A year after launching one of the world’s largest COVID-19 antibody surveys, Texas CARES, public health experts at The University of Texas Health Science Center at Houston (UTHealth Houston) and the Texas Department of State Health Services (DSHS) are estimating that over 75% of Texans have COVID-19 antibodies.
Tag: serological COVID-19 tests
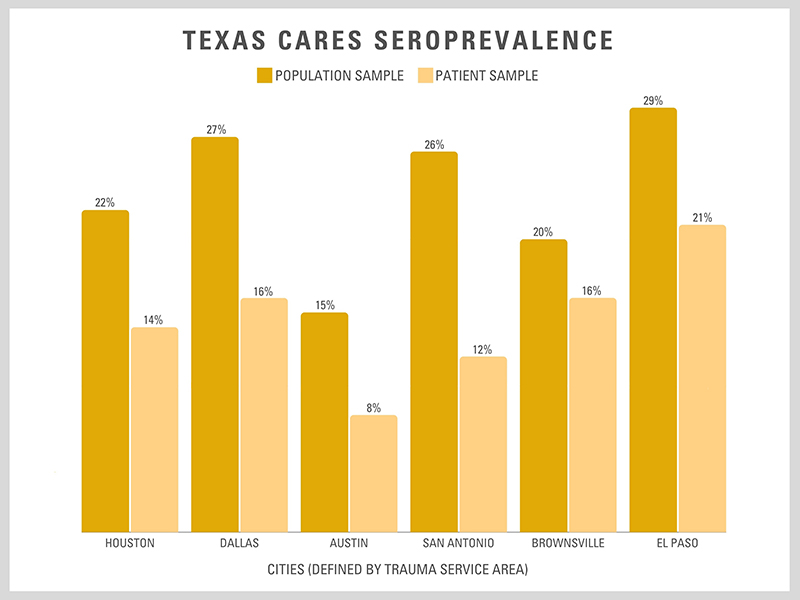
More than 20% of Texans may have COVID-19 antibodies, serological assessment finds
Four months after launching the nation’s largest COVID-19 serological testing assessment, Texas CARES, researchers at The University of Texas Health Science Center at Houston (UTHealth) and the Texas Department of State Health Services (DSHS) have compiled preliminary data estimating that 14% to 24% of Texans have COVID-19 antibodies.
Public health experts at DSHS and UTHealth collaborate on nation’s largest COVID-19 serological testing assessment
To help public health professionals and scientists better understand the spread of COVID-19 in Texas and the immune response it causes in individuals, researchers at The University of Texas Health Science Center at Houston (UTHealth) are partnering with the Texas Department of State Health Services (DSHS) to launch the Texas Coronavirus Antibody Response Survey (Texas CARES). Texas CARES will determine the proportion of people throughout Texas who have COVID-19 antibodies, indicating a past infection and presumably some degree of immune protection.
New York State Department of Health Grants Emergency Use Authorization to Mount Sinai for Quantitative COVID-19 Antibody Test
The Clinical Laboratories of The Mount Sinai Hospital has received emergency use authorization from the New York State Department of Health (NYSDOH) for quantitative use of Mount Sinai’s COVID-19 antibody test, making Mount Sinai’s lab the first in the country to run an authorized, fully quantitative antibody test that can deliver a precise numeric measurement of the level of antibodies in a patient’s blood
Mount Sinai study finds SARS-CoV-2 induces robust antibody responses stable for at least 3 months
Research from the Mount Sinai Health System, just posted to pre-print server MedRxiv (https://www.medrxiv.org/content/10.1101/2020.07.14.20151126v1), shows that the vast majority (more than 90%) of infected individuals with mild-to-moderate COVID 19 experience robust IgG antibody responses against the viral spike protein. The researchers also show…
Penn Medicine Launches COVID-19 Convalescent Plasma Study
Convalescent plasma therapy for COVID-19 patients — an experimental approach of giving a transfusion of plasma collected from a donor who has recovered from COVID-19 to a patient with an active infection — is the focus of a new two-part research initiative at Penn Medicine. Researchers will first collect plasma from people who have recovered from their infection under a donor research protocol. The second part involves conducting clinical trials to test the safety and efficacy of giving that plasma to moderately and severely ill hospitalized patients.