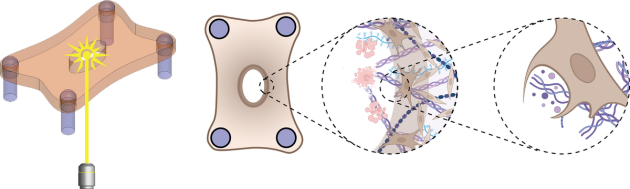
Researchers at Boston University and Harvard University uncovered this cellular capability by simulating both cuts and burns in a miniature skin tissue model and monitoring how fibroblasts responded. The results of a study published in APL Bioengineering show that in scorched samples, fibroblasts consumed and removed broken down tissue — a process that scientists previously thought was solely carried out by immune cells. The authors of the study suggest that the finding is an important first step toward the development of future therapies that speed up and optimize the healing of burns.
“Our bodies need to clear dead tissue out of wounds before it begins repair. But if that phase takes too long, then the wound won’t repair at all,” said Boston University professor Jeroen Eyckmans, lead author of the study. “You want to shorten that first period as much as possible.”
In the clinic, healthcare professionals already take an “out with the old, in with the new” approach, prioritizing the cleaning of burns. However, the science of how the body responds differently to burns and other injury types on a cellular level is lacking, limiting therapeutic options, Eyckmans said.
To begin peeling back the curtain on the healing process at a more fundamental level, Eyckmans and his colleagues previously engineered a microtissue model composed of fibroblasts buried within a tiny sheet composed of collagen — one of the primary components of the scaffolding, or extracellular matrix (ECM), that supports cells within skin.
Initially, the researchers created wounds in the model exclusively with a half-millimeter wide diamond blade, however, the lacerations were so small, it was difficult to examine how the cells responded. In the new study, they zapped tissues with lasers as well, simulating burn injuries that would be larger and more straightforward to study.
The researchers compared how the cells responded in both injury types by assessing microscopic images collected in the hours following the injuries. With a clear view of the wounded microtissue, they noticed many similarities between the two wound types but several key differences as well.
While the knife damaged the tissue immediately at the site of the incision — sparing the cells and ECM adjacent to the wound — heat generated by the laser created a ring of damaged tissue around where the laser struck.
The knife wound began closing within minutes as expected, but the burn began widening over the course of many hours. The researchers were caught by surprise by the removal of damaged tissue in the burns, as their model excluded any immune cells, which normally lead the charge in removing damaged tissue.
Close inspection revealed that fibroblasts were clearing out the ECM seared by the laser. Further experiments, wherein the team inhibited specific proteins within the fibroblasts, suggested that the cells were ingesting ECM through a process called phagocytosis, which is commonly carried out by immune cells.
“To my knowledge, this is the first study showing that fibroblasts can exhibit phagocytosis in a wound healing context,” Eyckmans said. “Now we have an additional cell type to target that could speed up wound clearance.”
Through a drug, genetic modification or some other means, future clinical treatments could potentially coax fibroblasts to initiate or accelerate ECM removal, facilitating burn healing during the critical early phase.
The laser injury model permitted Eyckmans and his colleagues to take a first look at wound clearance at the cellular level, but the team does not intend to stop there. They have their sights set on extending their model to examine how other cell types, such as those in vasculature, clear and mend wounds.
“Wound healing is an incredibly multifaceted and complicated process, but the insights gleaned from this injury model fill in an important piece of the puzzle. The findings could create a path for new clinical treatments down the line,” said David Rampulla, Ph.D., director of the Division of Discovery Science and Technology at the National Institute of Biomedical Imaging and Bioengineering (NIBIB).
This study was supported by a grant from NIBIB (R21EB028491), as well as grants from the National Science Foundation (2036842) and the Office of the Director at the National Institutes of Health (S10OD028571).
This Science Highlight describes a basic research finding. Basic research increases our understanding of human behavior and biology, which is foundational to advancing new and better ways to prevent, diagnose, and treat disease. Science is an unpredictable and incremental process—each research advance builds on past discoveries, often in unexpected ways. Most clinical advances would not be possible without the knowledge of fundamental basic research.
Study reference: Megan Griebel et al. Fibroblast clearance of damaged tissue following laser ablation in engineered microtissues. APL Bioengineering (2023). DOI: 10.1063/5.0133478
About the artwork: The second image is licensed under the Creative Commons Attribution 4.0 International license.