The Wistar Institute is pleased to announce a research collaboration with Accelerated Biosciences Corp. aimed at creating a platform based on human trophoblast stem cells to explore new immunotherapies that use Accelerated Bio’s proprietary technology.
Tag: Immunotheapy
Oral Probiotic Delivers Colitis Treatment Directly to Gut in Multiple Animal Models
Article title: Oral administration of CXCL12-expressing Limosilactobacillus reuteri improves colitis by local immunomodulatory actions in preclinical models Authors: Emelie Öhnstedt, Cristian Doñas, Kristel Parv, Yanhong Pang, Hava Lofton Tomenius, Macarena Carrasco López, Venkata Ram Gannavarapu, Jacqueline Choi, Maria Ovezik, Peter Frank, Margareth…

Roswell Park Insights on Role of Exportin 1 Protein Suggest Strategy for Improving Cancer Immunotherapy
Research by Hemn Mohammadpour, DVM, PhD, and colleagues at Roswell Park Comprehensive Cancer Center offers new insights into tumor biology and may lay the groundwork for more effective cancer immunotherapy. Their preclinical findings were published today in the journal Cellular & Molecular Immunology.
Researchers develop a new way to safely boost immune cells to fight cancer
Researchers in the College of Engineering explore a cancer immunotherapy treatment that involves activating the immune cells in the body and reprogramming them to attack and destroy cancer cells. This therapeutic method frequently uses cytokines, small protein molecules that act as intercellular biochemical messengers and are released by the body’s immune cells to coordinate their response.
New cell-based immunotherapy offered for melanoma
Siteman Cancer Center, based at Barnes-Jewish Hospital and Washington University School of Medicine in St. Louis, is one of the first centers nationwide to offer a newly approved cell-based immunotherapy that targets melanoma.
UC San Diego Awarded $8M to Expand Stem Cell Therapy Clinical Trials
UC San Diego Alpha Stem Cell Clinic awarded $8M to expand clinical trials of novel stem cell therapies. The CIRM award will advance partnerships between academic and industry experts in San Diego to expedite clinical trials for patients with difficult-to-treat diseases.
Cancer Patients Treated with Immunotherapy Can Safely Receive mRNA COVID-19 Vaccines, According to JNCCN Study
New research published in the October 2022 issue of JNCCN—Journal of the National Comprehensive Cancer Network confirms the safety of mRNA vaccines in people with cancer undergoing immunotherapy treatment.
Mount Sinai Researcher Receives Prestigious Award From the American Association of Indian Scientists in Cancer Research
Nina Bhardwaj, MD, PhD, Director of Immunotherapy at The Tisch Cancer Institute at Mount Sinai, has received the 2022 Lifetime Achievement in Cancer Research award from the American Association of Indian Scientists in Cancer Research (AAISCR).
Chula Excellence Cancer Center collabs with medical specialists from various fields to enhance treatment capabilities.
Chula now has an Excellence Chulalongkorn Comprehensive Cancer Center bringing together medical specialists from various areas of expertise to attend to cancer patients using the latest academic and technological know-how to enhance the quality of life and the possibilities of being cured for patients of all types of cancer.
Therapy Using Dual Immune System Cells Effectively Controls Neuroblastoma
A newly developed immunotherapy that simultaneously uses modified immune-fighting cells to home in on and attack two antigens, or foreign substances, on cancer cells was highly effective in mice implanted with human neuroblastoma tissue.
Roswell Park and University of Chicago Medicine Comprehensive Cancer Centers Awarded Nearly $9M for Ovarian Cancer Research
For years, scientists at Roswell Park Comprehensive Cancer Center and the University of Chicago Medicine Comprehensive Cancer Center have devoted themselves to research to better understand ovarian cancer.
First in the World! Chulalongkorn Hospital Successfully Treats a Breast Cancer Patient with Immunotherapy
Queen Sirikit Center for Breast Cancer, King Chulalongkorn Memorial Hospital, Thai Red Cross Society (Chulalongkorn Hospital) has become the world’s first institution to have successfully used immunotherapy to treat a breast cancer patient who is now in complete remission with minimal side effects and uplifted quality of life.
Dual Immunotherapy Regimen Delays Cancer Progression in Patients with Advanced Melanoma
A treatment regimen for patients with advanced melanoma that combines the immunotherapy agents relatlimab (anti-LAG-3) and nivolumab (anti-PD-1) delayed time to cancer progression significantly more than nivolumab alone, according to results of a study to be presented June 6 at the 2021 American Society of Clinical Oncology (ASCO) annual meeting.
Ludwig Lausanne Receives Team Science Award from the Society for Immunotherapy of Cancer
Ludwig Cancer Research is proud to announce that the Lausanne Branch of the Ludwig Institute for Cancer Research has received a Team Science Award from the Society for Immunotherapy of Cancer (SITC).
MD Anderson and Allogene Therapeutics announce strategic collaboration to accelerate advancement of allogeneic CAR T therapy (AlloCAR T)
MD Anderson and Allogene today announced a strategic five-year collaboration for preclinical and clinical investigation of allogeneic CAR T cell therapies.

Ludwig Study Finds a Common Nutritional Supplement Might Boost the Effects of Cancer Immunotherapy
A Ludwig Cancer Research study has uncovered a mechanism by which the tumor’s harsh internal environment sabotages T lymphocytes, leading cellular agents of the anticancer immune response.

Hackensack Meridian CDI Scientists Find One-Two Punch Working against Stubborn Pancreatic Cancer in Preclinical Models
Changing gene expression, then deploying immune checkpoint inhibitors, shows promise in battling one of the most treatment-resistant types of cancer in preclinical models
This MicroRNA Might Help Detect, Treat Ovarian Cancer
In cell and mouse models, one microRNA showed promise as a biomarker for early stage ovarian cancer and may help make immunotherapy treatment more effective.

Targeted Drugs and Immunotherapies May Lower Risk of Therapy-Related Hematologic Cancers
While breakthrough treatments have emerged for several cancers over the last two decades, driving striking improvements in survival and other clinical outcomes, too little is known about the risk of therapy-related hematologic cancers following targeted and immunotherapeutic approaches. In a study to be presented at the American Society of Clinical Oncology (ASCO) 2020 virtual meeting, a Roswell Park Comprehensive Cancer Center team reports that in many cases, these newer treatment approaches may reduce the risk of therapy-related myelodysplastic syndrome or acute myeloid leukemia (tMDS/AML) compared to chemotherapy-based treatment strategies.

Scientists identify promising immunotherapy combination for pediatric brain cancer
Scientists at Sanford Burnham Prebys have discovered that combining immunotherapy with a drug called tumor necrosis factor (TNF) eradicated a deadly type of pediatric brain tumor in mice. The discovery, published in Nature Neuroscience, is expected to lead to a clinical trial to test the benefits of the treatment in patients. The findings also hold implications for other cancers that do not respond to immunotherapy.
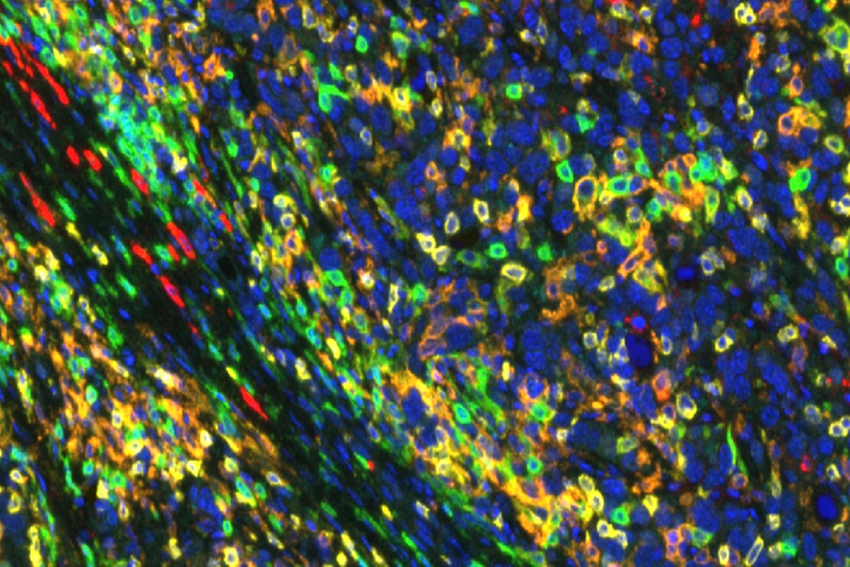
Roswell Park Team Proposes Strategy for Making Pancreatic Tumors Respond to Checkpoint Inhibition
A possible new strategy for treating pancreatic cancer highlights the promise of collaboration between experts in both precision medicine and immunology. The findings from a team led by Agnieszka Witkiewicz, MD, and Erik Knudsen, PhD, at Roswell Park Comprehensive Cancer Center and published today in the journal Gut suggest a combination treatment approach that can make some breakthrough immunotherapy drugs effective for more patients with pancreatic cancer.

Massive study reveals how “hypermutated” malignant brain tumors with many mutations escape chemotherapy and immunotherapy
An analysis of more than 10,000 gliomas and clinical outcomes reported in Nature by scientists in Boston and Paris found that glioma patients whose tumors were hypermutated actually had no significant benefit when treated with checkpoint blockers.

Combination creates powerful central memory T cells for cellular therapy
MD Anderson researchers find that treating T cells with panobinostat and IL-21 re-programs them to a powerful central memory T cell type that persists longer.
UAH research into developing artificial lymph nodes has immunotherapy implications
Research into engineering artificial organs that mimic the functions of human lymph nodes at The University of Alabama in Huntsville (UAH) has garnered one of its professors a $507,777 National Science Foundation (NSF) Faculty Early Career Development Program (CAREER) Award.
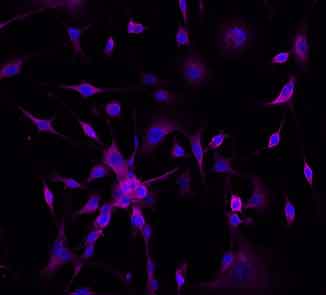
Designer Probiotic Treatment for Cancer Immunotherapy
Columbia Engineers have engineered probiotics to safely deliver immunotherapies within tumors, including nanobodies against two proven therapeutic targets—PD-L1 and CTLA-4. Continuously released by bacteria, the drugs continue to attack the tumor after just one dose, facilitating an immune response resulting in tumor regression. The versatile probiotic platform can also be used to deliver multiple immunotherapies simultaneously, enabling the release of effective therapeutic combinations within the tumor for more difficult-to-treat cancers like colorectal cancer.

Ping-Chih Ho Appointed Associate Member of Ludwig Institute for Cancer Research
Ludwig Cancer Research congratulates Ping-Chih Ho on his appointment as Associate Member at the Lausanne Branch of the Ludwig Institute for Cancer Research.
Tip Sheet: Mesh loaded with T cells shrinks tumors; second dose of CAR-T cells shows potential; and gene-edited cells stay safe as immunotherapy attacks cancer
Below are summaries of recent Fred Hutch research findings with links for additional background and media contacts.
Fred Hutch at ASH: Latest CAR T data – BCMA, CD19, CD20 – plus new insights on transplantation, gene therapy – and more
The 61st American Society of Hematology Annual Meeting and Exposition will take place Dec. 7–10 in Orlando, Florida
MD Anderson and Takeda Announce Collaboration to Accelerate the Development of Clinical-Stage, Off-The-Shelf CAR NK-Cell Therapy Platform
MD Anderson and Takeda today announced an exclusive license agreement and research agreement to develop CAR NK-cell therapies for the the treatment of B-cell malignancies.
Tip Sheet: How a failed Alzheimer’s drug boosts CAR T-cell therapy; a new type of anti-CRISPR; and how GVHD starts in the gut
Below are summaries of recent Fred Hutch research findings, with links for additional background and media contacts.

Investigational drug with immunotherapy may provide new therapeutic opportunity for patients previously treated for kidney and lung cancer
Investigational drug with immunotherapy may provide new therapeutic opportunity for patients previously treated for kidney and lung cancer. Pegilodecakin with pembrolizumab and nivolumab shown to be safe in Phase IB study