A new therapy developed by researchers at Rush University Medical Center is showing success as a way to prevent COVID-19 symptoms in mice.
Tag: covid 19 therapies
Baylor Scott & White Health Enrolls First Patients in the World in Trial for Inhaled Remdesivir
Earlier this month, Baylor Scott & White Research Institute enrolled the first four patients in the world on Gilead’s new clinical trial involving an investigational inhaled solution of remdesivir (NCT04539262). This is Gilead’s first trial in COVID-19 patients examining the safety and efficacy of an inhaled solution of the drug in an outpatient setting. The study of an inhaled solution asks whether this mode of delivery can help reduce the amount of virus from the airways earlier.

First Patients in NIH ACTIV-3 Clinical Trial Enroll in Dallas
On Wednesday in Dallas, just one day after the initiative was launched by the National Institutes of Health (NIH), Baylor Scott & White Research Institute enrolled the first patient in the world for the ACTIV-3 clinical trial. A second patient was enrolled the following day.

Baylor Scott & White Research Institute Expands Efforts in the Fight Against COVID-19
As the global response to the SARS-COV-2 virus that causes COVID-19 approaches 200 days, Baylor Scott & White Research Institute, the research and development arm of Baylor Scott & White Health, is accelerating its pace of bringing clinical trials online.
Baylor Scott & White Research Institute continues to mobilize staff and resources, including components needed to integrate critical patient-safety measures at every participating site within the Baylor Scott & White system for industry sponsored drug trials, investigator-initiated drug trials and research studies, and observational and data studies designed to help increase knowledge around case trends, viral epidemiology, and care best practices.
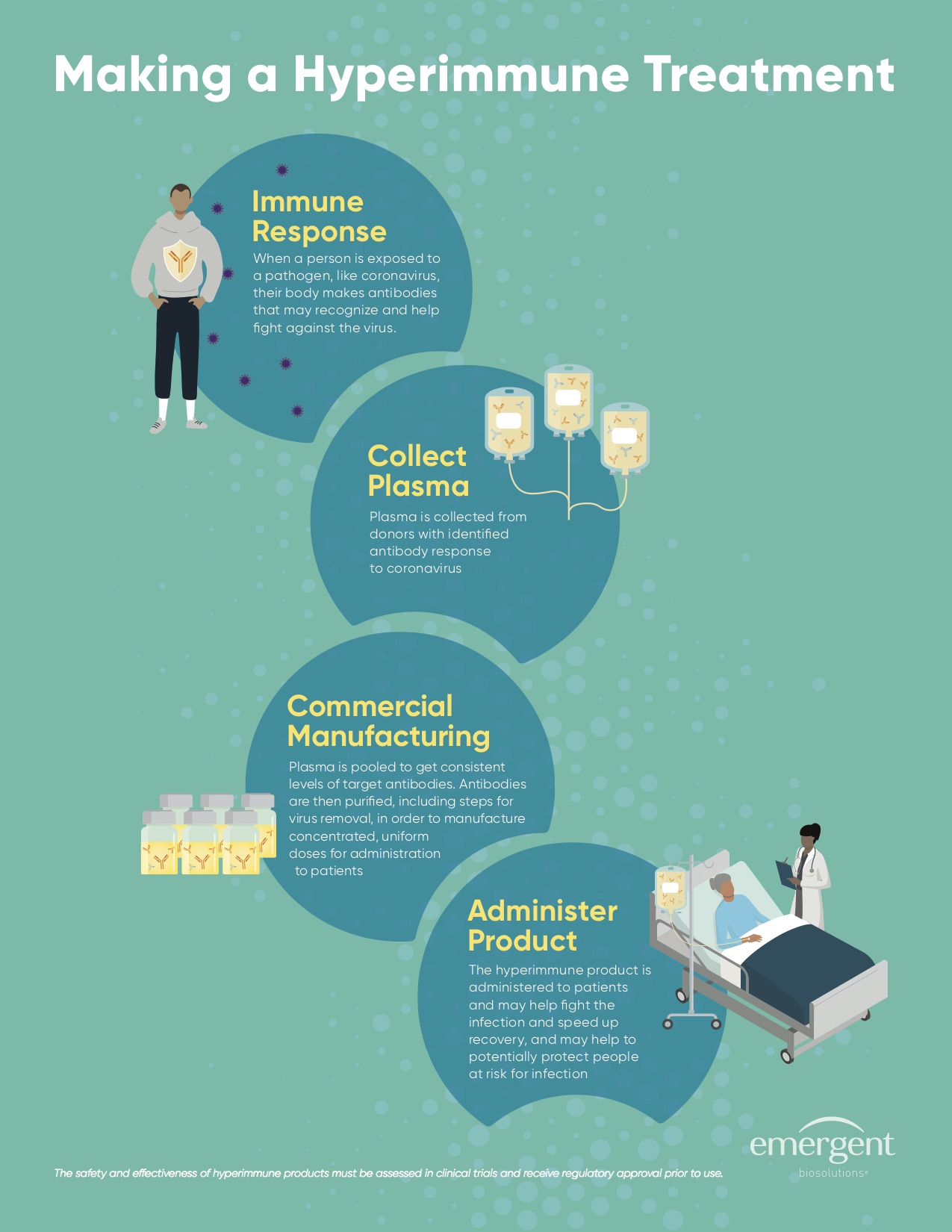
Mount Sinai Health System, Emergent BioSolutions, and ImmunoTek Bio Centers Form Collaboration to Develop Emergent’s COVID-19 Hyperimmune Globulin (COVID-HIG) Product Candidate with U.S. Department of Defense Funding
Mount Sinai and Emergent to conduct clinical trials to evaluate COVID-HIG for post-exposure prophylaxis of COVID-19 in front-line health care workers and to support a potential Expanded Access Program for military personnel with funding from the U.S. Department of Defense
– ImmunoTek to extend operating license and provide training to Mount Sinai to establish onsite plasma collection to support production of COVID-HIG
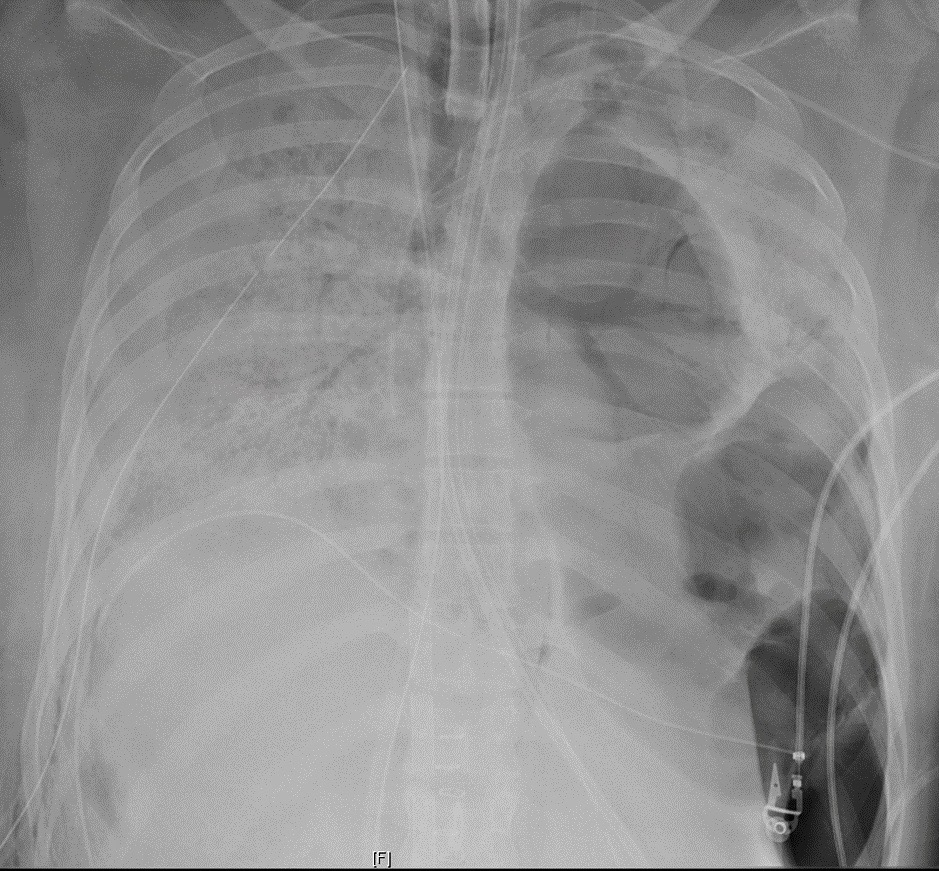
Lung Transplant Performed on a COVID-19 Patient at Northwestern Medicine
For the first time, surgeons at Northwestern Medicine performed a double-lung transplant on a patient whose lungs were damaged by COVID-19. The patient, a Hispanic woman in her 20s, spent six weeks in the COVID ICU on a ventilator and extracorporeal membrane oxygenation (ECMO), a life support machine that does the work of the heart and lungs.