Rabih Al-Kaysi’s molecular motors look like tiny worms, but they’re actually crystallized molecules that move in response to light. These machines could someday solve real-world problems, like being used as drug-delivery robots. The researchers will present their results at ACS Spring 2024.
Tag: Crystals
Phonon engineering in Yb:La2CaB10O19 crystal for extended lasing beyond the fluorescence spectrum
The direct lasing outside the fluorescence spectrum is deemed impossible owing to the ‘zero-gain’ cross-section.

Kirstin Alberi: Then and Now / 2012 Early Career Award Winner
Kirstin Alberi is Director of the Materials Science Center at the National Renewable Energy Laboratory. Her research into semiconductor materials shows that scientists can use light as a tool while depositing materials as a vapor and controlling the substrate’s temperature.
A project by Russian scientists to help create capsules for targeted drug delivery
Scientists from MIPT and ITMO University and their colleagues have studied the formation and growth of crystals from simple organic molecules into large associations. These experiments will help create capsules for targeted drug delivery to specific tissues in the human body. The scientific paper was published in the journal Crystal Growth & Design.
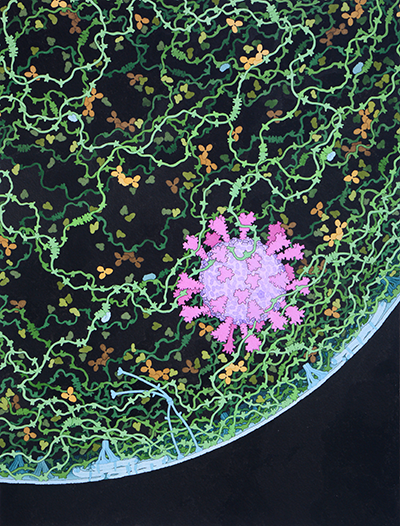
More than 1,000 SARS-CoV-2 Coronavirus Protein 3D Structures Available
New Brunswick, N.J. (March 3, 2021) – The 3D structures of more than 1,000 SARS-CoV-2 coronavirus proteins are freely available from the RCSB Protein Data Bank headquartered at Rutgers University–New Brunswick. The data bank reached the milestone this week, with 1,018 proteins as…

Socially distanced chemistry for kids: Join the U.S. Crystal Growing Competition!
“Now, more than ever, with so many kids being at home, they need fun, hands-on scientific activities,” says Jason Benedict, contest founder, dad, and an associate professor of chemistry in the UB College of Arts and Sciences.

Quantum Materials Quest Could Benefit From Graphene That Buckles
Graphene, an extremely thin two-dimensional layer of the graphite used in pencils, buckles when cooled while attached to a flat surface, resulting in beautiful pucker patterns that could benefit the search for novel quantum materials and superconductors, according to Rutgers-led research in the journal Nature. Quantum materials host strongly interacting electrons with special properties, such as entangled trajectories, that could provide building blocks for super-fast quantum computers. They also can become superconductors that could slash energy consumption by making power transmission and electronic devices more efficient.
‘Blinking” Crystals May Convert CO2 into Fuels
Imagine tiny crystals that “blink” like fireflies and can convert carbon dioxide, a key cause of climate change, into fuels. A Rutgers-led team has created ultra-small titanium dioxide crystals that exhibit unusual “blinking” behavior and may help to produce methane and other fuels, according to a study in the journal Angewandte Chemie. The crystals, also known as nanoparticles, stay charged for a long time and could benefit efforts to develop quantum computers.

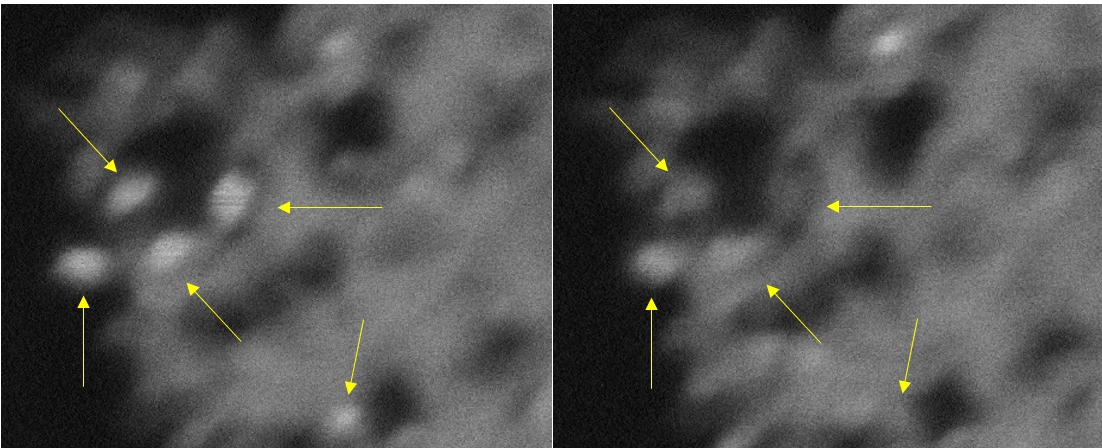
Study reveals breach of ‘dancing’ barrier governs crystal growth
Researchers at the University of Illinois at Chicago used computer-based simulations to analyze how atoms and molecules move in a solution and identified a general mechanism governing crystal growth that scientists can manipulate when developing new materials.