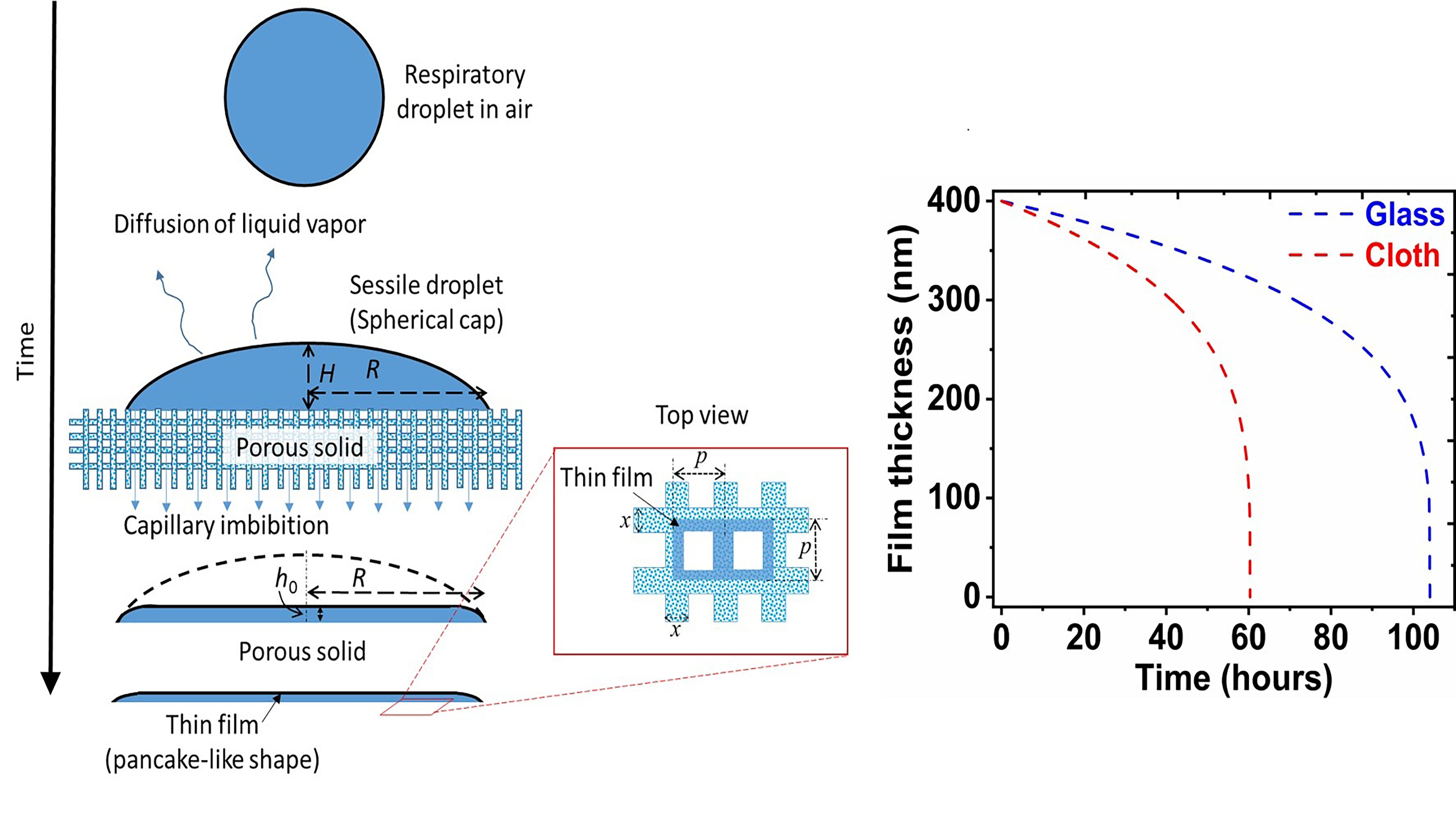
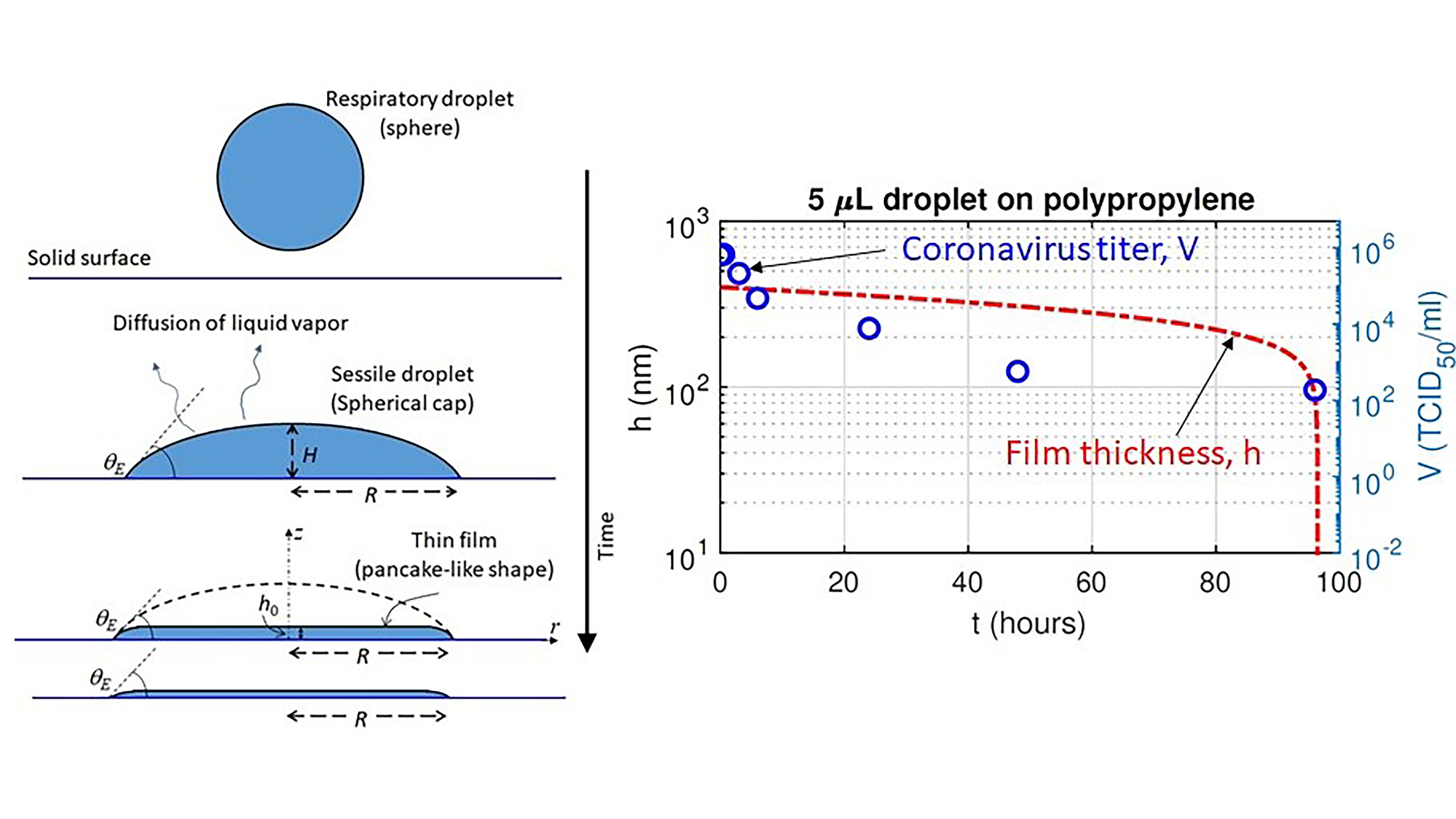
As COVID-19 spreads via respiratory droplets, researchers have become increasingly interested in the drying of droplets on impermeable and porous surfaces; surfaces that accelerate evaporation can decelerate the spread of the virus. In Physics of Fluids, researchers show a droplet remains liquid for a much shorter time on a porous surface, making it less favorable to survival of the virus. On paper and cloth, the virus survived for only three hours and two days, respectively.