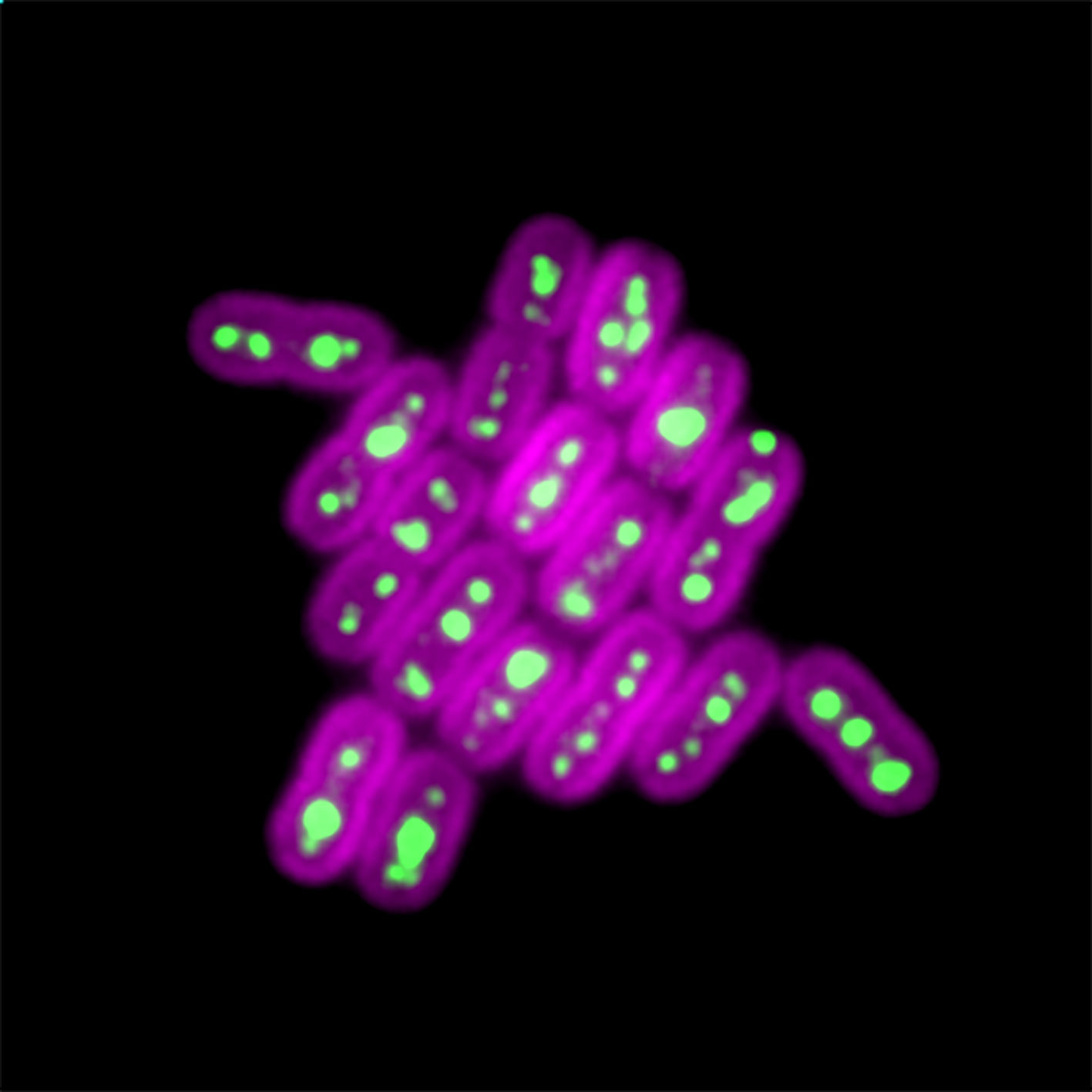
New CU Boulder research published today in Science Advances shows how these specialized compartments, known as carboxysomes, work and how their activity can be accurately measured. The research findings could ultimately lead to increased plant yields, novel antibiotics and improved production efficiency of renewable fuels.
“This was a 50-year question, a long-standing thing that people could never tackle,” said Jeffrey Cameron, an assistant professor in the Department of Biochemistry and coauthor of the new research.
In his work as a postdoctoral researcher, Cameron figured out how carboxysomes are formed: from the inside out. But he couldn’t determine their function in the cells because they struggled to grow under the microscope.
He wanted to answer the questions of, how long do carboxysomes last? How long are they active? What happens to cells without carboxysomes?
So, Cameron and his colleagues tracked specific enzymes within these cells’ carboxysomes, by tagging them with a green fluorescent marker. They then added chemicals to curb the ability of the cell to make new carboxysomes. Researchers tracked them for several days using live cell imaging as they were passed from mother to daughter cells.
Because the cells’ ability to grow was dependent on their ability to fix carbon, in this way they could directly track the effect of the carboxysome on the cells’ growth. The researchers found that by adjusting the amount of CO2 available to cells without carboxysomes, they could switch their growth on and off – effectively reactivating them at will from a dormant state.
And by filming thousands of cells and measuring all of them in the population, they uncovered several practical applications for engineering.
About 5% of these carboxysomes were ultra-productive and could maintain their growth rate for over seven generations.
“This would be like having the same brain passed on from your great, great, great, great, great grandmother that’s still functioning over that many generations,” said Cameron. “If we could understand the principles of why these ones are so active, we might be able to significantly improve plant growth.”
There is now a huge push to try to put carboxysomes into plants to improve photosynthesis, which could greatly enhance plant yields, said Cameron, also of the Renewable and Sustainable Energy Institute (RASEI) at CU Boulder.
Carboxysomes also have potential to serve as mini bioreactors, increasing the metabolism for the processes that create biofuels.
Meantime, pathogenic bacteria, such as Salmonella, contain similar microcompartments that give them metabolic advantage in certain environments. Because humans do not have cyanobacteria or bacterial microcompartments, it may be possible to develop novel antibiotics that specifically target these bacterial structures, he added.
“I want to know, how can I elucidate the basic principles that will allow engineers to move forward?” said Cameron.
Additional authors include lead author Nicholas Hill, graduate research associate in the Department of Biochemistry and Renewable and Sustainable Energy Institute; Jian Wei Tay, Image Analysis Specialist at the Renewable and Sustainable Energy Institute and BioFrontiers Institute; and Sabina Altus, graduate student, and David Bortz, associate professor, in the Department of Applied Mathematics at CU Boulder.
Original post https://alertarticles.info