The Science
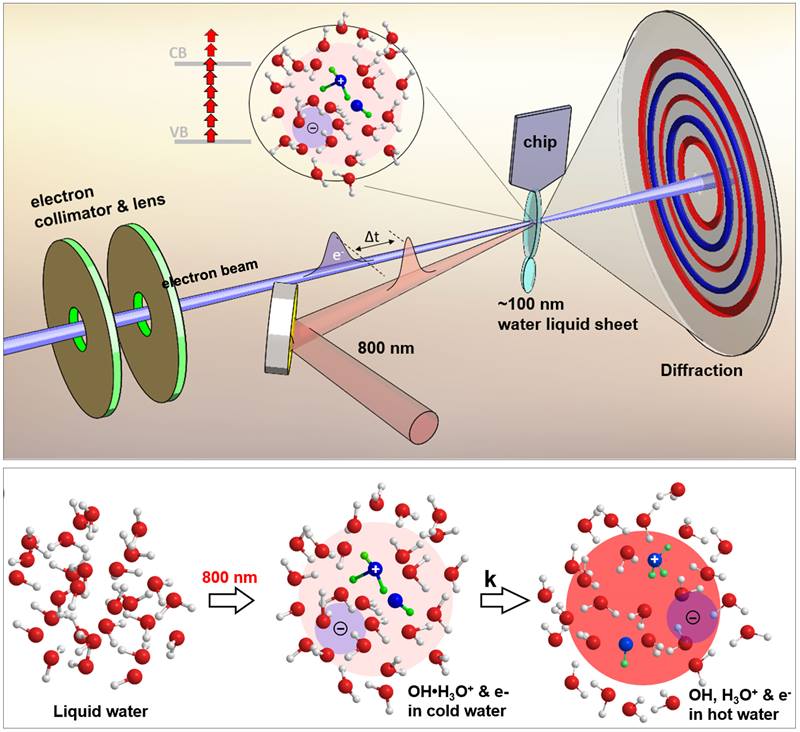
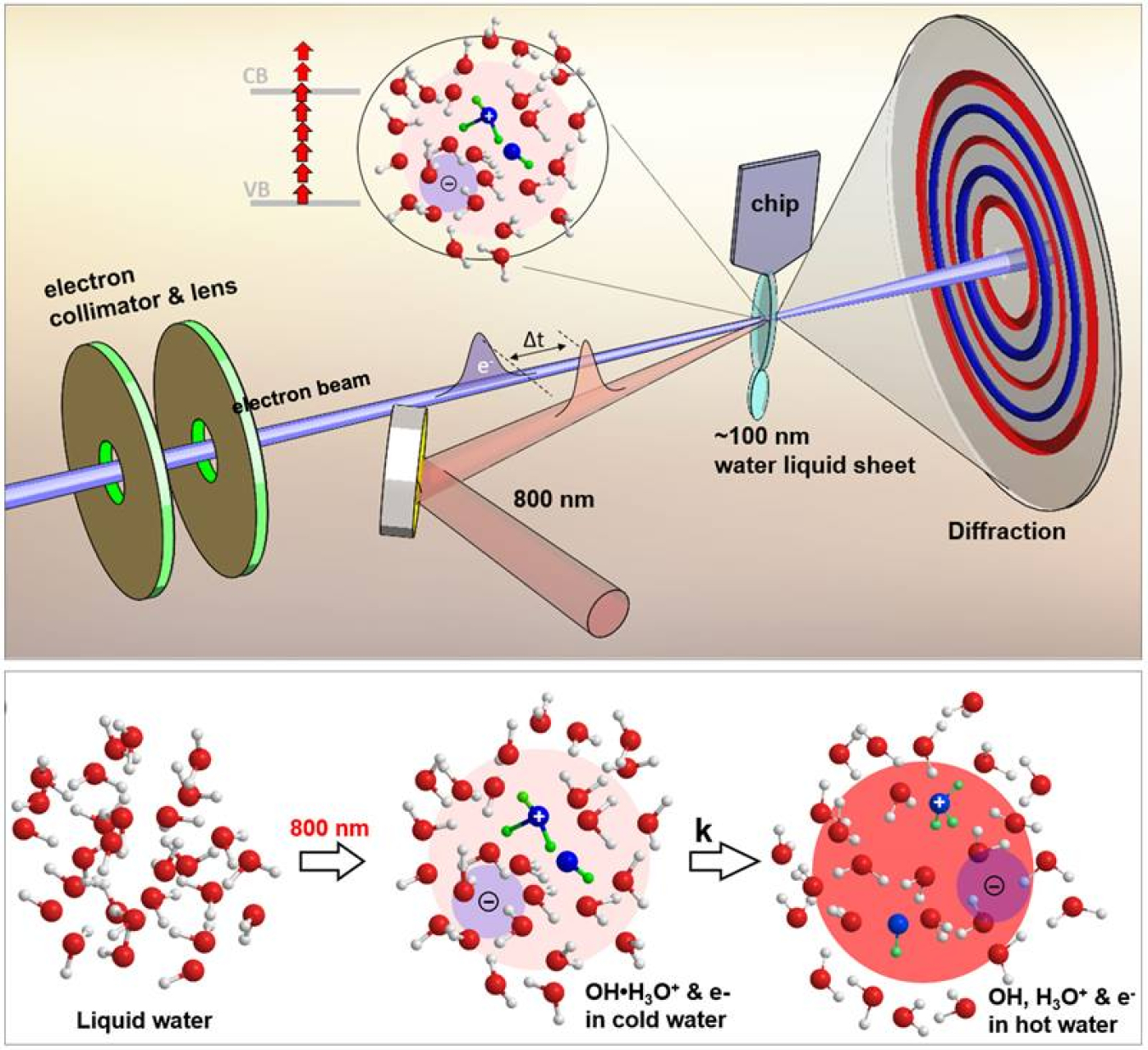
Water gives rise to ultrafast reactions when it is irradiated by high-energy particles of light called photons. Electrons absorb these high-energy photons and escape the water molecule. This leaves a positively charged water molecule. Protons or positively charged hydrogen atoms from this charged water molecule then jump to another water molecule. The result is the formation of hydroxyl (OH) radicals. This sequence of events happens in a trillionth of a second. It causes ultrafast structural changes and the redistribution of energy among neighboring water molecules. Scientists can capture this avalanche of ionization events in real time by putting together a series of high-resolution snapshots taken using pulses of high energy probing electron beams.
The Impact
The proton-transfer and ionization process in water leads to the formation of a hydroxyl-hydronium [OH(H3O+)] complex. This complex is important for many applications in biomedicine, chemistry, and engineering. These include radiotherapy, photon science, space travel, water disinfection, and nuclear engineering. In addition, OH radicals are common in space. Understanding their formation may therefore shed light on the origin of life. Despite the importance of these processes, scientists have been unable to measure them. Recent developments in liquid phase ultrafast electron diffraction (UED) techniques make them useful for understanding these processes in water.
Summary
Researchers probed ultrafast physio-chemical processes initiated by laser irradiation in liquid water using a novel liquid-phase ultrafast electron diffraction technique in a unique mega-electron volt ultrafast electron diffraction (MeV-UED) facility in the Linac Coherent Light Source (LCLS). The LCLS is a Department of Energy (DOE) Office of Science user facility at the SLAC National Accelerator Laboratory. The team included researchers from SLAC, Stanford University, and the University of Nebraska-Lincoln. The experiment required the creation of a thin (100-nanometer) liquid water jet, enabling the measurement of bonds (a few Angstroms) between oxygen atoms and oxygen and hydrogen atoms. After a short-pulse laser ionization, the ionized liquid water undergoes ultrafast proton transfer to form the OH(H3O+) complex within 140 femto-seconds. This radical-cation complex dissociates into individual OH and H3O+within 250 femto-seconds. The method uses MeV-UED to directly measure the inter-molecular O··O and O··H bond structures after short-pulse laser ionization in liquid water. This spatiotemporal resolution enables measurements of ultrafast structural rearrangement of molecules due to the formation of unstable complexes and energy redistribution. The researchers confirmed this ultrafast structural relaxation observed from these measurements using high-level time-dependent density functional theory and molecular-dynamics calculations. Real-time imaging of this unstable complex using MeV-UED can be crucial for fundamental science and numerous applications, ranging from radiotherapy, nuclear engineering, water disinfection, space-travel, and the search for extraterrestrial life.
Contact
Matthias Ihme
Stanford University, Department of Mechanical Engineering
SLAC National Accelerator Laboratory
[email protected]
Xijie Wang1
SLAC National Accelerator Laboratory
[email protected]
Funding
This research was supported by the Department of Energy (DOE) Office of Science, Office of Basic Energy Sciences, the Laboratory Directed Research and Development program at SLAC, the DFG Mercator Fellowship, and the National Energy Research Scientific Computing Center (NERSC), a DOE Office of Science user facility.
Publications
Lin, M.-F., et al., Imaging the short-lived hydroxyl-hydronium pair in ionized liquid water. Science 374, 92-95 (2021). [DOI: 10.1126/science.abg3091]
Related Links
Scientists capture the fleeting transition of water into a highly reactive state, SLAC Office of Communications
Scraped from https://www.sourcearu.com