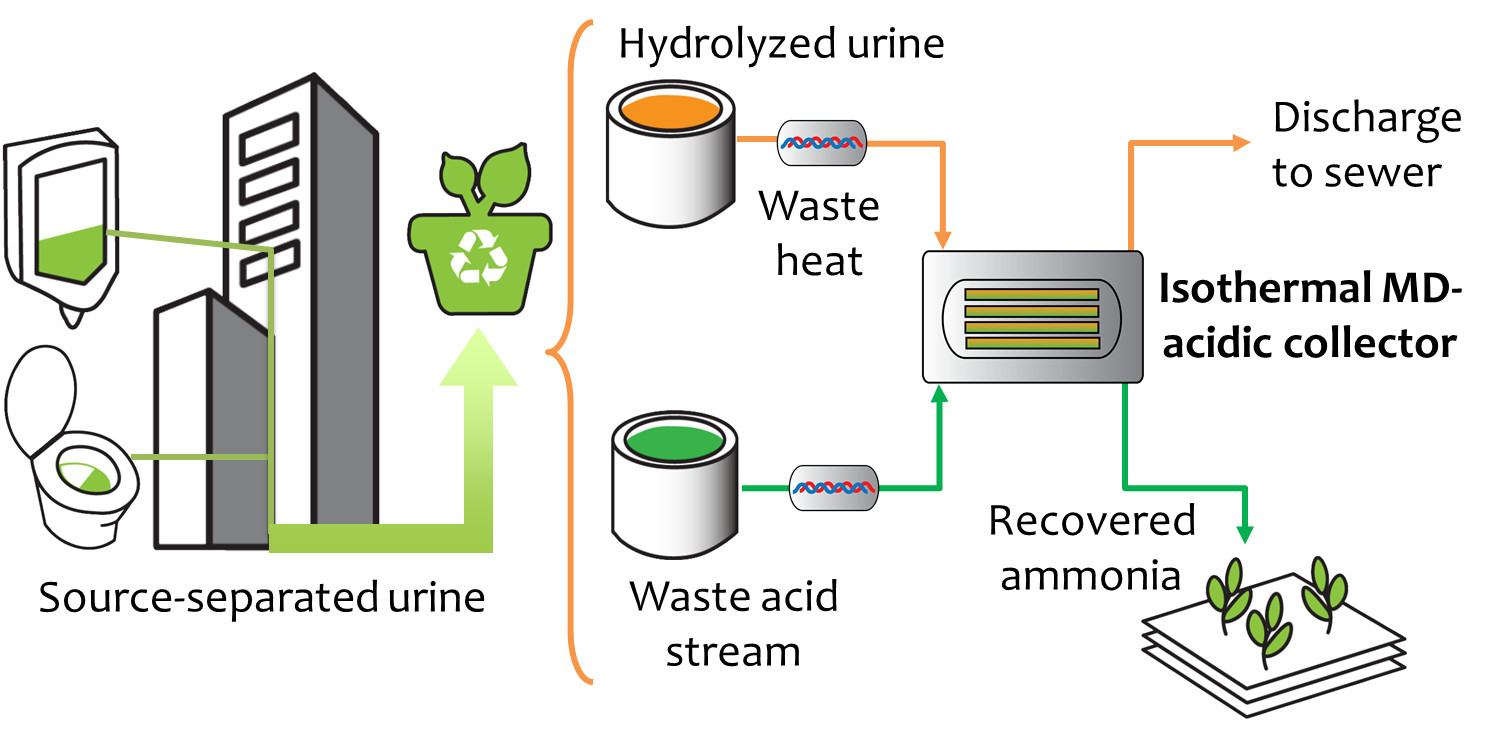
Sustainable Recovery of Nutrients from Urine
Most ammonia capture is done through the Haber-Bosch (HB) process, an energy-intensive technique used to produce fertilizer that accounts for 1-2% of the world’s annual energy consumption. Columbia engineers report they have recovered ammonia through a new method with a very low level of energy, approx 1/5 of the energy used by HB. And because the technique recycles ammonia in a closed loop, the ammonia can be recaptured for reuse in fertilizer, household cleaners, etc.
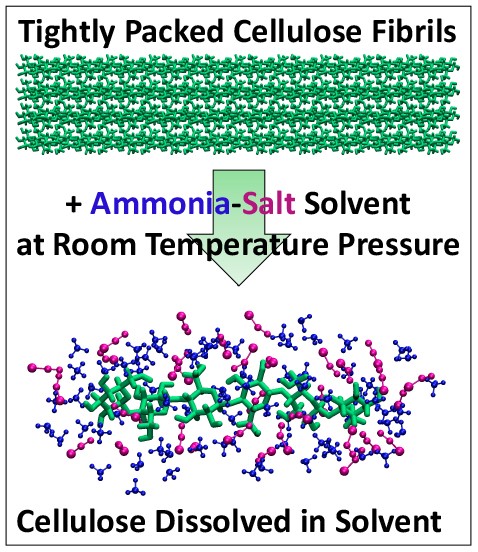
How to Make it Easier to Turn Plant Waste into Biofuels
Researchers have developed a new process that could make it much cheaper to produce biofuels such as ethanol from plant waste and reduce reliance on fossil fuels. Their approach, featuring an ammonia-salt based solvent that rapidly turns plant fibers into sugars needed to make ethanol, works well at close to room temperature, unlike conventional processes, according to a Rutgers-led study in the journal Green Chemistry.