Ferrum Health, an enterprise healthcare AI platform supporting over fifty third-party, industry-leading algorithms, proudly announces a strategic partnership with PaxeraHealth, a global leader in enterprise imaging solutions.

news, journals and articles from all over the world.

Ferrum Health, an enterprise healthcare AI platform supporting over fifty third-party, industry-leading algorithms, proudly announces a strategic partnership with PaxeraHealth, a global leader in enterprise imaging solutions.
The American Association of Neuromuscular & Electrodiagnostic Medicine (AANEM), is excited to announce John A. Morren, MD, as a plenary speaker at the 2024 AANEM Annual Meeting Oct. 15-18, in Savannah, Georgia.
To fill high demand positions, CHLA trainees undergo reskilling, a recent addition to the workforce glossary that refers to employees’ learning of new skills in order to pursue a different role within the same organization.
Researchers at the University of California San Diego have been awarded a $5.6 million grant to further their efforts within the All of Us Research Program, which aims to accelerate and promote equity in health research.
Cole-Parmer, an Antylia Scientific company, is a trusted global leader in laboratory essentials. We deliver a portfolio of reliable equipment and consumables to help speed scientific discovery in life science, clinical, academic and research laboratories.
The drug minocycline, an antibiotic that also decreases inflammation, failed to slow vision loss or expansion of geographic atrophy in people with dry age-related macular degeneration (AMD), according to a phase II clinical study at the National Eye Institute (NEI),…
TE Connectivity, a world leader in connectors and sensors, has acquired three companies focused on microfluidic cartridge and blister reagent package development, usability testing, clinical research and manufacturing. The companies are Toolbox Medical Innovations (USA), Wi Inc (USA) and microiLIQUID (Spain).
The combined entities leverage design and manufacturing capabilities in the United States and Europe and are focused on serving a global client base ranging from startups to multinationals in the life science, IVD and cell therapy markets.

GenScript USA Inc., the world’s leading research reagent provider, announced today that Brazil’s National Health Surveillance Agency (ANVISA) (Agência Nacional de Vigilância Sanitária) has authorized the use of the cPass™ SARS-CoV-2 Neutralization Antibody Detection Kit for detecting neutralizing antibodies. The cPass test is the first and only ANVISA authorized test for detecting neutralizing antibodies to SARS-CoV-2. Neutralizing antibodies specifically block the ability of a virus to infect a cell and are well-recognized to confer immunity.
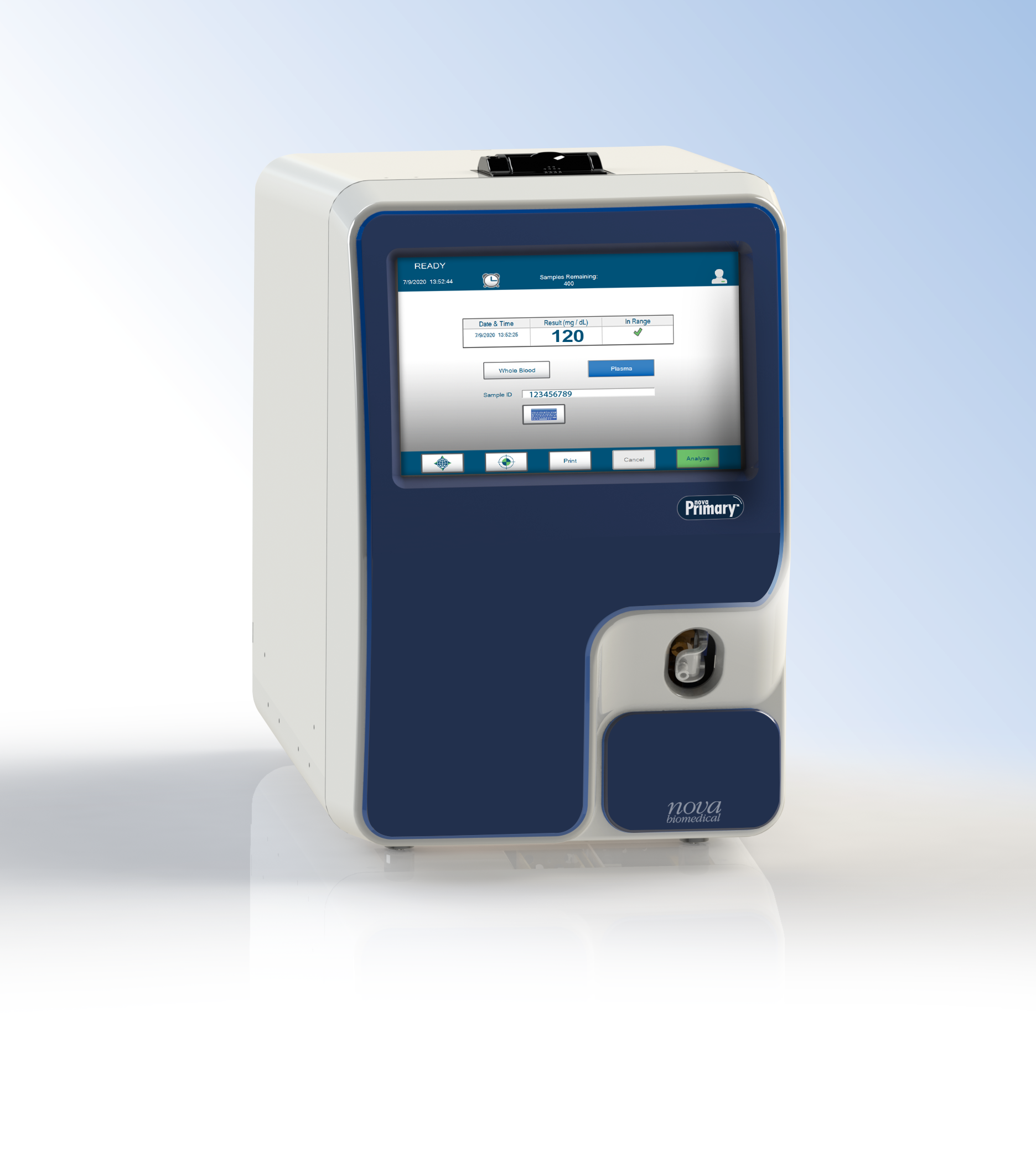
New glucose reference correlation analyzer replaces “gold standard” YSI 2300
Neuro-oncologist and renowned physician-scientist Ingo Mellinghoff will lead MSK’s distinguished Department of Neurology after previously serving as Acting Co-Chair.