“Two-dimensional nanomaterials are extremely thin ⎯ only several atoms thick ⎯ sheet-shaped materials,” said Utana Umezaki, a Rice graduate student who is a lead author on a study published in ACS Nano. “They behave very differently from materials we’re used to in daily life and can have really useful properties: They can withstand a lot of force, resist high temperatures and so on. To take advantage of these unique properties, we have to find ways to turn them into larger-scale materials like films and fibers.”
In order to maintain their special properties in bulk form, sheets of 2D materials have to be properly aligned ⎯ a process that often occurs in solution phase. Rice researchers focused on graphene, which is made up of carbon atoms, and hexagonal boron nitride, a material with a similar structure to graphene but composed of boron and nitrogen atoms.
“We were particularly interested in hexagonal boron nitride, which is sometimes called ‘white graphene’ and which, unlike graphene, doesn’t conduct electricity but has high tensile strength and is chemically resistant,” said Angel Martí, a professor of chemistry, bioengineering, materials science and nanoengineering and chair of Rice’s chemistry department. “One of the things that we realized is that the diffusion of hexagonal boron nitride in solution was not very well understood.
“In fact, when we consulted the literature, we found that the same was true for graphene. We couldn’t find an account of diffusion dynamics at the single molecule level for these materials, which is what motivated us to tackle this problem.”
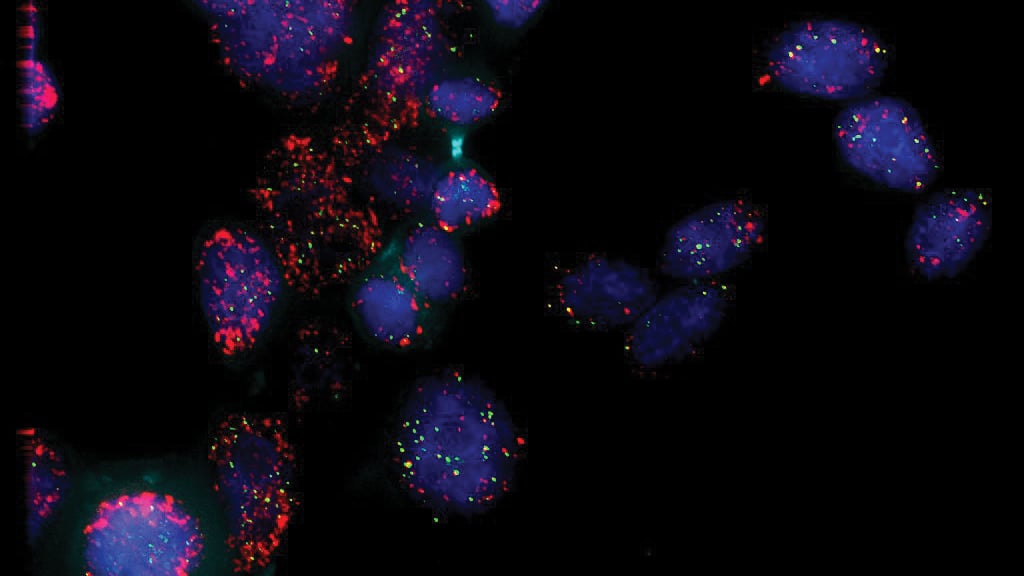
The researchers used a fluorescent surfactant, i.e. glowing soap, to tag the nanomaterial samples and render their motion visible. Videos of this motion allowed researchers to map out the trajectories of the samples and determine the relationship between their size and how they move.
“From our observation, we found an interesting trend between the speed of their movement and their size,” Umezaki said. “We could express the trend with a relatively simple equation, which means we can predict the movement mathematically.”
Graphene was found to move slower in the liquid solution, possibly due to the fact that its layers are thinner and more flexible than hexagonal boron nitride, giving rise to more friction. Researchers believe that the formula derived from the experiment could be used to describe how other 2D materials move in similar contexts.
“Understanding how diffusion in a confined environment works for these materials is important because ⎯ if we want to make fibers, for example ⎯ we extrude these materials through very thin injectors or spinnerets,” Martí said. “So this is the first step toward understanding how these materials start to assemble and behave when they are in this confined environment.”
As one of the first studies to investigate the hydrodynamics of 2D nanosheet materials, the research helps fill a gap in the field and could be instrumental to overcoming 2D material fabrication challenges.
“Our final objective with studying these building blocks is to be able to generate macroscopic materials,” Martí said.
Anatoly Kolomeisky, professor of chemistry and chemical and biomolecular engineering at Rice, and Matteo Pasquali, the A.J. Hartsook Professor of Chemical and Biomolecular Engineering and professor of chemistry and of materials science and nanoengineering, are corresponding authors on the study.
The research was supported by the National Science Foundation (1807737, 2108838), the Air Force Office of Scientific Research (FA9550-19-1-7045) and the Welch Foundation (C-2152, C-1668, C-1559).
-30-
This release can be found online at news.rice.edu.
Follow Rice News and Media Relations via Twitter @RiceUNews.
Peer-reviewed paper:
Brownian Diffusion of Hexagonal Boron Nitride Nanosheets and Graphene in Two Dimensions | ACS Nano | DOI: 10.1021/acsnano.3c11053
Authors: Utana Umezaki, Ashleigh D. Smith McWillams, Zhao Tang, Zhi Mei Sonia He, Ivan R. Siqueira, Stuart J. Corr, Hijun Ryu, Anatoly B. Kolomeisky, Matteo Pasquali, and Angel A. Martí
https://pubs.acs.org/doi/10.1021/acsnano.3c11053
Image downloads:
https://news-network.rice.edu/news/files/2024/01/240124_Umezaki_Fitlow_Fitlow_009-2cbf5109f3b7a353.jpg
CAPTION: Utana Umezaki is a Rice graduate student and lead author on a study published in ACS Nano. (Photo by Jeff Fitlow/Rice University)
https://news-network.rice.edu/news/files/2024/01/illustration-f36d0f2ce8ee71a9.jpg
(Image courtesy of Mario Norton/Rice University)
https://news-network.rice.edu/news/files/2024/01/240124_Umezaki_Fitlow_Fitlow_019-7432727baa775389.jpg
CAPTION: The researchers used a fluorescent surfactant, i.e. glowing soap, to tag the nanomaterial samples and render their motion visible. (Photo by Jeff Fitlow/Rice University)
https://news-network.rice.edu/news/files/2024/01/240124_Umezaki_Fitlow_Fitlow_023-99e76d75a5bc2b44.jpg
CAPTION: Utana Umezaki (left) and Angel Martí (Photo by Jeff Fitlow/Rice University)
https://news-network.rice.edu/news/files/2024/01/Kolomeisky_Pasquali-a2e8c11a41469240.jpg
CAPTION: Anatoly Kolomeisky (left) is a professor of chemistry and chemical and biomolecular engineering at Rice University. Matteo Pasquali is the A.J. Hartsook Professor of Chemical and Biomolecular Engineering and a professor of chemistry, materials science and nanoengineering. (Photos by Gustavo Raskoksy/Rice University)
Links:
Kolomeisky Research Group: https://kolomeisky.rice.edu/
Angel Martí Group at Rice University: https://martigroup.rice.edu/
Pasquali Research Group: https://pasquali.rice.edu/
Department of Bioengineering: https://bioengineering.rice.edu/
Carbon Hub: https://carbonhub.rice.edu/
Department of Chemistry: https://chemistry.rice.edu/
Department of Chemical and Biomolecular Engineering: https://chbe.rice.edu/
Department of Materials Science and NanoEngineering: https://msne.rice.edu/
Wiess School of Natural Sciences: https://naturalsciences.rice.edu
George R. Brown School of Engineering: https://engineering.rice.edu
About Rice:
Located on a 300-acre forested campus in Houston, Rice University is consistently ranked among the nation’s top 20 universities by U.S. News & World Report. Rice has highly respected schools of architecture, business, continuing studies, engineering, humanities, music, natural sciences and social sciences and is home to the Baker Institute for Public Policy. With 4,574 undergraduates and 3,982 graduate students, Rice’s undergraduate student-to-faculty ratio is just under 6-to-1. Its residential college system builds close-knit communities and lifelong friendships, just one reason why Rice is ranked No. 1 for lots of race/class interaction, No. 2 for best-run colleges and No. 12 for quality of life by the Princeton Review. Rice is also rated as a best value among private universities by Kiplinger’s Personal Finance.