The US Food and Drug Administration has approved Moderna’s mRNA vaccine for older adults to protect against respiratory syncytial virus (RSV). The vaccine, called mRESVIA, is for people ages 60 and older. According to a company press release, this marks…
Tag: Moderna
Improving gene therapy with tiny bubbles
Beyond vaccines, mRNA offers immense potential to fight disease, but targeting the genetic material to specific diseased cells is challenging—requiring a new method. To meet this need, researchers at the Case Western Reserve University School of Medicine, with a Global Research Fellowship award from Moderna Inc., are developing a process that essentially uses bubbles to overcome the problem.
Rutgers Expert Available to Discuss Mixing and Matching of COVID-19 Vaccines, Booster Shots
Rutgers infectious disease expert Shobha Swaminathan is available to discuss the mixing and matching of COVID-19 vaccines and boosters as the Food and Drugs Administration authorized booster shots for both the Johnson & Johnson and Moderna vaccines. “There is mounting…
10 questions you want answered on COVID-19
Should children under the age of 12 be vaccinated against COVID-19? Is it possible to achieve herd immunity? Will an 80 per cent vaccination rate keep us safe? In this video, UniSA epidemiologist Professor Adrian Esterman answers these questions and a lot more. The former World Health Organization consultant shares his expertise – 50 years’ worth – and argues for a different approach to tackling COVID-19.
https://www.youtube.com/watch?v=MLKC9q9yEto
Data and safety review board reports how it monitored the COVID-19 vaccine trials
Evaluation of three vaccine candidates during the COVID-19 pandemic fell to 12 experts of the federally appointed COVID-19 Vaccine Data and Safety Monitoring Board. This team has now taken the unusual step of publishing details of their review process in The Journal of Infectious Diseases.
Rutgers Expert Available to Discuss Dangers of Missing Second Doses of COVID-19 Vaccines
Rutgers scholar Stanley H. Weiss is available to discuss the trend of people skipping their second dose of COVID-19 vaccines and how this delays herd immunity. “The second shot leads to a huge boost of the body’s immune protection –…
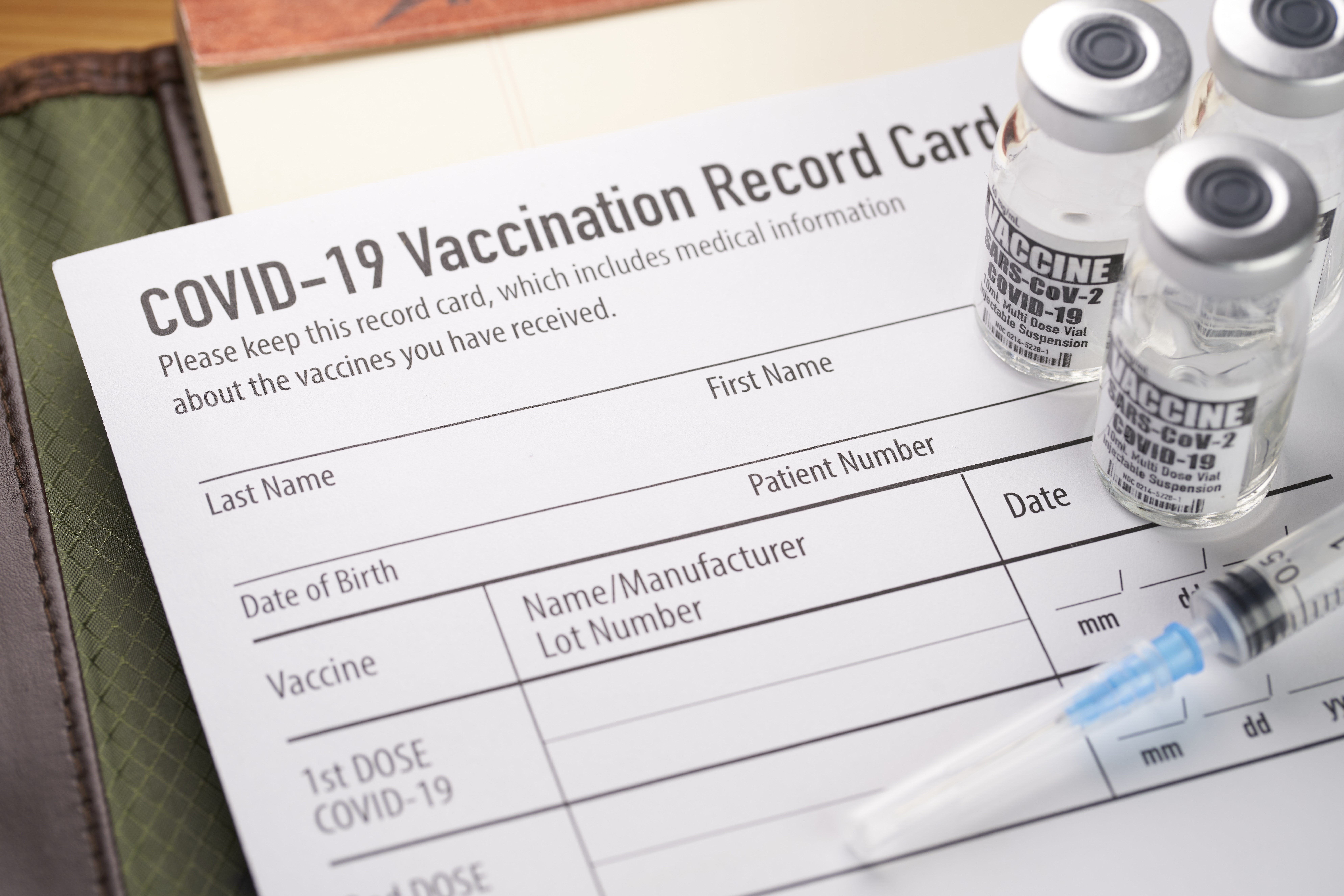
6 important things to know about your COVID-19 vaccine card
Everyone who gets vaccinated for COVID-19 in the U.S. receives a vaccine card. Sarah Lynch, clinical assistant professor of pharmacy practice at Binghamton University, State University of New York, offers insight into why vaccine cards are important and why you…
FAU Expert Answers Questions about COVID-19 Vaccines
FAU’s Joanna Drowos, D.O., M.P.H., M.B.A., provides answers to some of the most frequently asked questions regarding COVID-19 vaccines.

TTP El Paso Receives 3,000 doses of Moderna’s COVID-19 Vaccine
Three thousand doses of Moderna’s COVID-19 vaccine arrived at the Texas Tech Physicians of El Paso Alberta, Kenworthy and Transmountain clinics on Wednesday, Dec. 23.
Rutgers Experts Available to Discuss Moderna Coronavirus Vaccine Rollout
Rutgers medical experts are available to discuss the FDA’s approval of Moderna’s coronavirus vaccine for emergency use and its rollout. The Rutgers New Jersey Medical School is one of only two sites for the Phase 3 trial of the Moderna…
George Washington University Public Health/Medical Experts Available for Media Interviews on COVID-19 Vaccine
On Dec. 17, a U.S. Food and Drug Administration panel is expected to review the data on the Moderna COVID-19 vaccine, paving the way for approval. Researchers at the George Washington University led one of the 100 clinical sites testing…

Rutgers Expert Available to Discuss COVID-19 Vaccine Distribution Priorities
Perry N. Halkitis, dean of the Rutgers School of Public Health, is available to discuss the priorities for COVID-19 vaccination distribution. “It is essential that we prioritize vaccinations for healthcare providers and nursing home residents, but they are not the…

UNLV Immunologist on the Differences Between Two Leading COVID-19 Vaccine Candidates
Millions around the world have waited for news about a COVID-19 vaccine, regarding it as the beginning of the end for the global pandemic and a herald for the eventual return to “normal life.” Recent announcements from pharmaceutical companies Pfizer and Moderna that their respective late-stage vaccine trials have shown a 90% or better effectiveness rate have received international applause, excitement furthered with estimates that doses could be ready as early as December.
Rutgers Vaccine Expert Available to Discuss Moderna’s FDA Submission
Rutgers’ principal investigator for the Moderna phase 3 coronavirus vaccine clinical trial at Rutgers New Jersey Medical School is available to discuss the drugmaker’s submission of its vaccine to the U.S. Food and Drug Administration for regulatory approval. “It is…
Rutgers Infectious Disease Expert Available to Discuss Moderna’s Coronavirus Vaccine
Rutgers’ principal investigator for the Moderna phase 3 coronavirus vaccine clinical trial at Rutgers New Jersey Medical School is available to discuss the drugmaker’s vaccine, which the company has announced is 94.5 percent effective. Moderna intends to seek an emergency…