MIMEDX is the latest addition to the RegeneratOR Innovation Accelerator, located in the Regenerative Medicine Hub (RegenMed Hub), a rapidly growing regenerative medicine ecosystem based in the Innovation Quarter of Winston-Salem.
Tag: Biologics
Infection Rates in Psoriatic Arthritis Patients on Biologics Have Decreased, According to National Data
New research presented this week at ACR Convergence, the American College of Rheumatology’s annual meeting, shows significant decreases in infections among people with psoriatic arthritis over the years 2012-2017.
Early Combined Treatment with Biologic and Conventional DMARDs Could be Effective for Polyarticular Juvenile Arthritis
New research presented this week at ACR Convergence, the American College of Rheumatology’s annual meeting, found that patients started on early, aggressive treatment with a combination of biologic and conventional disease modifying antirheumatic drugs (DMARDs) achieved clinically inactive disease in children with polyarticular juvenile idiopathic arthritis (JIA) more frequently compared to other treatment plans 24 months after starting treatment.
ACR Releases Position Statement on Patient Safety and Site of Service for Biologics
The American College of Rheumatology (ACR) has released an updated position statement on patient safety and site of service for biologics outlining several reasons why the ACR strongly believes infusions should be administered in a monitored health care setting with onsite supervision by a provider with appropriate training in biologic infusions.
June 2021 Issue of Neurosurgical Focus: “Biologics and Spinal Fusion”
Announcement of articles in the June 2021 issue of Neurosurgical Focus.
Black Patients with RA Less Likely to Receive a Biologic, More Likely to Be Treated with Glucocorticoids Than Whites
A new study reveals that Black patients with rheumatoid arthritis were less likely to be prescribed a biologic treatment and more likely to use glucocorticoids, which carry a risk of serious long-term side effects. This study highlights ongoing racial disparities in the care of patients with rheumatic disease. Details of the study was shared at ACR Convergence, the American College of Rheumatology’s annual meeting.

AAOS Advances Biologics Initiative with Innovative Dashboard
The American Academy of Orthopaedic Surgeons (AAOS) continues to demonstrate its commitment to advancing the quality of musculoskeletal care in a fully transparent and scientific way. Debuting today as a new member benefit, the AAOS Biologics Dashboard is a dynamic online tool designed to help orthopaedic surgeons navigate the approval status of biologic-based interventions. The development of the AAOS Biologics Dashboard is just one of several efforts within the Academy’s Biologics Initiative that offers evidence-based guidance to the musculoskeletal health community. An additional effort is the revision of two biologics-related position statements, recently approved by the AAOS Board of Directors.
The Tri-Institutional Therapeutics Discovery Institute, Inc. Renews Partnerships with Takeda and Bridge Medicines, LLC
To date, work done within the Tri-I TDI has resulted in the launch of two New York City–based companies and the licensing of six therapeutic discovery programs.

Aphea.Bio Joins the International Phytobiomes Alliance
Aphea.Bio has joined the International Phytobiomes Alliance as a sponsoring partner, both organizations announce today
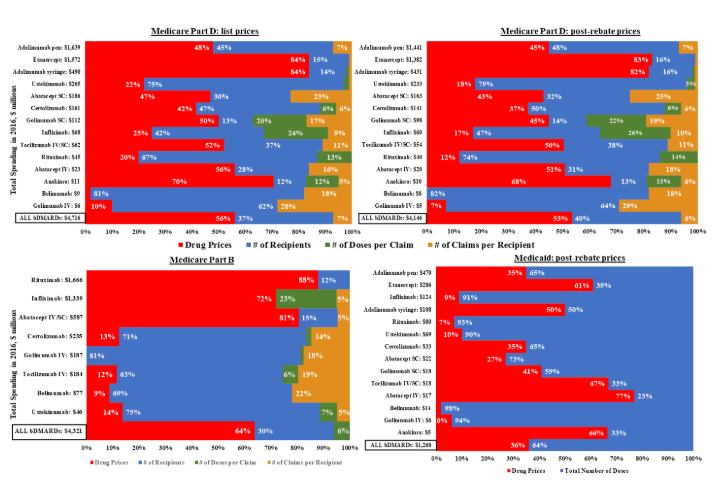
Post-Market Price Changes Alone Account for Most Recent Spending Growth for Biologics, and Rebates Have Little Impact
New research findings presented at the 2019 ACR/ARP Annual Meeting found that annual spending on biologic DMARDS (biologics) by U.S. public programs and beneficiaries nearly doubled from 2012 to 2016.
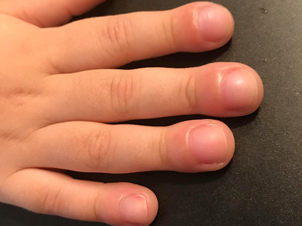
Aggressive Screening Urged to Detect Rare Lung Complication Among Children with Severe Form of Arthritis
A study led by experts at Cincinnati Children’s reports new details about a rare lung complication affecting children with systemic JIA. The complications appear related to how some patients react to treatments called “biologics.”